Advancing Optical Material Precursors: Selective Exo-Norbornene Derivative Manufacturing via Palladium Catalysis
The landscape of advanced optical materials is undergoing a significant transformation, driven by the demand for higher performance cyclic olefin polymers (COP) and cyclic olefin copolymers (COC). At the heart of this evolution lies the precise synthesis of norbornene derivatives, specifically those with controlled stereochemistry. Patent CN101778809B introduces a groundbreaking methodology for the production of norbornene derivatives where the substituent at the 5-position adopts an exclusive exo configuration. This technical breakthrough addresses a longstanding bottleneck in the manufacturing of optical films, phase difference membranes, and semiconductor sealing materials. By leveraging a palladium-catalyzed reductive Heck reaction, this process bypasses the thermodynamic limitations of traditional cycloaddition routes, offering a pathway to high-purity intermediates essential for next-generation display technologies. For R&D leaders and procurement strategists in the electronic chemicals sector, understanding this shift from non-selective thermal reactions to catalytic precision is crucial for securing supply chains capable of meeting the rigorous specifications of modern optoelectronics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-substituted norbornene derivatives has relied heavily on the Diels-Alder reaction between cyclopentadiene derivatives and styrene-based olefins. While chemically straightforward, this thermal cycloaddition suffers from inherent stereochemical ambiguity. As illustrated in the reaction scheme below, the Diels-Alder reaction typically yields a mixture of endo and exo isomers, often with a ratio favoring the endo form due to secondary orbital interactions. This lack of selectivity creates a massive downstream burden; separating these stereoisomers is notoriously difficult because they possess similar physical properties, often requiring energy-intensive fractional distillation or preparative HPLC, which drastically reduces overall process yield. Furthermore, attempts to isomerize the unwanted endo fraction into the desired exo form using acid or base catalysts have proven largely ineffective due to the comparable thermodynamic stability of both isomers. Consequently, manufacturers relying on this legacy technology face inconsistent batch quality and inflated production costs, limiting their ability to scale high-purity optical grade monomers efficiently.
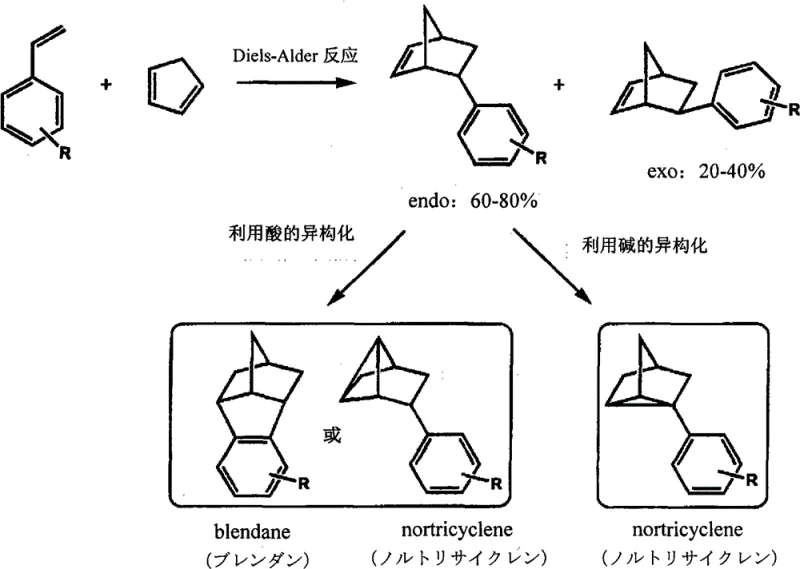
The Novel Approach
In stark contrast to the thermal limitations of the past, the methodology disclosed in CN101778809B utilizes a transition metal-catalyzed strategy to achieve unprecedented stereocontrol. By reacting a norbornadiene derivative with an aryl bromide in the presence of a specific palladium catalyst and phosphine ligand system, the process selectively constructs the carbon-carbon bond to favor the exo configuration exclusively. This reductive Heck-type coupling avoids the concerted mechanism of the Diels-Alder reaction, instead proceeding through a stepwise organometallic cycle that is kinetically controlled by the steric bulk of the ligand. The result is a reaction profile that suppresses the formation of the endo isomer almost entirely, as demonstrated by the high selectivity observed in experimental data. This shift allows producers to bypass complex purification trains, directly accessing the target exo-norbornene derivative with high conversion rates. For the supply chain, this means a more robust and predictable manufacturing process that is less susceptible to the variability inherent in thermal equilibrium reactions.
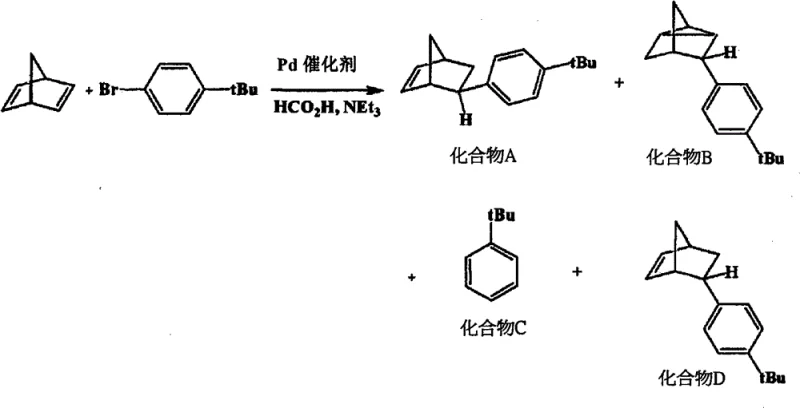
Mechanistic Insights into Pd-Catalyzed Reductive Heck Coupling
The core of this innovation lies in the intricate interplay between the palladium center and the specialized phosphine ligands defined in general formulas (1) and (2) of the patent. The reaction initiates with the oxidative addition of the aryl bromide to the Pd(0) species, forming an aryl-palladium(II) intermediate. Crucially, the choice of ligand—such as tri-tert-butylphosphine or bulky biaryl phosphines—creates a steric environment that directs the subsequent insertion of the norbornadiene double bond. Unlike standard Heck reactions that eliminate to form a diene, this process employs a reducing agent, typically formic acid or a formate salt, to intercept the alkyl-palladium intermediate. This hydride transfer facilitates reductive elimination, releasing the saturated exo-5-aryl-norbornene product and regenerating the active Pd(0) catalyst. The presence of a base, such as potassium hydroxide or carbonate, is also critical to neutralize the hydrogen bromide byproduct and maintain the catalytic cycle's efficiency. This mechanistic pathway effectively decouples the stereochemical outcome from thermodynamic equilibrium, allowing kinetic control to dictate the exclusive formation of the exo isomer.
From an impurity control perspective, this catalytic system offers distinct advantages over radical or thermal pathways. The specificity of the palladium-ligand complex minimizes side reactions such as polymerization of the norbornadiene or homocoupling of the aryl bromide. The patent data indicates that by optimizing the molar ratio of the base and reducing agent, the formation of byproducts like tricyclic compounds or reduced arenes can be suppressed to negligible levels. This high level of chemoselectivity is vital for electronic applications where trace metallic or organic impurities can degrade the optical transparency or electrical insulation properties of the final polymer. The ability to tune the ligand structure further allows for adaptation to various substituted aryl bromides, providing a versatile platform for synthesizing a wide range of functionalized norbornene monomers tailored for specific refractive index or glass transition temperature requirements.
How to Synthesize Exo-Norbornene Derivatives Efficiently
Implementing this catalytic protocol requires careful attention to reaction conditions to maximize the benefits of the exo-selective pathway. The process generally involves charging a reactor with the norbornadiene substrate, the aryl bromide coupling partner, and the palladium catalyst system under an inert atmosphere. The addition of a reducing agent and a base is timed to coincide with the heating phase, typically maintained between 80°C and 120°C to ensure optimal turnover without degrading the sensitive organometallic species. Following the reaction, standard workup procedures involving extraction and distillation yield the high-purity product. The detailed standardized synthesis steps for this specific transformation are outlined in the guide below.
- Prepare the reaction system by combining a norbornadiene derivative, a bromine compound (aryl bromide), a palladium source, and a specific phosphine ligand in a suitable solvent.
- Add a reducing agent such as formic acid or a formate salt, along with a base like potassium hydroxide or carbonate, to facilitate the reductive Heck coupling.
- Heat the mixture to 80-120°C under inert atmosphere, then isolate the exo-selective product through distillation or chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Pd-catalyzed synthesis route represents a strategic opportunity to optimize cost structures and enhance supply reliability. The primary economic driver is the elimination of isomer separation steps. In traditional manufacturing, a significant portion of raw material value is lost during the purification of endo/exo mixtures, often discarding the unwanted isomer or subjecting it to low-yield recycling processes. By selectively producing the exo isomer directly, this technology drastically improves atom economy and reduces the consumption of solvents and energy associated with extensive purification. This efficiency translates into substantial cost savings in electronic chemical manufacturing, allowing for more competitive pricing of high-performance optical resins without compromising margin.
- Cost Reduction in Manufacturing: The streamlined process eliminates the need for complex chromatographic separations or multi-stage distillations required to isolate exo isomers from Diels-Alder mixtures. By removing these unit operations, manufacturers can significantly reduce capital expenditure on purification equipment and lower operational costs related to energy and solvent recovery. Furthermore, the high conversion rates achieved with the palladium catalyst minimize raw material waste, ensuring that expensive aryl bromide precursors are utilized effectively. This lean manufacturing approach supports a more sustainable cost model, particularly important when scaling production for large-volume applications like LCD panels.
- Enhanced Supply Chain Reliability: Relying on a catalytic process with high selectivity reduces the risk of batch-to-batch variability that often plagues thermal cycloadditions. The robustness of the palladium-phosphine system ensures consistent product quality, which is critical for maintaining long-term contracts with display manufacturers who require strict adherence to optical specifications. Additionally, the use of commercially available aryl bromides and norbornadiene derivatives simplifies the raw material sourcing strategy. This accessibility mitigates supply risks associated with specialized or hard-to-source intermediates, ensuring a continuous flow of materials even during market fluctuations.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are amenable to large-scale batch processing, utilizing standard stainless steel reactors common in fine chemical facilities. The ability to operate at moderate temperatures (80-120°C) reduces the thermal load on plant infrastructure compared to high-pressure Diels-Alder processes. Moreover, the improved selectivity leads to a cleaner waste stream with fewer organic byproducts, simplifying effluent treatment and helping facilities meet increasingly stringent environmental regulations. This scalability ensures that the technology can grow alongside market demand for advanced optical materials, from pilot batches to multi-ton annual production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this selective synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: Why is exo-selectivity critical for norbornene-based optical polymers?
A: Exo-isomers provide superior thermal stability and specific birefringence properties required for high-performance retardation films in LCDs, whereas endo-isomers often lead to crystallization issues and inferior optical clarity.
Q: How does this Pd-catalyzed method improve upon traditional Diels-Alder synthesis?
A: Traditional Diels-Alder reactions produce inseparable mixtures of endo and exo isomers. This novel reductive Heck approach selectively yields the exo-configuration, eliminating costly and yield-reducing separation steps.
Q: What are the key ligand requirements for this catalytic system?
A: The process requires bulky, electron-rich phosphine ligands, such as tri-tert-butylphosphine or specific biaryl phosphines, to stabilize the palladium center and enforce the steric environment necessary for exo-selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Exo-Norbornene Derivatives Supplier
As the demand for high-performance optical materials continues to surge, securing a supply partner with deep technical expertise in advanced catalytic synthesis is essential. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging cutting-edge methodologies like the Pd-catalyzed reductive Heck reaction to deliver superior exo-norbornene derivatives. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without sacrificing quality. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the exacting standards required for optical film and semiconductor applications.
We invite you to collaborate with us to optimize your material sourcing strategy. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. Contact us today to request specific COA data and route feasibility assessments, and discover how our advanced manufacturing capabilities can drive efficiency and innovation in your supply chain.
