Advanced Pd-Catalyzed Synthesis of Chiral 1,3-Disubstituted-3-Arylpropene Compounds for Commercial Scale-Up
Advanced Pd-Catalyzed Synthesis of Chiral 1,3-Disubstituted-3-Arylpropene Compounds for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing chiral carbon-carbon bonds, particularly for the synthesis of non-steroidal anti-inflammatory drugs (NSAIDs) and their precursors. Patent CN103467228A introduces a groundbreaking preparation method for chiral 1,3-disubstituted-3-arylpropene compounds, utilizing a palladium-catalyzed allyl-aryl coupling reaction. This technology represents a significant leap forward in synthetic efficiency, enabling the direct use of chiral allyl alcohols and arylboronic acids to generate high-value intermediates with exceptional stereocontrol. For R&D directors and process chemists, this patent offers a viable pathway to access complex chiral scaffolds that were previously difficult to synthesize without extensive protection-deprotection sequences or harsh reagents. The ability to transform readily available starting materials into pharmaceutically relevant structures under mild conditions addresses critical bottlenecks in the development of next-generation therapeutic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1,3-disubstituted-3-arylpropene skeletons has relied heavily on the use of highly reactive organometallic reagents such as aryl Grignard, aryl zinc, or aryl aluminum species. These conventional approaches suffer from severe practical limitations, including the necessity for stringent anhydrous and anaerobic conditions, which drastically increase operational costs and safety risks in a manufacturing environment. Furthermore, the requirement to pre-prepare these sensitive nucleophiles adds extra synthetic steps, reducing the overall atom economy and generating substantial amounts of metal-containing waste that requires costly disposal protocols. Additionally, many traditional methods lack the necessary stereospecificity, often yielding racemic mixtures that necessitate expensive and yield-loss-inducing resolution steps to obtain the single enantiomer required for biological activity. The reliance on harsh reaction conditions also limits the substrate scope, preventing the incorporation of sensitive functional groups that are increasingly common in modern drug design.
The Novel Approach
In stark contrast to these legacy techniques, the methodology disclosed in CN103467228A utilizes a palladium-complex catalyzed coupling between chiral allyl alcohols and arylboronic acids, bypassing the need for pre-activated electrophiles or aggressive nucleophiles. 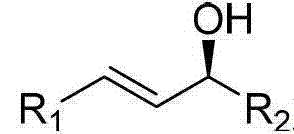 This innovative strategy leverages the hydroxyl group of the allyl alcohol directly as a leaving group, activated in situ by the interaction with the boronic acid and the palladium catalyst.
This innovative strategy leverages the hydroxyl group of the allyl alcohol directly as a leaving group, activated in situ by the interaction with the boronic acid and the palladium catalyst. 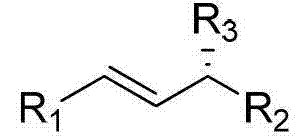 The reaction proceeds efficiently in common organic solvents such as toluene or tetrahydrofuran at moderate temperatures ranging from 20°C to 80°C. This shift not only simplifies the operational workflow by eliminating the need for cryogenic conditions or strict moisture exclusion but also significantly enhances the safety profile of the process. By employing stable and commercially abundant arylboronic acids, the method ensures a reliable supply chain for raw materials, making it an attractive option for large-scale industrial applications where consistency and safety are paramount.
The reaction proceeds efficiently in common organic solvents such as toluene or tetrahydrofuran at moderate temperatures ranging from 20°C to 80°C. This shift not only simplifies the operational workflow by eliminating the need for cryogenic conditions or strict moisture exclusion but also significantly enhances the safety profile of the process. By employing stable and commercially abundant arylboronic acids, the method ensures a reliable supply chain for raw materials, making it an attractive option for large-scale industrial applications where consistency and safety are paramount.
Mechanistic Insights into Pd-Catalyzed Allyl-Aryl Coupling
The core of this technological advancement lies in the sophisticated catalytic cycle mediated by the palladium-phosphine complex. The reaction initiates with the coordination of the palladium catalyst to the chiral allyl alcohol, facilitated by the presence of the arylboronic acid which assists in activating the hydroxyl moiety. This activation allows for an oxidative addition step that generates a crucial pi-allyl palladium intermediate, a species that retains the stereochemical information of the starting material. Subsequently, a transmetallation event occurs where the aryl group from the boronic acid is transferred to the palladium center. The cycle concludes with a reductive elimination step that forms the new carbon-carbon bond, releasing the chiral 1,3-disubstituted-3-arylpropene product and regenerating the active palladium species. Crucially, this mechanism is stereospecific, typically proceeding with inversion of configuration, which allows chemists to predictably access either enantiomer simply by selecting the appropriate enantiomer of the starting allyl alcohol. 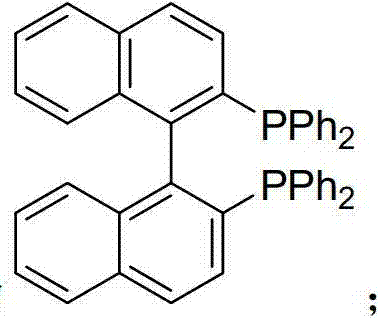 The choice of ligand, such as the bisphosphine ligands depicted in the patent examples like rac-BINAP or DPPB, plays a pivotal role in stabilizing the palladium intermediate and ensuring high turnover numbers, thereby minimizing the residual metal content in the final API intermediate.
The choice of ligand, such as the bisphosphine ligands depicted in the patent examples like rac-BINAP or DPPB, plays a pivotal role in stabilizing the palladium intermediate and ensuring high turnover numbers, thereby minimizing the residual metal content in the final API intermediate.
From an impurity control perspective, this mechanism offers distinct advantages over traditional cross-coupling reactions. Because the reaction does not require strong bases, side reactions such as base-mediated elimination or epimerization of sensitive chiral centers are effectively suppressed. The mild conditions also prevent the decomposition of thermally labile functional groups, resulting in a cleaner crude reaction profile that simplifies downstream purification. The high stereospecificity ensures that the formation of diastereomeric impurities is minimized, which is critical for meeting the stringent purity specifications demanded by regulatory agencies for pharmaceutical ingredients. Furthermore, the use of arylboronic acids, which are generally stable to air and water, reduces the risk of forming hazardous byproducts associated with the hydrolysis of more reactive organometallic reagents, thus enhancing the overall environmental footprint of the synthesis.
How to Synthesize Chiral 1,3-Disubstituted-3-Arylpropene Efficiently
The implementation of this synthesis route in a laboratory or pilot plant setting follows a streamlined protocol designed for maximum efficiency and reproducibility. The process begins with the precise weighing of the palladium source, such as Pd2(dba)3CHCl3, and the selected phosphine ligand, which are dissolved in the chosen organic solvent to form the active catalytic species. To this mixture, the chiral allyl alcohol substrate and the arylboronic acid coupling partner are added in a specific molar ratio optimized to drive the reaction to completion while minimizing excess reagent costs. The reaction vessel is then heated to the prescribed temperature, typically between 50°C and 80°C, and maintained for a duration of 4 to 48 hours depending on the specific substrate reactivity. Detailed standard operating procedures for scaling this reaction from gram to kilogram scale are available upon request, ensuring that the transition from discovery to manufacturing is seamless and compliant with GMP standards.
- Prepare the catalytic system by mixing a palladium source such as Pd2(dba)3CHCl3 with a chiral or achiral phosphine ligand like rac-BINAP in an organic solvent.
- Add the chiral allyl alcohol substrate and the arylboronic acid coupling partner to the reaction mixture and heat to temperatures between 20°C and 80°C.
- Monitor the reaction progress, then remove the solvent under reduced pressure and purify the resulting chiral product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the raw material portfolio; by utilizing stable arylboronic acids and readily synthesizable allyl alcohols, companies can reduce their dependency on specialized, hazardous reagents that often suffer from volatile pricing and long lead times. This shift to more commoditized starting materials enhances supply chain resilience, ensuring continuous production even during market fluctuations. Moreover, the elimination of strong bases and the reduction in reaction severity lower the requirements for specialized corrosion-resistant equipment, allowing for the utilization of standard glass-lined or stainless steel reactors commonly found in existing manufacturing facilities. This compatibility significantly reduces capital expenditure requirements for process implementation.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the high atom economy and the reduction in unit operations. By avoiding the pre-preparation of organometallic reagents and eliminating the need for cryogenic cooling or extensive drying of solvents, the energy consumption per kilogram of product is substantially lowered. The high yields and stereospecificity observed in the patent examples mean that less raw material is wasted on off-spec byproducts, directly improving the cost of goods sold (COGS). Additionally, the simplified workup procedure, which often involves simple solvent evaporation and chromatography rather than complex aqueous quenches and extractions, reduces labor costs and solvent usage, contributing to a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply chain stability. Since the reagents are not sensitive to air or moisture, the logistics of transporting and storing raw materials become less complex and costly. There is no need for specialized containers or inert atmosphere handling during the charging of reactors, which minimizes the risk of batch failures due to operator error or equipment leakage. This reliability ensures consistent output quality and quantity, allowing supply chain planners to maintain tighter inventory controls and meet delivery commitments to downstream API manufacturers with greater confidence. The broad substrate scope also means that alternative suppliers for specific arylboronic acids can be sourced easily without requiring extensive re-validation of the process.
- Scalability and Environmental Compliance: Scaling this reaction to commercial volumes is facilitated by its exothermic profile and lack of hazardous gas evolution. The absence of strong bases and toxic heavy metal reagents (other than the catalytic amount of palladium which can be recovered) simplifies waste treatment protocols, aligning with increasingly stringent environmental regulations. The process generates significantly less saline waste compared to traditional methods, reducing the burden on wastewater treatment facilities. This environmental compliance not only mitigates regulatory risk but also enhances the corporate sustainability profile, a factor that is becoming increasingly important for multinational pharmaceutical clients evaluating their supplier base for green chemistry credentials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed coupling technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to existing industry standards. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their current production pipelines. For further technical discussions or custom feasibility studies, our team is prepared to provide detailed data packages.
Q: What are the advantages of using unactivated allyl alcohols in this coupling reaction?
A: Using unactivated allyl alcohols directly eliminates the need for pre-functionalization steps required for traditional electrophiles like halides or esters. This approach significantly improves atom economy and reduces waste generation, as the hydroxyl group serves as the leaving group activated in situ by the boronic acid and palladium catalyst.
Q: How does this method ensure high stereoselectivity for chiral drug intermediates?
A: The reaction proceeds through a stereospecific mechanism where the stereochemistry of the product is strictly determined by the stereochemistry of the starting chiral allyl alcohol. The process typically results in chirality inversion, allowing for predictable synthesis of specific enantiomers essential for pharmaceutical applications like Naproxen production.
Q: Does this process require harsh reaction conditions or strong bases?
A: No, unlike conventional methods that often require strong bases or harsh nucleophiles like Grignard reagents, this patented method operates under mild conditions (20°C to 80°C) without the addition of external alkali. This tolerance simplifies the workup procedure and enhances compatibility with sensitive functional groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 1,3-Disubstituted-3-Arylpropene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality chiral intermediates in the development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is managed with precision and care. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify enantiomeric excess and chemical purity at every stage of the manufacturing process. Our facility is equipped to handle the specific requirements of palladium-catalyzed reactions, including efficient metal scavenging technologies to ensure residual metal levels are well below regulatory limits.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this greener, more efficient route. We encourage you to contact us today to obtain specific COA data for our reference standards and to schedule a consultation for route feasibility assessments, ensuring your supply chain is built on a foundation of innovation and reliability.
