Advanced Silver-Free Synthesis of Tetraammine Platinum Salts for Industrial Catalysis
The landscape of platinum coordination chemistry is undergoing a significant transformation driven by the urgent need for greener, more cost-effective synthetic routes in the fine chemical and catalysis sectors. A pivotal advancement in this domain is detailed in patent CN115010193A, which discloses a novel preparation method for tetraazaplatinum(II) acid salts, including critical derivatives like tetraammine platinum nitrate and acetate. This technology represents a paradigm shift away from the historically dominant, silver-dependent methodologies that have plagued the industry with high costs and complex waste streams. By leveraging a two-step one-pot reaction utilizing platinum dioxide hydrate and hydrazine reduction, this innovation addresses the longstanding challenges of chlorine contamination and low overall yields. For R&D directors and procurement strategists alike, understanding the mechanistic underpinnings and commercial implications of this patent is essential for securing a competitive edge in the supply of high-purity platinum intermediates. The following analysis dissects the technical superiority of this silver-free approach and its potential to redefine supply chain reliability for global manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tetraammine platinum salts has been inextricably linked to the use of expensive silver salts, a dependency that creates substantial bottlenecks in both cost efficiency and environmental compliance. Traditional pathways, such as those cited in CN101367557A and CN103113412A, typically commence with the preparation of dichlorotetraammine platinum or platinum dichloride, necessitating a subsequent reaction with silver nitrate or silver acetate to precipitate chloride ions. This reliance on silver introduces a dual burden: the exorbitant cost of the silver reagent itself and the generation of massive quantities of silver chloride waste sludge. Furthermore, these multi-step processes often suffer from cumulative yield losses, with comprehensive calculations indicating an overall yield of merely 50% when starting from sponge platinum. The physical entrapment of product within insoluble by-products like silver chloride or calcium oxalate further complicates purification, leading to products with residual chlorine levels that often exceed 100ppm, a critical failure for applications requiring ultra-low halogen content.
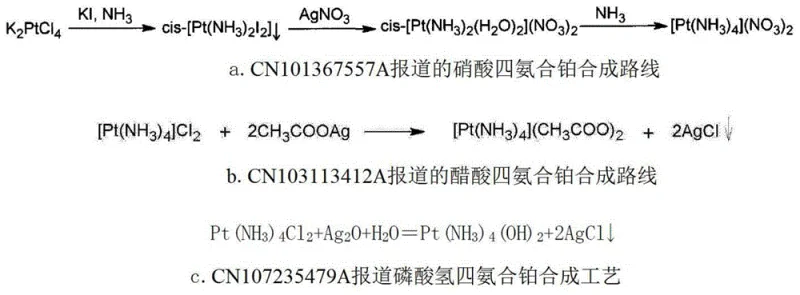
The Novel Approach
In stark contrast to the cumbersome legacy protocols, the methodology outlined in CN115010193A introduces a streamlined, silver-free trajectory that fundamentally alters the economic and operational profile of tetraammine platinum production. This innovative route utilizes hydrated platinum dioxide, a +4 valence platinum source, as the starting material, thereby bypassing the need for chloroplatinic acid and the associated chloride removal steps. The process employs a quantitative reduction strategy using hydrazine hydrate to convert the intermediate +4 nitrogen-containing platinum complex directly into the target +2 valence complex. By eliminating the silver precipitation step entirely, the new method avoids the formation of difficult-to-separate silver chloride waste, resulting in a much cleaner reaction profile. The operational simplicity is further enhanced by the ability to conduct the reaction under atmospheric pressure with straightforward temperature controls, facilitating a comprehensive yield of approximately 90% and product purity exceeding 99%, which stands as a testament to the robustness of this modern synthetic design.
Mechanistic Insights into Hydrazine-Mediated Reduction of Platinum Complexes
The core chemical innovation driving this process lies in the precise control of platinum oxidation states and ligand coordination within a unified reaction vessel. The mechanism initiates with the coordination of nitrogen-containing ligands, such as ammonia or ethylenediamine, to the +4 valent platinum center of the hydrated platinum dioxide. This step forms a stable intermediate complex, represented generally as [Pt(NR)m(OH)n](X)j, where the stoichiometry is carefully balanced to ensure complete solubilization of the platinum source. The subsequent addition of hydrazine hydrate acts as a potent reducing agent, facilitating the transfer of electrons to the platinum center. This reduction event triggers the release of nitrogen gas and water as benign by-products, while simultaneously stabilizing the platinum in the desired +2 oxidation state coordinated by four nitrogen atoms. The elegance of this mechanism is its atom economy; unlike silver salt metathesis which generates stoichiometric amounts of solid waste, this redox process generates volatile or soluble by-products that do not interfere with product isolation.
From an impurity control perspective, this mechanism offers a decisive advantage in managing halogen content, a critical parameter for catalyst longevity and performance. Traditional methods relying on chloroplatinic acid inherently introduce chloride ions that are notoriously difficult to remove completely, often requiring multiple recrystallization steps that erode yield. In the hydrazine reduction pathway, the absence of chloride in the primary starting materials means that the final product naturally exhibits chlorine levels below 20ppm, as verified by elemental analysis in the patent examples. This ultra-low halogen profile is achieved without the need for aggressive ion-exchange resins or extensive washing protocols, which can sometimes lead to product degradation. The result is a high-purity tetraammine platinum salt that is ideally suited for sensitive applications in petrochemical catalysis and pharmaceutical intermediate synthesis, where trace metal impurities can poison active sites or compromise downstream reaction fidelity.
How to Synthesize Tetraammine Platinum Salts Efficiently
The practical implementation of this synthesis route is designed for scalability and reproducibility, making it an attractive option for industrial adoption. The process is characterized by a two-step sequence that can be executed in a single reactor, minimizing equipment footprint and handling time. Operators begin by dissolving the appropriate ammonium salt and nitrogen ligand in distilled water, followed by the addition of hydrated platinum dioxide under heated stirring conditions until a clear solution is obtained. This homogeneity is crucial for ensuring uniform reduction in the subsequent step. Once the intermediate is formed, the solution is diluted and treated with a controlled addition of hydrazine hydrate, where temperature management plays a key role in preventing runaway reactions while ensuring complete conversion. The detailed standardized operating procedures for this synthesis are provided in the guide below.
- Dissolve ammonium salt and nitrogen ligand in water, add hydrated platinum dioxide, and react at 50-90°C until clear.
- Dilute the concentrated solution and slowly add hydrazine hydrate reducing agent at 60°C.
- Heat to boil, concentrate until solid precipitates, filter, wash with ethanol, and vacuum dry to obtain the target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this silver-free synthesis protocol offers profound strategic benefits that extend far beyond simple raw material substitution. The most immediate impact is observed in the drastic reduction of direct material costs, as the elimination of silver nitrate and silver acetate removes one of the most expensive line items from the bill of materials. Silver prices are notoriously volatile and subject to speculative market forces, creating budget uncertainty for manufacturers; by decoupling production from silver dependency, companies can achieve significantly more stable and predictable cost structures. Furthermore, the simplification of the workflow from a multi-step sequence involving filtration of heavy metal sludges to a streamlined one-pot process reduces labor hours and energy consumption. This operational efficiency translates into substantial cost savings in manufacturing overhead, allowing for more competitive pricing in the global marketplace without compromising margin integrity.
- Cost Reduction in Manufacturing: The removal of silver salts from the process equation eliminates the need for purchasing high-value precious metal reagents that offer no value in the final product. Additionally, the avoidance of silver chloride waste generation removes the costly burden of hazardous waste disposal and the complex logistics associated with recovering silver from sludge. This dual saving mechanism—reduced input costs and reduced waste management expenses—creates a powerful economic incentive for adopting this technology. The higher overall yield of approximately 90% compared to the 50% of traditional methods means that less platinum feedstock is required to produce the same amount of finished goods, further amplifying the return on investment for every kilogram of platinum purchased.
- Enhanced Supply Chain Reliability: Relying on silver salts introduces a single point of failure in the supply chain, as silver availability can be constrained by mining outputs and geopolitical factors. By shifting to a process based on hydrated platinum dioxide and hydrazine, manufacturers diversify their supplier base to include more abundant and stable chemical commodities. The simplified process flow also reduces the risk of production delays caused by equipment fouling or filtration bottlenecks common in silver precipitation steps. This increased robustness ensures a more consistent delivery schedule for downstream customers, enhancing the reputation of the supplier as a reliable partner capable of meeting just-in-time delivery requirements even during periods of market volatility.
- Scalability and Environmental Compliance: The environmental footprint of this new method is markedly smaller, aligning with increasingly stringent global regulations regarding heavy metal discharge. The absence of silver-containing wastewater simplifies effluent treatment processes, reducing the capital expenditure required for compliance infrastructure. Moreover, the reaction conditions—operating at atmospheric pressure and moderate temperatures—are inherently safer and easier to scale from pilot plant to full commercial production. This ease of scale-up allows manufacturers to respond rapidly to surges in demand for tetraammine platinum salts, ensuring that supply can be expanded quickly to capture market opportunities without the need for extensive new facility construction or complex safety certifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into existing production lines. The clarity provided here aims to resolve uncertainties regarding product quality, process safety, and economic viability.
Q: How does this new method reduce chlorine content compared to traditional routes?
A: By starting with platinum dioxide hydrate instead of chloroplatinic acid and avoiding silver chloride precipitation, the process achieves chlorine levels below 20ppm, significantly lower than the 100ppm limit of older methods.
Q: What are the economic benefits of eliminating silver salts in platinum synthesis?
A: Eliminating expensive silver nitrate or silver acetate removes a major raw material cost and avoids the generation of silver chloride waste, which simplifies purification and reduces environmental disposal costs.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process operates at atmospheric pressure with simple temperature control and uses common reagents like hydrazine hydrate, making it highly suitable for large-scale commercial manufacturing with yields around 90%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetraammine Platinum Salts Supplier
The technological breakthroughs presented in CN115010193A underscore the immense potential for optimizing the production of critical platinum intermediates, yet realizing this potential requires a partner with deep technical expertise and proven manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that ensure every batch meets the exacting standards required for high-performance catalysis and pharmaceutical applications. We understand that consistency is key in fine chemical manufacturing, and our state-of-the-art facilities are designed to deliver the high-purity tetraammine platinum salts your operations demand.
We invite you to engage with our technical procurement team to discuss how we can tailor our manufacturing solutions to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages of switching to our optimized supply chain. We encourage you to contact us today to索取 specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of reliability, quality, and cost-efficiency. Let us be your trusted partner in navigating the complexities of platinum chemistry.
