Advanced Synthesis of Quinolonyl Lactam Antimicrobials via Organosilicon-Mediated Cyclization
The pharmaceutical landscape is constantly evolving with the demand for more potent broad-spectrum antimicrobial agents, and patent CN1159809A presents a significant breakthrough in the synthesis of quinolonyl lactam antimicrobials (QLA). This intellectual property discloses a sophisticated process for preparing compounds that combine a quinolone moiety with a lactam-containing moiety through a specialized linking group. The core innovation lies in a two-step methodology that first couples a quinolone precursor with a lactam compound to form a stable intermediate, followed by a crucial cyclization step mediated by an organosilicon compound. This approach effectively overcomes the historical challenges associated with synthesizing these complex hybrid molecules, particularly the degradation of products and the insolubility of components in standard organic solvents. By leveraging mild reaction conditions and organosilicon chemistry, the process ensures that sensitive functional groups within the substrate and product remain intact, offering a robust pathway for generating high-purity antimicrobial intermediates suitable for further pharmaceutical development.
Traditional methods for ligating quinolone and lactam moieties have long been plagued by significant technical limitations that hinder efficient commercial production. Conventional ligation techniques often rely on harsh reagents and polar solvents, such as water, which can lead to the degradation of the final product and poor solubility of the quinolone or related heterocyclic components. These adverse conditions frequently result in low overall yields and the formation of complex impurity profiles that are difficult to purify on a large scale. Furthermore, the use of strong bases for ring closure in prior art processes can compromise the stability of the beta-lactam ring, a critical pharmacophore responsible for the antimicrobial activity. The inability to maintain the integrity of sensitive functional groups during synthesis has historically restricted the structural diversity of QLA compounds that could be practically manufactured, limiting the exploration of potentially superior therapeutic candidates.
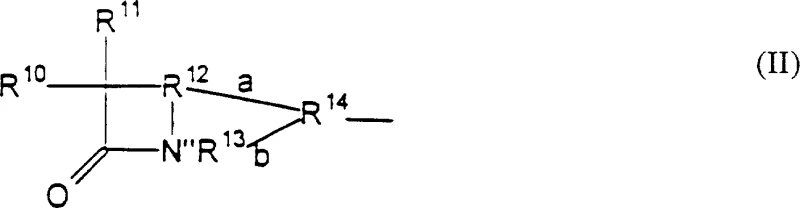
The novel approach detailed in the patent data introduces a paradigm shift by utilizing an organosilicon compound during the linking and cyclization steps, which fundamentally alters the reaction environment to favor product stability. This method allows for the surprisingly efficient synthesis of QLA precursors under reaction conditions where the solubility of the quinolone precursor is optimized without resorting to harsh polar solvents. The mild nature of these reaction conditions means that sensitive functional groups in the reaction substrate and product can withstand the synthesis process without degradation. In addition, these methods are particularly useful when used in conjunction with quinolone and related heterocyclic moiety ring closure methods, providing more synthetic flexibility for the preparation of such molecules. If prepared according to the prior art, these molecules would typically only be achievable with low to moderate yields, but this new process significantly improves both yield and product purity, enabling the practical realization of complex dual-action antimicrobial structures.
Mechanistic Insights into Organosilicon-Mediated Cyclization
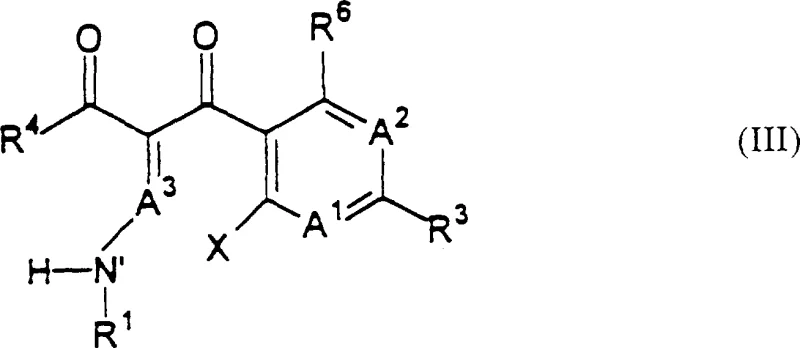
The mechanistic elegance of this process centers on the strategic use of organosilicon compounds to facilitate the formation of the quinolone ring system while preserving the integrity of the attached lactam moiety. The reaction begins with the coupling of a compound of Formula (III), which serves as a quinolone precursor, with a lactam-containing compound of Formula (II). This coupling step forms an intermediate compound where the two pharmacophores are linked but the quinolone ring is not yet fully closed or aromatized in its final active form. The subsequent cyclization step involves reacting this intermediate with an organosilicon compound, such as chlorotrimethylsilane or hexamethyldisilazane, which acts as a silylating agent. This reagent promotes the intramolecular cyclization required to form the quinolone nucleus under mild thermal conditions, typically ranging from about 0°C to 110°C, avoiding the extreme temperatures and strong bases that would otherwise hydrolyze the sensitive beta-lactam ring. The organosilicon species likely activates specific oxygen or nitrogen atoms within the precursor, facilitating nucleophilic attack and ring closure without generating the highly basic environments that are detrimental to lactam stability.
Impurity control is inherently built into this mechanism through the avoidance of degradation pathways common in traditional synthesis. By eliminating the need for harsh polar solvents and strong bases, the process minimizes the formation of hydrolysis byproducts that often arise from the opening of the beta-lactam ring or the decomposition of the quinolone core. The use of protecting groups for carboxyl, hydroxyl, and amino functions prior to the coupling step further enhances purity by preventing unwanted competing side reactions during the ligation phase. These protecting groups, such as esters for carboxylic acids or silyl ethers for hydroxyls, are stable under the mild coupling conditions but can be readily removed in a final deprotection step after cyclization is complete. This orthogonal protection strategy ensures that the final antimicrobial compound is obtained with a clean impurity profile, reducing the burden on downstream purification processes and ensuring that the final active pharmaceutical ingredient meets stringent regulatory specifications for safety and efficacy.
How to Synthesize Quinolonyl Lactam Antimicrobials Efficiently
The synthesis of these advanced antimicrobial intermediates requires precise control over reaction parameters to maximize the benefits of the organosilicon-mediated pathway. The process generally involves dissolving the quinolone precursor and the lactam component in suitable organic solvents such as dichloromethane, tetrahydrofuran, or acetonitrile, ensuring that all reactants remain in solution throughout the transformation. Temperature control is critical, particularly during the coupling step where temperatures are preferably maintained below 0°C, and often between -20°C to -15°C, to prevent premature side reactions. Following the formation of the intermediate, the addition of the organosilicon reagent initiates the cyclization, which proceeds efficiently at slightly elevated temperatures up to 50°C. The detailed standardized synthesis steps for executing this protocol are outlined in the guide below.
- Couple a compound of Formula (III) with a lactam-containing compound of Formula (II) to form an intermediate compound, optionally using protecting groups.
- Cyclize the intermediate compound by reacting it with an organosilicon compound to form the final quinolonyl lactam structure.
- Perform deprotection steps if protecting groups were used to remove them and obtain the pharmaceutically active antimicrobial agent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain professionals, the adoption of this synthesis methodology offers distinct strategic advantages that translate directly into operational resilience and cost efficiency. The primary benefit stems from the significant reduction in manufacturing complexity achieved by replacing harsh, degradation-prone conditions with mild, controlled organosilicon chemistry. This shift eliminates the need for expensive and hazardous reagents that require specialized handling and disposal, thereby streamlining the production workflow. Furthermore, the improved solubility of intermediates in common organic solvents reduces the reliance on exotic solvent systems, simplifying solvent recovery and recycling operations. These factors collectively contribute to a more streamlined manufacturing process that is easier to manage and less prone to the variability that often plagues complex multi-step syntheses involving sensitive biological motifs.
- Cost Reduction in Manufacturing: The elimination of harsh reaction conditions and the associated need for extensive purification steps to remove degradation byproducts leads to substantial cost savings. By avoiding the use of strong bases and polar solvents that compromise yield, the process maximizes the conversion of raw materials into valuable product, reducing the effective cost per kilogram of the intermediate. Additionally, the mild conditions reduce the energy consumption required for heating and cooling cycles, and the compatibility with standard organic solvents lowers the cost of solvent procurement and waste treatment. The ability to use protecting groups that are easily introduced and removed further optimizes the material throughput, ensuring that the overall cost of goods sold is minimized without sacrificing quality.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route significantly enhances supply chain reliability by reducing the risk of batch failures due to product degradation. Traditional methods often suffer from inconsistent yields caused by the sensitivity of the beta-lactam ring to reaction conditions, leading to unpredictable production schedules. In contrast, the organosilicon-mediated process provides a more consistent and reproducible outcome, allowing for more accurate forecasting and inventory planning. The use of commercially available starting materials and reagents, such as standard organosilicon compounds and protected lactams, ensures that the supply of raw materials is stable and not subject to the bottlenecks often associated with specialized or custom-synthesized reagents. This reliability is crucial for maintaining continuous production lines and meeting the demanding delivery timelines of global pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of standard unit operations and common solvents that are well-understood in industrial settings. The mild reaction conditions reduce the safety risks associated with exothermic reactions or the handling of corrosive bases, making the process safer to operate at large volumes. From an environmental perspective, the reduction in waste generation due to higher yields and fewer purification steps aligns with green chemistry principles and regulatory requirements for sustainable manufacturing. The ability to recycle solvents and the reduced load on wastewater treatment systems due to the absence of harsh aqueous waste streams further enhance the environmental profile of the process. This compliance with environmental standards not only mitigates regulatory risk but also appeals to increasingly eco-conscious stakeholders in the pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry stakeholders. Understanding these details is essential for evaluating the feasibility of integrating this process into existing manufacturing frameworks or for sourcing these specific intermediates from qualified suppliers. The insights provided here clarify the scope of the technology and its practical implications for drug development and production.
Q: What are the advantages of using organosilicon compounds in this synthesis?
A: The use of organosilicon compounds allows for cyclization under milder reaction conditions compared to traditional strong base methods. This prevents the degradation of sensitive functional groups in both the quinolone and lactam moieties, resulting in higher product purity and yield.
Q: Which types of lactam moieties are compatible with this process?
A: The process is highly versatile and supports a wide range of beta-lactam structures, including penems, carbapenems, cephems, carbacephems, and monocyclic beta-lactams, allowing for the creation of diverse antimicrobial libraries.
Q: How does this method address solubility issues in quinolone synthesis?
A: Traditional methods often suffer from insolubility of quinolone precursors in organic solvents. This novel approach utilizes specific solvent systems and mild conditions that maintain good solubility of the quinolone precursor throughout the coupling and cyclization steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinolonyl Lactam Antimicrobial Intermediates Supplier
As the demand for next-generation antimicrobial agents continues to grow, the ability to reliably source high-quality intermediates is paramount for pharmaceutical developers. NINGBO INNO PHARMCHEM stands ready to support your projects with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team possesses deep expertise in handling sensitive beta-lactam and quinolone chemistries, ensuring that the stringent purity specifications required for clinical and commercial batches are consistently met. We utilize rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and purity of every lot, guaranteeing that the intermediates supplied meet the highest international standards for safety and efficacy.
We invite you to engage with our technical procurement team to discuss how we can tailor our manufacturing capabilities to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of how our optimized processes can reduce your overall development costs. We encourage potential partners to contact us to obtain specific COA data for our catalog items or to request route feasibility assessments for custom synthesis projects. Our commitment to transparency and technical excellence ensures that we are not just a vendor, but a strategic partner dedicated to accelerating your path to market with reliable, high-performance pharmaceutical intermediates.
