Advanced Enzymatic Resolution of Indoline-2-Carboxylic Acid for Commercial Scale-Up
Advanced Enzymatic Resolution of Indoline-2-Carboxylic Acid for Commercial Scale-Up
The pharmaceutical industry's relentless pursuit of high-purity chiral intermediates has found a significant breakthrough in the biocatalytic resolution of (S)-indoline-2-carboxylic acid, a critical scaffold for cardiovascular therapeutics like Perindopril. Patent CN110592045B details a sophisticated methodology employing a stereoselective recombinant esterase derived from Bacillus aryabhattai, engineered for expression in Escherichia coli. This technology addresses the longstanding challenges of achieving high enantiomeric excess while maintaining cost-effective manufacturing protocols. By leveraging genetic engineering to overexpress a specific carboxylesterase (SEQ ID NO. 1), the process facilitates the kinetic resolution of racemic (R,S)-indoline-2-ethyl formate with remarkable efficiency. The reported conversion rates hover around 45%, yielding the desired (S)-enantiomer with an optical purity consistently exceeding 96% e.e. This innovation represents a paradigm shift from traditional chemical synthesis, offering a reliable pharmaceutical intermediate supplier pathway that aligns with modern green chemistry principles and stringent regulatory requirements for API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically pure (S)-indoline-2-carboxylic acid has been plagued by cumbersome multi-step chemical sequences that impose significant burdens on both cost and environmental compliance. Traditional routes often rely on L-phenylalanine as a chiral source, necessitating a series of harsh transformations including nitration, bromination, and intramolecular cyclization, followed by nitro reduction and diazotization. These processes not only involve hazardous reagents but also generate substantial volumes of toxic waste, complicating downstream processing and waste management. Alternatively, resolution methods utilizing chiral resolving agents like (R)-alpha-phenylethylamine suffer from low theoretical yields (maximum 50%) and the high cost of recovering and recycling the resolving agent. Furthermore, chemical reduction methods involving tin and hydrochloric acid introduce heavy metal contamination risks, requiring extensive purification steps to meet pharmacopeial standards. The complexity of these legacy routes is visually represented in the intricate reaction schemes previously employed in the industry.
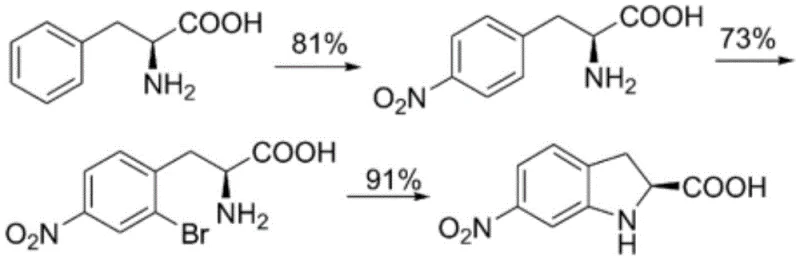
Another conventional approach involves the use of ortho-halo benzaldehydes condensed with acetic anhydride, followed by enzymatic aminolysis and cyclization. While this introduces a biocatalytic step, it still relies on complex chemical precursors and multiple isolation stages. The reliance on stoichiometric chiral auxiliaries or expensive transition metal catalysts in these older methodologies creates a bottleneck for cost reduction in API manufacturing, particularly when scaling to commercial volumes. The accumulation of impurities from side reactions in these harsh chemical environments further compromises the quality of the final product, often necessitating recrystallization steps that erode overall yield.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes a highly specific recombinant esterase to perform a direct kinetic resolution of the racemic ethyl ester substrate. This method bypasses the need for protecting groups, heavy metal reducers, or stoichiometric chiral resolving agents. The process operates under mild physiological conditions, typically at 30°C in a phosphate buffer system at pH 7.0, which preserves the integrity of the sensitive indoline ring structure. By employing whole-cell biocatalysis or purified enzyme preparations, the reaction achieves high stereoselectivity, effectively hydrolyzing the (S)-ester while leaving the (R)-ester intact. This selectivity eliminates the formation of difficult-to-remove diastereomeric impurities common in chemical resolution. The simplicity of the work-up procedure, involving merely pH adjustment and extraction, drastically simplifies the operational workflow. This streamlined protocol not only enhances the throughput of high-purity pharmaceutical intermediates but also significantly lowers the barrier to entry for manufacturers seeking to optimize their supply chains for cardiovascular drug components.
Mechanistic Insights into Recombinant Esterase Catalysis
The core of this technological advancement lies in the unique amino acid sequence of the stereoselective esterase (SEQ ID NO. 1), which exhibits a specialized active site architecture capable of distinguishing between the enantiomers of indoline-2-ethyl formate. The enzyme functions through a classic serine hydrolase mechanism, where a catalytic triad facilitates the nucleophilic attack on the carbonyl carbon of the ester bond. However, the steric constraints within the enzyme's binding pocket are exquisitely tuned to accommodate the (S)-configuration of the substrate while sterically hindering the (R)-enantiomer. This molecular recognition ensures that hydrolysis occurs almost exclusively on the desired isomer, leading to the observed high enantiomeric excess values of over 96%. The gene encoding this enzyme (SEQ ID NO. 2) was cloned from Bacillus aryabhattai and optimized for expression in E. coli BL21 using the pET-28a vector system, allowing for high-density fermentation and robust enzyme production. The use of a His-tag fusion protein further enables efficient purification via Ni-NTA affinity chromatography, ensuring that the biocatalyst itself meets high purity standards before being deployed in the resolution process.
From an impurity control perspective, the enzymatic nature of the reaction inherently minimizes side products. Unlike chemical hydrolysis which might proceed via non-selective acid or base catalysis leading to racemization or ring-opening byproducts, the enzyme maintains strict regio- and stereoselectivity. The reaction medium, being an aqueous buffer, prevents the solvation of non-polar impurities that often plague organic solvent-based syntheses. Furthermore, the mild pH conditions prevent the degradation of the indoline nucleus, which can be susceptible to oxidation or polymerization under acidic or basic stress. The result is a crude product profile that is significantly cleaner than that obtained from chemical routes, reducing the load on downstream purification units. This mechanistic precision translates directly into commercial value, as it reduces the number of unit operations required to achieve pharmaceutical grade purity, thereby enhancing the overall process mass intensity (PMI) of the manufacturing campaign.
How to Synthesize (S)-Indoline-2-Carboxylic Acid Efficiently
The implementation of this biocatalytic route requires a systematic approach to strain construction, fermentation, and biotransformation to ensure consistent quality and yield. The process begins with the precise cloning of the esterase gene followed by the optimization of induction conditions to maximize enzyme activity per gram of wet cell weight. Once the biocatalyst is prepared, the resolution reaction is conducted in a controlled aqueous environment where substrate concentration and temperature are tightly regulated to maintain enzyme stability. The following guide outlines the standardized operational framework derived from the patent examples, providing a roadmap for technical teams to replicate this high-efficiency synthesis.
- Clone the stereoselective esterase gene (SEQ ID NO. 2) from Bacillus aryabhattai into a pET-28a vector and transform into E. coli BL21.
- Culture the recombinant bacteria, induce expression with IPTG, and harvest wet cells or purify the enzyme via Ni-NTA chromatography.
- Perform kinetic resolution of racemic (R,S)-indoline-2-ethyl formate in phosphate buffer at 30°C to obtain (S)-indoline-2-carboxylic acid with >96% ee.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic resolution technology offers profound strategic advantages that extend beyond mere technical feasibility. The shift from multi-step chemical synthesis to a single-step biocatalytic resolution fundamentally alters the cost structure of producing (S)-indoline-2-carboxylic acid. By eliminating the need for expensive chiral resolving agents and hazardous reducing agents like tin chloride, the raw material costs are significantly reduced. Moreover, the avoidance of heavy metals simplifies the regulatory filing process and reduces the risk of supply chain disruptions caused by environmental compliance issues. The ability to use whole wet cells as the catalyst source further drives down costs by removing the need for expensive enzyme purification steps in large-scale applications, making the process economically viable for bulk manufacturing.
- Cost Reduction in Manufacturing: The elimination of stoichiometric chiral auxiliaries and heavy metal reagents removes major cost drivers associated with traditional synthesis. Since the maximum theoretical yield of a resolution is 50%, the ability to recover and recycle the unreacted (R)-ester or racemize it for reuse further enhances the economic efficiency of the process. The mild reaction conditions also reduce energy consumption related to heating and cooling, contributing to substantial cost savings in utility expenditures. Additionally, the simplified downstream processing reduces the consumption of organic solvents and chromatography media, lowering the overall variable cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: Relying on fermentation-derived enzymes provides a more stable and scalable supply source compared to extraction from natural sources or synthesis of complex chemical catalysts. The recombinant E. coli strain can be banked and scaled from shake flasks to industrial fermenters with predictable kinetics, ensuring consistent supply continuity. This biological manufacturing platform is less susceptible to the geopolitical and logistical volatility often associated with the sourcing of rare earth metals or specialized chemical reagents. The robustness of the enzyme under process conditions also means longer catalyst lifetimes and fewer batch failures, securing the production schedule against unexpected technical delays.
- Scalability and Environmental Compliance: The aqueous nature of the reaction system aligns perfectly with green chemistry initiatives, drastically reducing the generation of halogenated waste streams and heavy metal sludge. This environmental advantage simplifies the permitting process for new manufacturing facilities and reduces the liability associated with waste disposal. The process is inherently scalable; the parameters established in the patent (e.g., substrate loading of 25-50 g/L) can be directly translated to kiloliter-scale reactors without significant re-optimization. This scalability ensures that the technology can meet the growing global demand for cardiovascular medications without the need for building entirely new types of chemical infrastructure, leveraging existing fermentation capabilities instead.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented enzymatic resolution technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on performance metrics and operational feasibility. Understanding these details is crucial for technical teams evaluating the integration of this biocatalytic route into their existing manufacturing portfolios.
Q: What is the optical purity achieved by this enzymatic resolution method?
A: The patented process utilizing the recombinant esterase achieves an enantiomeric excess (e.e.) of over 96%, specifically reaching up to 97.16% in optimized examples, which is superior to wild-type bacterial strains.
Q: How does this biocatalytic route compare to traditional chemical synthesis?
A: Unlike traditional chemical routes that require hazardous reagents like tin/hydrogen chloride or complex multi-step chiral auxiliary syntheses, this enzymatic method operates under mild aqueous conditions (30°C, pH 7.0), significantly reducing environmental impact and purification complexity.
Q: Is the recombinant esterase suitable for large-scale industrial production?
A: Yes, the gene is expressed in E. coli BL21, a robust host for fermentation. The process allows for the use of whole wet cells or freeze-dried cells, facilitating easy scale-up from laboratory grams to metric ton quantities without the need for expensive cofactor regeneration systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-Indoline-2-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving cardiovascular medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced chiral HPLC and NMR techniques to verify enantiomeric excess and chemical identity. Our facility is equipped to handle the fermentation and downstream processing required for this enzymatic route, guaranteeing a consistent supply of this vital pharmaceutical building block.
We invite potential partners to engage with our technical procurement team to discuss how this advanced biocatalytic technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the specific economic benefits of switching to this greener, more efficient method. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring that your development programs proceed without interruption.
