Scalable Synthesis of 3,6-Dichloro-2-aminopyridine via Hofmann Degradation for Commercial API Production
Scalable Synthesis of 3,6-Dichloro-2-aminopyridine via Hofmann Degradation for Commercial API Production
The pharmaceutical and agrochemical industries constantly seek robust synthetic routes for halogenated heterocycles, particularly for complex intermediates like 3,6-dichloro-2-aminopyridine. A pivotal advancement in this domain is detailed in Chinese patent CN102875455A, which discloses a highly efficient synthesis method starting from 3,6-dichloro-2-pyridinecarboxylic acid. This innovation represents a significant departure from traditional lithiation-based strategies, offering a pathway that is not only chemically elegant but also commercially viable for large-scale operations. By leveraging a classic Hofmann degradation mechanism, the process circumvents the severe safety hazards and exorbitant costs associated with cryogenic organometallic chemistry. For R&D directors and procurement specialists alike, this technology signals a shift towards more sustainable and cost-effective manufacturing of S1P1 receptor agonist intermediates, ensuring a stable supply chain for downstream API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the state-of-the-art synthesis, as documented in patent WO2009154775A1, relied heavily on a challenging organolithium strategy. This conventional route begins with 6-chloro-2-aminopyridine, which must be protected and then subjected to directed ortho-lithiation using n-butyllithium at extremely low temperatures of -78°C. The reliance on n-butyllithium introduces substantial economic and safety burdens, as this reagent is not only expensive but also pyrophoric, requiring rigorous anhydrous and oxygen-free handling protocols. Furthermore, the cryogenic conditions demand specialized equipment and high energy consumption for cooling, drastically inflating operational expenditures. Perhaps most critically from a quality control perspective, slight deviations in temperature control during the lithiation step can lead to the formation of regioisomers, specifically 5,6-dichloro-2-aminopyridine, which are notoriously difficult to separate from the desired product, thereby compromising overall yield and purity.
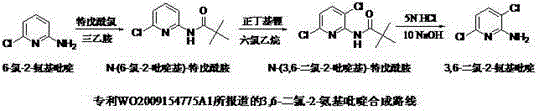
The Novel Approach
In stark contrast, the methodology presented in CN102875455A utilizes 3,6-dichloro-2-pyridinecarboxylic acid, a commercially abundant agrochemical intermediate known as Picloram acid, as the starting material. This strategic choice inherently solves the regioselectivity issue because the chlorine atoms are already positioned correctly on the pyridine ring before the amination step occurs. The process involves a straightforward acylation to form an acid chloride, followed by ammonolysis to generate the amide intermediate, and finally, a Hofmann degradation to convert the amide into the primary amine. This route operates under mild thermal conditions, typically between 5°C and 75°C, completely eliminating the need for cryogenic cooling. The transition from a lithiation-based approach to a degradation-based approach fundamentally alters the risk profile of the synthesis, making it accessible for standard chemical manufacturing facilities without the need for exotic infrastructure.
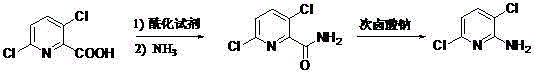
Mechanistic Insights into Hofmann Degradation and Impurity Control
The core chemical transformation in this novel process is the Hofmann degradation, a reaction that converts a primary amide into a primary amine with one fewer carbon atom, although in this specific heterocyclic context, it effectively replaces the carbonyl group with an amino group while retaining the ring structure integrity. The mechanism proceeds through the formation of an N-haloamide intermediate upon reaction with sodium hypohalite (generated in situ from bromine or chlorine and sodium hydroxide). Under basic conditions, this intermediate undergoes deprotonation and rearrangement to form an isocyanate, which is subsequently hydrolyzed to the amine. This mechanistic pathway is highly specific and does not involve the generation of reactive carbanions that could attack alternative positions on the pyridine ring. Consequently, the formation of the troublesome 5,6-dichloro isomer, which plagues the lithiation route due to non-selective metalation, is theoretically impossible in this degradation pathway. This intrinsic selectivity ensures that the crude product profile is significantly cleaner, reducing the burden on downstream purification steps.
From an impurity control standpoint, the use of 3,6-dichloro-2-pyridinecarboxylic acid as the precursor provides a robust defense against structural impurities. Since the starting material is a commodity chemical produced on a multi-ton scale for herbicide applications, its quality is well-established and consistent. The acylation step using thionyl chloride or phosphorus oxychloride is a standard, high-yielding transformation that generates gaseous byproducts (SO2, HCl) which are easily removed, driving the reaction to completion. The subsequent ammonolysis is performed at controlled low temperatures (5-25°C) to prevent side reactions, yielding the amide intermediate in over 96% crude yield without the need for intermediate purification. This telescoping capability allows the crude amide to be fed directly into the degradation step, minimizing material loss and solvent usage, which is a critical factor in maintaining high overall process efficiency and product quality.
How to Synthesize 3,6-Dichloro-2-aminopyridine Efficiently
The synthesis protocol outlined in the patent offers a streamlined workflow that balances reaction kinetics with safety. The process begins with the activation of the carboxylic acid using thionyl chloride in toluene at moderate temperatures (50-60°C), followed by the careful addition of ammonia source to form the amide. The second stage involves the generation of sodium hypobromite or hypochlorite, which is added to the amide in alkaline solution. The reaction is initiated at low temperatures (8-10°C) to control the exotherm during the formation of the N-haloamide, and then heated to 70-75°C to drive the rearrangement and hydrolysis to completion. Detailed standardized operating procedures for scaling this reaction from laboratory to pilot plant are essential for maintaining these precise thermal profiles.
- Suspend 3,6-dichloro-2-pyridinecarboxylic acid in toluene and react with thionyl chloride at 50-60°C to form the acid chloride, followed by ammonolysis at 5-25°C to yield the amide intermediate.
- Prepare sodium hypohalite solution and add dropwise to the amide in sodium hydroxide solution at 8-10°C, then heat to 70-75°C to effect Hofmann degradation.
- Extract the reaction mixture with chloroform, dry over magnesium sulfate, and recrystallize the crude product from toluene or hexane to obtain high-purity 3,6-dichloro-2-aminopyridine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the CN102875455A process translates into tangible strategic advantages regarding cost stability and supply security. The most immediate impact is the drastic reduction in raw material costs. By substituting the expensive and hazardous n-butyllithium with commodity reagents like thionyl chloride, ammonia, and sodium hydroxide, the variable cost per kilogram of the final product is significantly lowered. Furthermore, the starting material, 3,6-dichloro-2-pyridinecarboxylic acid, is a high-volume agrochemical intermediate with a mature global supply chain, ensuring consistent availability and shielding the project from the volatility often seen with specialized fine chemical reagents. This shift from scarce, high-cost reagents to abundant, low-cost commodities creates a much more resilient cost structure for long-term production contracts.
- Cost Reduction in Manufacturing: The elimination of cryogenic operations removes a major energy cost center. Maintaining reactors at -78°C requires significant refrigeration capacity and insulation, whereas the new process operates near ambient temperature or with mild heating (up to 75°C). Additionally, the removal of n-butyllithium eliminates the need for specialized quenching and waste treatment protocols associated with pyrophoric materials, further reducing operational overhead and waste disposal costs. The high crude yield of the amide intermediate (>96%) also means less raw material is wasted, directly improving the mass balance and overall economic efficiency of the manufacturing campaign.
- Enhanced Supply Chain Reliability: Relying on a starting material that is already produced on a scale of over 1000 tons annually for other industries guarantees a secure supply base. Unlike custom-synthesized precursors that may face bottlenecks, 3,6-dichloro-2-pyridinecarboxylic acid is readily available from multiple suppliers globally. This abundance reduces lead times for raw material procurement and mitigates the risk of production stoppages due to upstream shortages. The simplified process flow, which avoids sensitive organometallic steps, also reduces the likelihood of batch failures due to operator error or equipment malfunction, ensuring a more predictable delivery schedule for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently safer and easier to scale. The absence of air-sensitive reagents means that standard stainless steel reactors can be used without the need for extensive inert gas purging or specialized glove-box techniques. This simplifies the engineering requirements for scale-up from kilograms to metric tons. From an environmental perspective, the process avoids the generation of lithium salts and complex organometallic waste streams. The byproducts are primarily inorganic salts and gases that are easier to treat and neutralize, facilitating compliance with increasingly stringent environmental regulations and reducing the burden on wastewater treatment facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, focusing on the practical implications for industrial adoption. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: Why is the Hofmann degradation route superior to the lithiation method for 3,6-dichloro-2-aminopyridine?
A: The Hofmann degradation route described in CN102875455A avoids the use of expensive and hazardous n-butyllithium and eliminates the need for cryogenic conditions (-78°C). Furthermore, because the starting material (3,6-dichloro-2-pyridinecarboxylic acid) already possesses the correct substitution pattern, it prevents the formation of difficult-to-separate isomers like 5,6-dichloro-2-aminopyridine, which are common in the lithiation pathway.
Q: What are the key cost drivers reduced in this new synthesis process?
A: The primary cost reductions come from replacing the expensive reagent n-butyllithium with commodity chemicals like thionyl chloride and sodium hypohalite. Additionally, the starting material, 3,6-dichloro-2-pyridinecarboxylic acid (Picloram acid), is a mass-produced agrochemical intermediate available at low cost, and the elimination of energy-intensive cryogenic cooling significantly lowers utility expenses.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is highly scalable. It operates at mild temperatures (room temperature to 75°C) using standard solvents like toluene and chloroform. The absence of air-sensitive reagents and extreme低温 conditions simplifies engineering requirements, making it ideal for multi-ton production in standard stainless steel reactors without specialized cryogenic infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,6-Dichloro-2-aminopyridine Supplier
The technological breakthroughs encapsulated in CN102875455A highlight the immense potential for optimizing the production of critical heterocyclic intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative patent routes into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this Hofmann degradation route are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of 3,6-dichloro-2-aminopyridine meets the exacting standards required for pharmaceutical applications, free from the isomeric impurities that plague older methods.
We invite you to collaborate with us to leverage this cost-effective and scalable technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how switching to this optimized synthesis path can enhance your project's economics and reliability.
