Advanced Synthesis of 3,4,5-Trisubstituted Isoxazoles for Oncology Drug Development
Introduction to Novel Antitumor Isoxazole Scaffolds
The landscape of oncology drug discovery is constantly evolving, driven by the urgent need for novel scaffolds that can overcome resistance mechanisms in various tumor types. Patent CN115710234A introduces a significant advancement in this field by disclosing a series of 3,4,5-trisubstituted isoxazole compounds with demonstrated antitumor activity. These heterocyclic compounds are not merely theoretical constructs; they represent a tangible opportunity for developing next-generation therapeutics targeting pancreatic, breast, colon, liver, and lung cancers. The core innovation lies in the versatile substitution pattern allowed at the R1, R3, R4, and R5 positions, enabling medicinal chemists to fine-tune lipophilicity, electronic properties, and steric bulk to optimize binding affinity against biological targets. As a reliable pharmaceutical intermediate supplier, understanding the structural nuances of these molecules is critical for supporting downstream drug development programs.
The structural diversity offered by this patent is substantial, encompassing phenyl, styryl, ethynylphenyl, and morpholinylphenyl groups among others. This breadth of chemical space suggests that these isoxazole derivatives can serve as privileged structures in medicinal chemistry. The ability to access such a wide array of analogs through a unified synthetic strategy is a major value proposition for R&D teams looking to build robust structure-activity relationship (SAR) libraries. By leveraging these intermediates, pharmaceutical companies can accelerate their lead optimization phases, potentially reducing the time-to-market for critical oncology treatments. The integration of these scaffolds into existing pipelines offers a strategic advantage in the competitive landscape of cancer therapy development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of complex heterocyclic systems like trisubstituted isoxazoles has been fraught with challenges that hinder efficient production. Conventional methods often rely on harsh reaction conditions, such as strong acids or bases and elevated temperatures, which can lead to poor regioselectivity and the formation of difficult-to-remove impurities. Furthermore, older synthetic routes frequently require multiple protection and deprotection steps, significantly increasing the step count and overall cost of goods sold (COGS). The use of exotic or unstable reagents in traditional protocols also poses supply chain risks, making it difficult to secure consistent quality for clinical trial materials. These limitations create bottlenecks in the drug development process, delaying the availability of promising candidates for preclinical and clinical evaluation.
The Novel Approach
In contrast, the methodology described in CN115710234A offers a streamlined and modular approach that addresses many of these historical pain points. The novel strategy utilizes palladium-catalyzed cross-coupling reactions, specifically Sonogashira coupling, to construct the carbon-carbon bonds necessary for the isoxazole core. This modern catalytic approach operates under milder conditions, often utilizing inert atmospheres and moderate temperatures to ensure high fidelity in bond formation. The modularity of the route allows for the late-stage introduction of diverse functional groups, meaning that a single intermediate can be diverted into multiple final products with minimal additional processing. This flexibility is paramount for cost reduction in API manufacturing, as it reduces the need for maintaining separate inventory lines for every single analog.
Mechanistic Insights into Pd-Catalyzed Cyclization and Coupling
To fully appreciate the technical robustness of this synthesis, one must delve into the mechanistic details of the catalytic cycles employed. The process begins with the formation of an ynone intermediate via the coupling of an acid chloride and a terminal alkyne. This step is facilitated by a dual catalyst system comprising Pd(PPh3)2Cl2 and CuI, which activates the alkyne for nucleophilic attack on the acyl chloride. The resulting ynone is then converted into an O-methyl oxime, a crucial precursor that sets the stage for the subsequent cyclization. The precision of this transformation is vital, as the geometry of the oxime influences the regioselectivity of the ring closure. Understanding these mechanistic subtleties allows process chemists to troubleshoot potential issues related to catalyst poisoning or incomplete conversion.
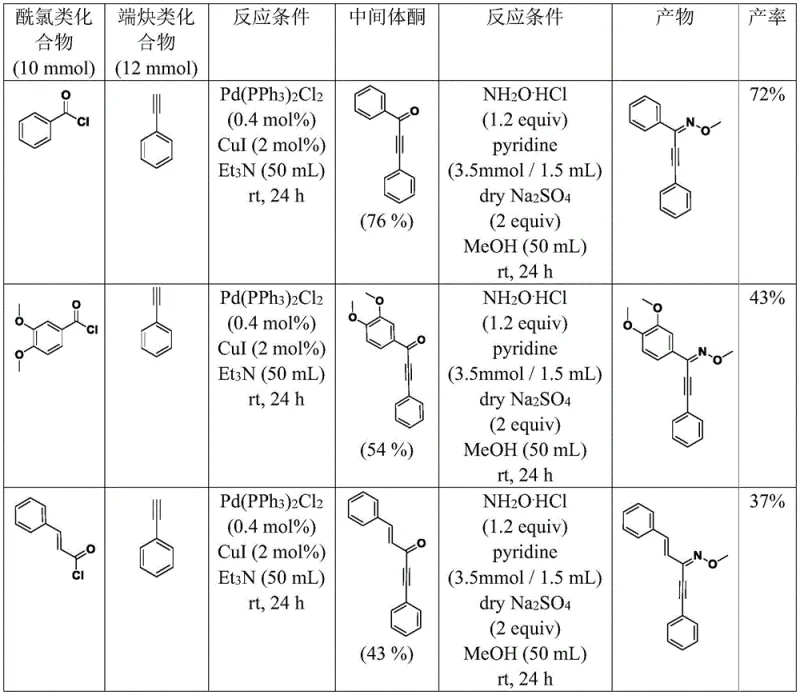
The subsequent cyclization steps vary depending on the desired substitution pattern, utilizing distinct routes (Route 2 through Route 5) tailored for specific structural outcomes. For instance, Route 2 involves a palladium-catalyzed reaction with 1,2-naphthoquinone and an alkene, proceeding through an oxidative cyclization mechanism. This pathway is particularly elegant as it constructs the isoxazole ring while simultaneously installing complex side chains. The use of specific oxidants and ligands ensures that the catalytic cycle turns over efficiently, minimizing the accumulation of palladium black and other inactive species. By controlling the stoichiometry of reagents and the reaction atmosphere, the process achieves high purity profiles, which is essential for meeting the stringent regulatory requirements of pharmaceutical intermediates. This level of mechanistic control translates directly into reproducible batch-to-batch consistency.
How to Synthesize 3,4,5-Trisubstituted Isoxazoles Efficiently
The practical execution of this synthesis requires careful attention to reaction parameters and purification techniques. The patent outlines a clear sequence starting from readily available acid chlorides and alkynes, moving through the oxime intermediate, and culminating in the final cyclized product. Each step is optimized for yield and purity, utilizing standard workup procedures such as aqueous washes and column chromatography. For R&D teams aiming to replicate or scale this chemistry, the detailed conditions provided in the patent serve as a valuable blueprint. The standardized protocols reduce the experimental burden, allowing scientists to focus on biological evaluation rather than method development. Detailed standardized synthesis steps are provided in the guide below.
- Perform Sonogashira coupling between acid chlorides and terminal alkynes using Pd(PPh3)2Cl2 and CuI catalysts to form ynone intermediates.
- Convert ynones to O-methyl oximes using methoxylamine hydrochloride and pyridine in methanol solvent.
- Execute specific cyclization routes (Route 2-5) using palladium catalysts and appropriate coupling partners like alkenes or aryl halides to form the final isoxazole core.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers significant strategic benefits for procurement and supply chain management. The reliance on commodity chemicals such as acid chlorides, terminal alkynes, and common solvents like triethylamine and methanol ensures a stable and resilient supply chain. Unlike processes that depend on bespoke or single-source reagents, this methodology leverages a global market of raw materials, mitigating the risk of supply disruptions. This stability is crucial for maintaining continuous production schedules, especially when scaling up to meet the demands of clinical trials or commercial launch. The predictability of raw material availability allows for more accurate forecasting and inventory planning, reducing the carrying costs associated with safety stock.
- Cost Reduction in Manufacturing: The streamlined nature of the synthesis contributes to substantial cost savings throughout the production lifecycle. By eliminating unnecessary protection and deprotection steps, the overall material throughput is improved, leading to less waste and lower disposal costs. Additionally, the use of efficient palladium catalysts, even at low loadings, maximizes atom economy and reduces the consumption of expensive metal resources. The ability to perform reactions at room temperature or with moderate heating further lowers energy consumption, aligning with green chemistry principles and reducing utility expenses. These cumulative efficiencies result in a more competitive cost structure for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The modular design of the synthetic route enhances supply chain reliability by decoupling the production of different analogs. Since the key intermediates are common to multiple final products, manufacturers can produce these building blocks in larger batches and store them for later diversification. This strategy reduces lead times for high-purity pharmaceutical intermediates, as the longest lead time steps are completed in advance. Furthermore, the robustness of the catalytic systems means that production is less susceptible to minor variations in raw material quality, ensuring consistent output even when sourcing from different vendors. This resilience is a key factor in securing long-term supply agreements with major pharmaceutical partners.
- Scalability and Environmental Compliance: Scaling complex organic syntheses often presents challenges related to heat transfer and mixing, but this process is designed with scalability in mind. The reaction conditions are compatible with standard stainless steel reactors, and the exothermic profiles are manageable, reducing the need for specialized cryogenic equipment. From an environmental standpoint, the process generates less hazardous waste compared to traditional stoichiometric methods. The solvents used are largely recoverable and recyclable, supporting sustainability goals and simplifying compliance with increasingly strict environmental regulations. This alignment with eco-friendly manufacturing practices enhances the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these isoxazole compounds. These insights are derived directly from the patent specifications and are intended to clarify the capabilities and limitations of the technology. By understanding these aspects, stakeholders can make informed decisions regarding the integration of these intermediates into their development pipelines. The answers reflect the current state of the art as described in the intellectual property documentation.
Q: What are the key advantages of the patented synthesis route for isoxazole intermediates?
A: The patented method utilizes modular palladium-catalyzed cross-coupling reactions which allow for high structural diversity at the 3, 4, and 5 positions. This flexibility enables rapid optimization of pharmacological properties without requiring entirely new synthetic pathways for each analog.
Q: How does this method address impurity control in pharmaceutical manufacturing?
A: The process employs specific catalyst systems such as Pd(PPh3)2Cl2/CuI under controlled inert atmospheres, which significantly reduces side reactions. Furthermore, the use of mild reaction conditions, often at room temperature or moderate heating, minimizes thermal degradation and byproduct formation.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the methodology relies on commercially available starting materials like acid chlorides and terminal alkynes. The reaction conditions utilize standard organic solvents and established catalytic cycles, making the transition from laboratory scale to industrial metric-ton production feasible and robust.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted Isoxazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of oncology drug development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your needs from early-stage research through to full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest standards of quality and consistency. Our commitment to excellence makes us a trusted partner for pharmaceutical companies seeking to advance their antitumor candidates.
We invite you to contact our technical procurement team to discuss your specific requirements. We are prepared to provide a Customized Cost-Saving Analysis tailored to your project's unique constraints and goals. By collaborating with us, you gain access to specific COA data and route feasibility assessments that will empower your decision-making process. Let us help you accelerate your path to market with reliable supply and technical expertise.
