Scalable Synthesis of O-Glycoside Nitrones: A Strategic Route for High-Purity Pharmaceutical Intermediates
Scalable Synthesis of O-Glycoside Nitrones: A Strategic Route for High-Purity Pharmaceutical Intermediates
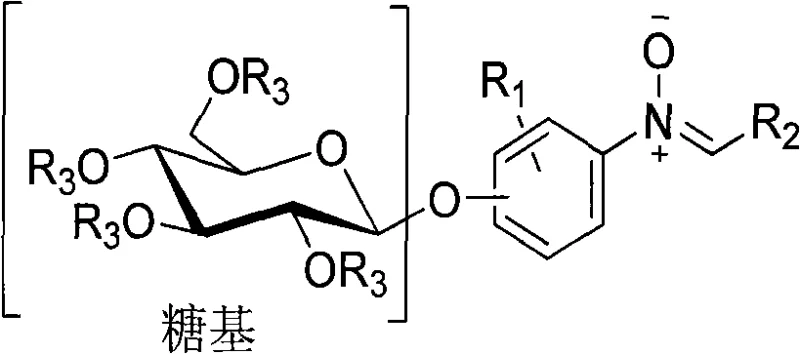
The development of novel heterocyclic compounds with enhanced biological profiles remains a cornerstone of modern medicinal chemistry. Patent CN101824057A introduces a robust and versatile synthetic methodology for producing O-glycoside nitrone compounds, a class of molecules that uniquely combine the pharmacokinetic benefits of glycosides with the radical-scavenging and 1,3-dipolar reactivity of nitrones. This technology addresses the critical need for water-soluble, biologically active intermediates that can serve as precursors for complex alkaloids or function directly as therapeutic agents. By leveraging naturally abundant reducing sugars such as glucose and lactose, the disclosed process offers a sustainable pathway to high-value fine chemicals. The strategic fusion of a sugar moiety with a nitrone functional group not only modulates solubility but also imparts specific stereochemical configurations, specifically the single beta-type, which is often difficult to achieve with conventional non-enzymatic methods. For R&D teams seeking reliable sources of chiral building blocks, this patent represents a significant advancement in carbohydrate chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of nitrone-containing glycosides has been plagued by issues regarding stereocontrol and solubility. Conventional routes often rely on harsh acidic conditions that can lead to the degradation of sensitive carbohydrate backbones or the formation of anomeric mixtures (alpha/beta), necessitating difficult and yield-reducing separation processes. Furthermore, many existing nitrone derivatives suffer from poor aqueous solubility, limiting their bioavailability and utility in physiological environments. The reliance on expensive, specialized starting materials or transition metal catalysts that require rigorous removal steps further complicates the supply chain for these intermediates. In large-scale manufacturing, the inability to consistently produce a single stereoisomer can result in batch-to-batch variability, posing significant regulatory hurdles for pharmaceutical applications. Additionally, traditional methods often lack the modularity to easily introduce diverse aromatic substituents, restricting the chemical space available for structure-activity relationship (SAR) studies.
The Novel Approach
The methodology outlined in CN101824057A overcomes these barriers through a modular, four-step sequence that prioritizes stereochemical integrity and operational simplicity. By initiating the synthesis with the peracetylation of readily available reducing sugars, the process protects the hydroxyl groups while activating the anomeric center for subsequent substitution. The use of a red phosphorus and liquid bromine system for bromination ensures high conversion to the reactive glycosyl bromide intermediate under mild thermal control (0-25°C). Crucially, the employment of phase-transfer catalysis in the glycosylation step allows for the efficient coupling of the sugar bromide with various hydroxy-aromatic aldehydes in a biphasic system, driving the reaction towards the desired beta-configuration. This approach eliminates the need for cryogenic conditions or exotic reagents, making it highly amenable to commercial scale-up. The final condensation with N-substituted hydroxylamines proceeds smoothly in aromatic solvents, yielding the target nitrones with high purity and defined structural characteristics.
Mechanistic Insights into the Four-Step Glycosylation and Nitrone Formation
The core of this synthetic strategy lies in the precise manipulation of the anomeric center to ensure exclusive beta-selectivity. The process begins with the acid-catalyzed acetylation of sugars like glucose using acetic anhydride and zinc chloride at 85-100°C. This step converts the hemiacetal into a fully protected peracetylated sugar, stabilizing the molecule for subsequent transformations. Following this, the generation of the glycosyl bromide is achieved via reaction with red phosphorus and bromine. This halogenation step is critical as it creates a good leaving group at the anomeric position, facilitating the nucleophilic attack by the phenolic oxygen of the aromatic aldehyde in the next stage. The reaction conditions are tightly controlled between 0-25°C to prevent side reactions and decomposition of the labile bromo-sugar intermediate.
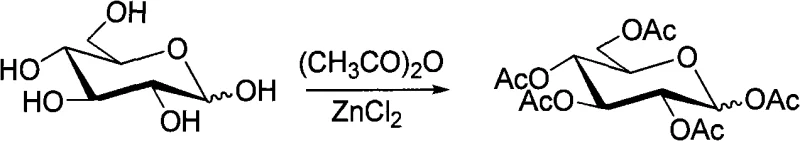
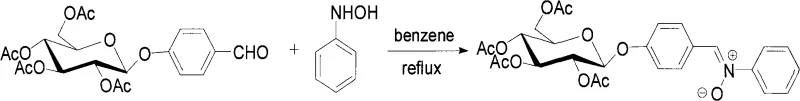
The glycosylation step utilizes a phase-transfer catalyst, such as tetrabutylammonium chloride, to bridge the organic and aqueous phases containing the bromo-sugar and the deprotonated hydroxy-aldehyde respectively. This interfacial catalysis promotes the SN2-like displacement of the bromide ion by the phenoxide, resulting in the inversion of configuration that leads to the stable beta-glycosidic linkage. The final transformation involves the condensation of the resulting O-benzaldehyde glycoside with N-substituted hydroxylamines (e.g., N-phenylhydroxylamine or N-benzylhydroxylamine). This reaction, typically conducted in benzene under reflux, forms the characteristic C=N(O)- bond of the nitrone. The electron-withdrawing nature of the nitrone group, combined with the steric bulk of the sugar moiety, contributes to the stability and unique electronic properties of the final molecule, making it an effective 1,3-dipole for further cycloaddition reactions or as a spin trap for free radicals.
How to Synthesize O-Glycoside Nitrone Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing reproducibility and safety. The procedure is designed to minimize purification bottlenecks by utilizing crystallization and standard extraction techniques rather than complex chromatography at every stage. For instance, the initial acetylation product can often be purified by simple recrystallization from ethanol, while the bromination mixture is worked up via aqueous extraction and drying. The glycosylation reaction progress is conveniently monitored by TLC, allowing operators to quench the reaction precisely upon completion to maximize yield. Detailed standardized synthesis steps for implementing this route are provided in the guide below.
- Acetylate reducing sugars (e.g., glucose) with acetic anhydride using ZnCl2 catalyst at 85-100°C to form peracetylated sugar.
- React peracetylated sugar with red phosphorus and liquid bromine in acetate at 0-25°C to generate bromo-sugar intermediates.
- Perform phase-transfer catalyzed glycosylation between bromo-sugar and hydroxy-aromatic aldehydes at 50-65°C to yield O-benzaldehyde glycosides.
- Condense the O-benzaldehyde glycoside with N-substituted hydroxylamine in benzene under reflux to obtain the final O-glycoside nitrone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the economic viability of this synthetic route is driven by the utilization of commodity-grade raw materials. Glucose, lactose, acetic anhydride, and simple aromatic aldehydes are globally available in bulk quantities, insulating the supply chain from the volatility associated with specialized fine chemical precursors. The elimination of precious metal catalysts significantly reduces the cost of goods sold (COGS) and simplifies the regulatory dossier by removing the need for extensive heavy metal residue testing. Furthermore, the reaction conditions are relatively mild, operating at temperatures that do not require extreme cooling or high-pressure equipment, thereby lowering capital expenditure (CAPEX) for reactor infrastructure. The high atom economy of the acetylation and condensation steps contributes to reduced waste generation, aligning with modern green chemistry principles and lowering disposal costs.
- Cost Reduction in Manufacturing: The process avoids the use of expensive chiral auxiliaries or enzymes, relying instead on the inherent chirality of natural sugars to dictate stereochemistry. This intrinsic stereocontrol eliminates the costly resolution steps often required in racemic syntheses. Additionally, the phase-transfer catalysis system allows for the use of cheaper solvents like chloroform and water mixtures, rather than expensive anhydrous aprotic solvents, further driving down operational expenses. The high yields reported in the examples, particularly in the final nitrone formation step (often exceeding 80-90%), ensure that raw material costs are effectively translated into product output without significant loss.
- Enhanced Supply Chain Reliability: Because the starting materials are fundamental chemicals with established global supply networks, the risk of supply disruption is minimized. The robustness of the synthetic steps means that minor variations in reagent quality can be tolerated without compromising the final product specification, providing flexibility in vendor selection. The ability to synthesize a wide range of derivatives by simply swapping the aromatic aldehyde or the hydroxylamine component allows manufacturers to respond quickly to changing market demands without retooling the entire production line. This modularity ensures a steady supply of diverse intermediates for downstream drug discovery programs.
- Scalability and Environmental Compliance: The batch processes described are inherently scalable from gram to multi-ton scales using standard stirred-tank reactors. The workup procedures involve standard liquid-liquid extractions and crystallizations, which are well-understood unit operations in the fine chemical industry. The use of acetic acid byproducts, which can be recovered and recycled, enhances the environmental profile of the process. Moreover, the absence of toxic heavy metals simplifies wastewater treatment protocols, ensuring compliance with stringent environmental regulations in major manufacturing hubs. This ease of scale-up facilitates the commercial production of complex pharmaceutical intermediates with consistent quality.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. Understanding these nuances is essential for process chemists and project managers evaluating this route for potential licensing or contract manufacturing opportunities. The answers are derived directly from the experimental data and claims presented in the patent documentation.
Q: What is the stereochemical outcome of the glycosylation step in this patent?
A: The synthesis method described in CN101824057A specifically yields O-glycoside nitrone compounds with a single beta-type configuration, ensuring high stereopurity which is critical for biological activity.
Q: Can this process be adapted for disaccharides like lactose?
A: Yes, the patent explicitly demonstrates the versatility of the method by successfully applying the same four-step sequence to lactose, producing heptaacetyl-beta-lactose derivatives with comparable efficiency.
Q: What are the key advantages of using phase transfer catalysts in this synthesis?
A: The use of phase transfer catalysts like tetrabutylammonium chloride allows the glycosylation reaction to proceed efficiently in a biphasic chloroform-water system at moderate temperatures (50-65°C), simplifying workup and improving yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable O-Glycoside Nitrone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced glycosylation technologies in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity O-glycoside nitrone intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex carbohydrate chemistry allows us to offer customized solutions that optimize both cost and quality for our global partners.
We invite you to collaborate with us to leverage this innovative synthetic route for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your project timelines while reducing overall manufacturing costs.
