Advancing Dialkyl Succinyl Succinate Production with Safe Magnesium Catalysis for Global Markets
The chemical industry is constantly evolving towards safer and more efficient synthetic pathways, and the production of dialkyl succinyl succinate stands as a prime example of this technological shift. Patent CN102093219A introduces a groundbreaking methodology that replaces traditional, hazardous alkali metal catalysts with a robust magnesium alkoxide system. This innovation not only addresses the critical safety concerns associated with handling metallic sodium and potassium but also delivers substantial improvements in product purity and overall process yield. For R&D directors and procurement specialists seeking a reliable fine chemical intermediate supplier, understanding this mechanistic pivot is essential for optimizing supply chains and reducing operational risks. The transition to alkaline earth metal catalysis represents a significant leap forward in the manufacturing of high-value intermediates used in advanced pigments and pharmaceutical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dialkyl succinyl succinate has relied heavily on the use of sodium methoxide or potassium ethoxide as condensation catalysts, a practice fraught with inherent logistical and safety challenges. The reliance on metallic sodium and potassium necessitates stringent safety protocols due to their extreme flammability and reactivity with moisture, which significantly increases processing and transportation costs for manufacturers. Furthermore, conventional methods often struggle with inconsistent reaction kinetics, leading to variable yields that typically hover around the 70% to 80% range, thereby impacting the economic viability of large-scale production. The formation of by-products and the difficulty in removing residual alkali metals from the final product often compromise the purity required for high-end applications such as quinacridone pigments. These legacy processes create a bottleneck for supply chain heads who require consistent quality and predictable lead times without the burden of managing hazardous waste streams.
The Novel Approach
The novel approach detailed in the patent utilizes magnesium alkoxide generated in situ from magnesium chips and alcohol, fundamentally altering the reaction landscape to favor safety and efficiency. By substituting volatile alkali metals with stable magnesium sources, the process eliminates the risk of spontaneous combustion and simplifies the storage and handling requirements for raw materials. This method employs a sophisticated solvent system comprising xylene and dimethyl sulfoxide (DMSO), which facilitates better heat transfer and prevents the caking of reactants that often plagues traditional batch reactions. The result is a more controlled exothermic profile that allows for higher reaction temperatures and more complete conversion of the starting dialkyl succinate. Consequently, this innovative route achieves superior product specifications, with purity levels reaching 99.2% and yields exceeding 88%, setting a new benchmark for cost reduction in specialty chemical manufacturing.
Mechanistic Insights into Magnesium-Catalyzed Claisen-Dieckmann Condensation
The core of this synthetic breakthrough lies in the dual-function catalytic mechanism where magnesium alkoxide drives both the intermolecular Claisen condensation and the subsequent intramolecular Dieckmann cyclization. Unlike sodium ions, magnesium cations possess a higher charge density which allows for tighter coordination with the carbonyl oxygen atoms of the succinate ester, effectively activating the substrate for nucleophilic attack. This enhanced activation lowers the energy barrier for enolate formation, promoting a smoother progression through the tetrahedral intermediate stages without the excessive side reactions often seen with harsher alkali bases. The presence of the DMSO co-solvent further stabilizes the magnesium enolate species, preventing premature decomposition and ensuring that the cyclization proceeds to form the six-membered ring structure with high fidelity. This precise control over the reaction pathway is crucial for minimizing impurity profiles and ensuring the structural integrity of the final cyclic diketone.
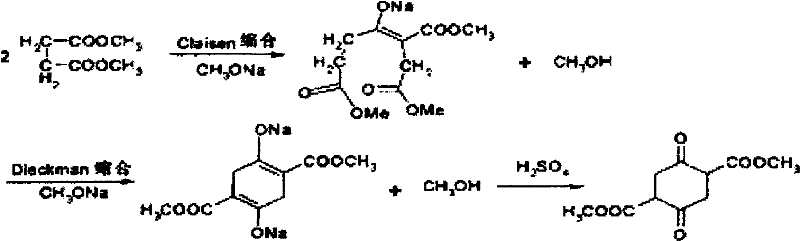
Impurity control in this magnesium-catalyzed system is achieved through the specific solubility characteristics of the intermediate magnesium salts and the careful management of the acidification step. As the reaction progresses, the magnesium salt of the product precipitates out of the xylene-DMSO matrix, effectively driving the equilibrium forward and separating the product from soluble by-products in real-time. Upon completion, the careful addition of dilute hydrochloric acid to pH 2 ensures the quantitative liberation of the free acid form without inducing hydrolysis of the ester groups, a common pitfall in less controlled acidic workups. The subsequent recrystallization from xylene leverages the differential solubility of the target molecule versus colored impurities, resulting in a white crystalline product that meets rigorous quality standards. This multi-stage purification embedded within the reaction design demonstrates a deep understanding of process chemistry, offering a robust solution for producing high-purity dialkyl succinyl succinate suitable for sensitive downstream applications.
How to Synthesize Dialkyl Succinyl Succinate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for replicating these high-yield results in a pilot or production setting, emphasizing the importance of solvent ratios and temperature control. Operators must first generate the magnesium alkoxide solution under an inert nitrogen atmosphere to prevent oxidation, followed by the precise addition of the co-solvent to optimize reaction homogeneity. The detailed standardized synthesis steps below outline the exact molar ratios and thermal profiles required to maximize efficiency while maintaining safety standards throughout the operation.
- Prepare magnesium alkoxide solution by reacting magnesium chips with alcohol solvent (methanol or ethanol) in xylene under nitrogen protection at 60-90°C.
- Add dimethyl sulfoxide (DMSO) as a co-solvent and introduce dialkyl succinate, maintaining reaction temperature between 95-120°C while fractionally distilling off generated alcohol.
- Acidify the magnesium salt precipitate with dilute hydrochloric acid, filter, wash, and recrystallize the crude product in xylene to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this magnesium-based technology translates directly into tangible operational benefits and risk mitigation strategies. The elimination of pyrophoric metals like sodium and potassium removes a major liability from the facility, reducing insurance premiums and the need for specialized hazardous material handling equipment. Furthermore, the improved yield and purity reduce the volume of raw materials required per unit of output, driving down the overall cost of goods sold without compromising on quality specifications. This process enhancement ensures a more stable supply of critical intermediates, shielding the supply chain from volatility associated with the sourcing and transport of dangerous reagents.
- Cost Reduction in Manufacturing: The substitution of expensive and hazardous metallic sodium with readily available magnesium chips results in significant raw material cost savings and reduced waste disposal expenses. By avoiding the complex neutralization and cleanup procedures required for alkali metal residues, manufacturers can streamline their production cycles and lower labor costs associated with safety monitoring. The higher reaction yield means less feedstock is wasted, directly improving the margin profile for every batch produced and enhancing the overall economic feasibility of the project.
- Enhanced Supply Chain Reliability: Sourcing magnesium is generally more stable and less regulated than sourcing reactive alkali metals, which are often subject to strict transportation restrictions and geopolitical supply fluctuations. This stability ensures that production schedules can be maintained without interruption due to raw material shortages or regulatory delays in shipping hazardous goods. Additionally, the safer nature of the reagents allows for broader logistics options, enabling faster delivery times and more flexible inventory management strategies for global distribution networks.
- Scalability and Environmental Compliance: The process is inherently scalable due to the use of common industrial solvents like xylene and ethanol, which are easily recovered and recycled in standard distillation units. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, facilitating easier permitting for capacity expansion and reducing the carbon footprint of the manufacturing site. This green chemistry approach not only future-proofs the production facility against regulatory changes but also appeals to end-customers who prioritize sustainable sourcing in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this magnesium-catalyzed synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation to provide clarity on process capabilities.
Q: Why is magnesium alkoxide preferred over sodium methoxide for this synthesis?
A: Magnesium alkoxide offers significant safety advantages by eliminating the fire hazards associated with metallic sodium and potassium. Additionally, it reduces raw material costs and simplifies transportation logistics while achieving higher yields and purity levels compared to traditional alkali metal catalysts.
Q: What is the role of DMSO in the reaction solvent system?
A: Dimethyl sulfoxide (DMSO) acts as a critical co-solvent that enhances the solubility of intermediates and stabilizes the reaction environment. Its presence allows for better control over the condensation process, preventing wall-caking and ensuring complete conversion of the starting ester into the cyclic product.
Q: Can this process be scaled for industrial production of pigment intermediates?
A: Yes, the process is designed for scalability, utilizing common industrial solvents like xylene and ethanol. The removal of hazardous alkali metals makes the scale-up process safer and more compliant with environmental regulations, making it ideal for large-scale manufacturing of quinacridone pigment precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dialkyl Succinyl Succinate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global fine chemical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of dialkyl succinyl succinate meets the highest standards required for pharmaceutical and specialty pigment applications.
We invite you to contact our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. Request a Customized Cost-Saving Analysis today to understand the economic impact of switching to this safer, high-yield process, and ask for specific COA data and route feasibility assessments tailored to your volume needs.
