Scalable Synthesis of Thermally Stable AIE Fluoroboron Dyes for Advanced Display Technologies
Scalable Synthesis of Thermally Stable AIE Fluoroboron Dyes for Advanced Display Technologies
The rapid evolution of the organic light-emitting diode (OLED) and bio-imaging sectors demands fluorescent materials that overcome the intrinsic limitations of traditional dyes, specifically aggregation-caused quenching and limited thermal robustness. Patent CN114605452A introduces a groundbreaking class of beta-dicarbonyl fluoroboron compounds engineered with propeller-shaped substituents to induce aggregation-induced emission (AIE). This technological leap addresses the critical bottleneck of fluorescence self-quenching in solid states while achieving a remarkable Stokes shift of up to 127 nm and thermal decomposition temperatures exceeding 440°C. For R&D directors and procurement specialists in the electronic chemicals sector, this patent represents a pivotal shift towards more reliable and high-performance luminescent materials that can withstand rigorous processing conditions without degradation.
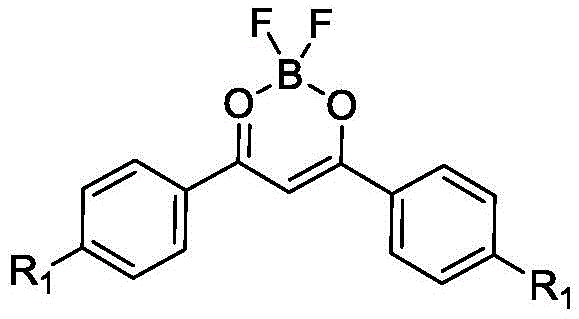
The development of high-efficiency fluorescent dyes has long been hindered by the trade-off between brightness in solution and brightness in the solid state. Conventional beta-dicarbonyl boron-fluorine complexes, while possessing high quantum yields in dilute solutions, typically suffer from severe aggregation-induced fluorescence quenching when concentrated or processed into films, rendering them unsuitable for many solid-state device applications. Furthermore, these legacy materials often exhibit small Stokes shifts of less than 50 nm, leading to significant self-absorption and reduced signal-to-noise ratios in sensing applications. The novel approach detailed in the patent circumvents these issues by integrating bulky, propeller-like aniline or carbazole substituents that physically prevent pi-pi stacking interactions, thereby unlocking strong emission in aggregated states and enabling red-shifted fluorescence through intramolecular charge transfer mechanisms.
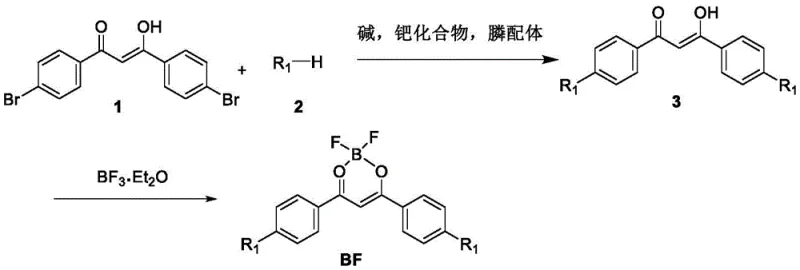
The mechanistic foundation of this synthesis relies on a sophisticated donor-acceptor (D-A) architecture where the electron-deficient six-membered boron fluoride ring acts as the acceptor and the electron-rich carbazole or aniline groups serve as donors. This push-pull electronic system is established through a palladium-catalyzed cross-coupling reaction, likely proceeding via a standard oxidative addition, transmetallation, and reductive elimination cycle typical of Buchwald-Hartwig aminations. The use of bulky phosphine ligands, such as tri-tert-butylphosphine, is critical in this mechanism as they facilitate the oxidative addition of the aryl bromide and stabilize the active palladium species, ensuring high conversion rates even with sterically hindered substrates. Following the formation of the beta-dicarbonyl intermediate, the subsequent complexation with boron trifluoride diethyl etherate creates a rigidified structure that restricts intramolecular rotation, a key factor in enhancing fluorescence quantum efficiency and thermal stability.
Impurity control in this synthesis is paramount for electronic grade materials, and the process utilizes robust purification strategies to ensure high purity. The initial coupling step generates a beta-dicarbonyl intermediate which is purified via column chromatography to remove unreacted amines, palladium residues, and homocoupling byproducts before the final complexation step. This two-stage purification approach is superior to one-pot methods as it prevents the formation of difficult-to-remove boron-complexed impurities early in the sequence. The final fluoroboron complex is then subjected to a second purification, ensuring that the final product meets the stringent purity specifications required for OLED manufacturing, where trace metal contaminants or organic impurities can drastically reduce device lifetime and efficiency.
How to Synthesize Aggregation-Induced Emission Fluoroboron Dyes Efficiently
The synthesis protocol outlined in the patent provides a reproducible pathway for generating these high-value intermediates, leveraging widely available starting materials and standard laboratory equipment. The process begins with the preparation of the key intermediate, 1,3-bis(4-bromophenyl)-3-hydroxypropenyl-ketone, which serves as the electrophilic core for subsequent functionalization. By reacting this core with specific aniline or carbazole derivatives under inert atmosphere protection, manufacturers can precisely tune the photophysical properties of the final dye. The detailed standardized synthesis steps below outline the precise stoichiometry, temperature controls, and workup procedures necessary to achieve the reported yields and purity levels consistently.
- Perform a palladium-catalyzed cross-coupling reaction between 1,3-bis(4-bromophenyl)-3-hydroxypropenyl-ketone and propeller-substituted aniline or carbazole derivatives using a base like cesium carbonate.
- Purify the resulting intermediate beta-dicarbonyl compound via column chromatography to ensure high purity before complexation.
- React the purified intermediate with boron trifluoride diethyl etherate in dry dichloromethane under reflux to form the final fluoroboron complex.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers substantial advantages in terms of raw material security and process scalability. The starting materials, including brominated acetophenones and various substituted anilines or carbazoles, are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. Furthermore, the reaction conditions utilize standard solvents like toluene and dichloromethane, which are easily recovered and recycled in an industrial setting, aligning with modern green chemistry initiatives and reducing waste disposal costs. The robustness of the palladium-catalyzed step allows for potential scale-up from gram to kilogram quantities without significant re-optimization, ensuring a steady supply of high-quality intermediates for downstream device fabrication.
- Cost Reduction in Manufacturing: The elimination of complex multi-step sequences required for traditional AIEgens significantly streamlines the production workflow. By utilizing a direct coupling strategy followed by a simple complexation step, the overall process time and labor costs are drastically reduced compared to iterative synthesis methods. Additionally, the high thermal stability of the final product minimizes material loss during high-temperature processing steps common in OLED device fabrication, leading to improved overall material utilization and lower cost per unit of finished display.
- Enhanced Supply Chain Reliability: The use of stable, non-hazardous reagents such as cesium carbonate and palladium acetate ensures that the manufacturing process is not subject to the strict regulatory hurdles associated with highly toxic or unstable precursors. This regulatory ease facilitates smoother logistics and faster customs clearance for international shipments. Moreover, the ability to store the intermediate beta-dicarbonyl compounds allows for a flexible production schedule where the final complexation can be performed on demand, reducing inventory holding costs and improving responsiveness to market fluctuations.
- Scalability and Environmental Compliance: The synthetic route avoids the use of exotic or environmentally persistent reagents, relying instead on well-established organometallic chemistry that is easily adapted to continuous flow reactors for large-scale production. The high yields reported in the patent examples, such as the 56% yield for the intermediate and 90% for the final complex in Example 1, indicate an atom-economical process that generates minimal waste. This efficiency not only lowers the environmental footprint but also translates directly into economic savings through reduced raw material consumption and waste treatment expenses.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluoroboron technology in industrial settings. These answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers evaluating this material for integration into their product lines.
Q: What is the primary advantage of this fluoroboron compound over traditional dyes?
A: Unlike traditional beta-dicarbonyl boron complexes that suffer from aggregation-caused quenching and small Stokes shifts, this novel compound features a propeller-shaped substituent that induces aggregation-induced emission (AIE) and achieves a massive Stokes shift of up to 127 nm.
Q: How does the thermal stability of this material compare to industry standards?
A: The synthesized compounds exhibit exceptional thermal stability, with a 5% mass loss temperature reaching as high as 440.9°C, which significantly exceeds the stability of many conventional organic fluorescent materials used in display applications.
Q: Can the emission wavelength be tuned for specific applications?
A: Yes, by selecting carbazole or aniline rotor units with different electron-donating abilities and steric hindrance, manufacturers can effectively adjust the fluorescence emission wavelength, quantum yield, and Stokes shift to meet specific optoelectronic requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluoroboron Compound Supplier
As the demand for advanced optoelectronic materials continues to surge, partnering with a manufacturer that possesses deep technical expertise in complex organic synthesis is essential for maintaining a competitive edge. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent quality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of fluoroboron compound meets the exacting standards required for high-performance OLED and bio-imaging applications.
We invite procurement leaders and R&D teams to collaborate with us to explore the full potential of these aggregation-induced emission materials. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments that will demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and product performance.
