Advanced Synthesis of Diphenyloxyphosphorus-Substituted Phenanthroimidazole for High-Efficiency OLEDs
Advanced Synthesis of Diphenyloxyphosphorus-Substituted Phenanthroimidazole for High-Efficiency OLEDs
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that simultaneously offer high color purity, efficient carrier transport, and thermal stability. Patent CN112194679A introduces a groundbreaking class of diphenyloxyphosphorus-substituted phenanthroimidazole compounds that address the critical challenge of efficiency roll-off in deep blue emitters. Unlike traditional planar molecules that suffer from severe aggregation-caused quenching due to pi-pi stacking, these novel compounds utilize a twisted spatial structure induced by the diphenyl phosphine oxide group. This structural modification effectively suppresses intermolecular interactions in the solid state, thereby preserving high fluorescence quantum efficiency. As a reliable OLED material supplier, understanding the nuanced chemical architecture of these emitters is essential for developing next-generation display technologies that require precise color coordinates and long operational lifetimes.
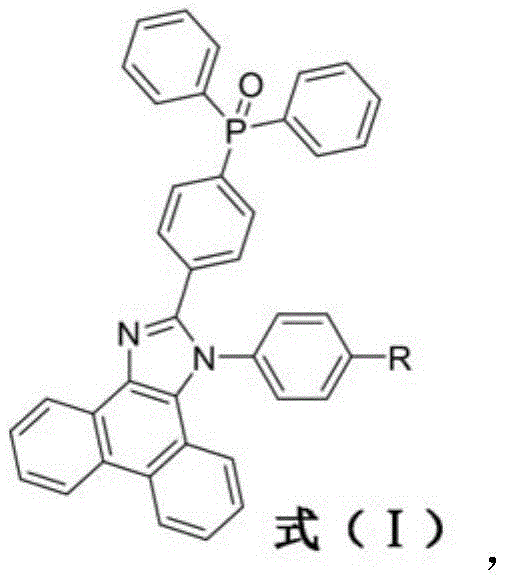
The core innovation lies in the specific donor-pi-acceptor configuration where the phenanthroimidazole unit serves as a robust electron donor and the diphenyl phosphine oxide functions as a potent electron acceptor. This balance is crucial because traditional modifications often result in unbalanced carrier injection, leading to device instability or undesirable spectral shifts. By integrating the phosphine oxide moiety, the invention achieves a delicate equilibrium that enhances pi-electron delocalization without compromising the deep blue emission characteristics. For procurement managers and R&D directors alike, this represents a significant opportunity to source high-purity electroluminescent materials that simplify device architecture while maximizing performance metrics such as external quantum efficiency and color gamut coverage.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of blue OLED materials has been plagued by the trade-off between color purity and charge transport efficiency. Conventional small molecule emitters often possess rigid planar structures that facilitate strong intermolecular pi-pi interactions in the aggregated state. These interactions lead to non-radiative decay pathways, commonly known as aggregation-caused quenching (ACQ), which drastically reduces the luminous efficiency of the device. Furthermore, attempts to modify these cores with strong electron-withdrawing groups to improve electron transport frequently result in excessive intramolecular charge transfer (ICT). This ICT phenomenon causes a significant red-shift in the emission spectrum, pushing the color out of the desirable deep blue region and lowering the color purity required for high-definition displays. Additionally, many existing synthetic routes rely on expensive palladium-catalyzed cross-couplings that introduce heavy metal impurities, necessitating costly and complex purification steps to meet the stringent standards of the electronics industry.
The Novel Approach
The methodology outlined in CN112194679A circumvents these issues through a strategic two-step synthesis that builds complexity efficiently. The process begins with the construction of a 4,4-bisbromophenanthroimidazole scaffold, which provides a stable and reactive platform for subsequent functionalization. The key breakthrough is the introduction of the diphenyl phosphine oxide group via a nickel-catalyzed coupling reaction. This specific functional group not only acts as an electron acceptor to balance carrier transport but also introduces steric bulk that twists the molecular geometry. This twisting physically prevents the molecules from packing too closely in the solid film, thus mitigating the quenching effects seen in planar analogues. The result is a material that maintains deep blue emission even in high-concentration films, offering a distinct advantage for cost reduction in electronic chemical manufacturing by reducing the need for complex host-guest doping strategies.
Mechanistic Insights into Ni-Catalyzed Phosphorylation and Cyclization
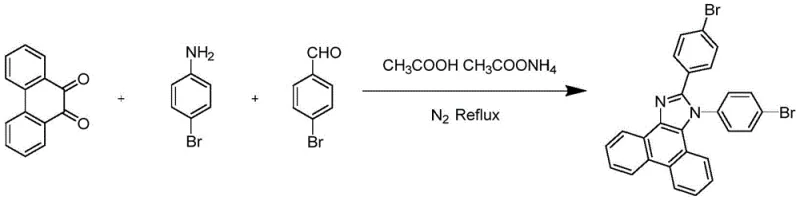
The synthetic pathway relies on a robust imidazole cyclization followed by a transition-metal catalyzed C-P bond formation. In the first stage, 4-bromobenzaldehyde, p-bromoaniline, and 9,10-phenanthrenequinone undergo a condensation reaction in the presence of ammonium acetate and glacial acetic acid. This multicomponent reaction proceeds through the formation of an imine intermediate which subsequently cyclizes to form the phenanthroimidazole core. The reaction conditions are optimized at temperatures between 115-125°C, typically around 120°C, ensuring complete conversion within 2 to 4 hours. The use of acetic acid as both solvent and catalyst facilitates the dehydration steps necessary for ring closure, while the ammonium acetate acts as a nitrogen source and buffer. This step is critical for establishing the rigid conjugated system that defines the optical properties of the final emitter, and the high yield reported (up to 82%) indicates a highly favorable thermodynamic profile for this transformation.
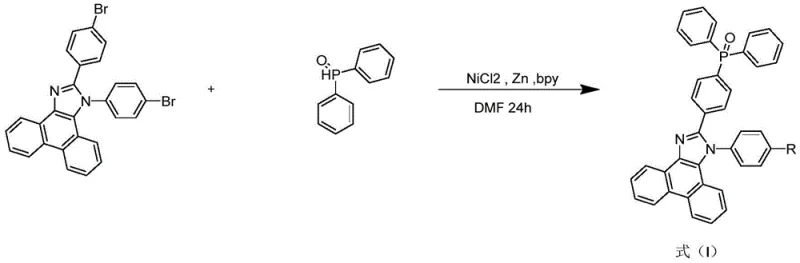
The second stage involves the installation of the phosphine oxide groups, which is the defining feature of this technology. The 4,4-bisbromophenanthroimidazole intermediate reacts with diphenylphosphine oxide in the presence of a nickel chloride and zinc powder catalyst system. This reaction, often referred to as a reductive cross-coupling or phosphorylation, replaces the bromine atoms with the phosphorus-containing moieties. The mechanism likely involves the oxidative addition of the aryl bromide to the nickel center, followed by transmetallation or radical processes mediated by the zinc reductant, and finally reductive elimination to form the C-P bond. Conducted in solvents like DMF or THF at 80-90°C for 18 to 36 hours, this step ensures deep conversion despite the steric hindrance of the bulky phosphine oxide. The choice of nickel over palladium is a strategic decision that lowers catalyst costs while maintaining high activity, which is vital for the commercial scale-up of complex organic semiconductors.
How to Synthesize Diphenyloxyphosphorus-Substituted Phenanthroimidazole Efficiently
The preparation of these advanced emitters follows a streamlined protocol designed for reproducibility and scalability. The process leverages common laboratory equipment such as two-necked flasks and standard heating mantles, making it accessible for pilot plant operations. The initial cyclization yields a white powder intermediate that can be purified via simple methanol washing or silica gel chromatography. The subsequent phosphorylation step requires an inert atmosphere to protect the sensitive nickel catalyst and phosphine reagents from oxidation. Following the reaction, the crude product is isolated through extraction with dichloromethane and brine, followed by vacuum distillation to remove solvents. Final purification is achieved using column chromatography with a petroleum ether and dichloromethane gradient, yielding the target compounds M1 or M2 as high-purity yellow or white powders suitable for vacuum deposition in OLED devices.
- Perform imidazole cyclization by reacting 4-bromobenzaldehyde, p-bromoaniline, and 9,10-phenanthrenequinone with ammonium acetate in glacial acetic acid at 120°C for 2 hours to form the bis-bromo intermediate.
- Conduct the phosphorylation step by reacting the bis-bromo intermediate with diphenylphosphine oxide using a Nickel Chloride and Zinc powder catalyst system in THF/DMF at 90°C for 24 hours.
- Purify the crude reaction mixture via vacuum distillation and silica gel column chromatography using petroleum ether and dichloromethane to isolate the final high-purity yellow or white powder products.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this synthetic route offers tangible benefits regarding cost structure and material availability. The reliance on commodity chemicals such as bromobenzaldehyde and phenanthrenequinone ensures a stable supply chain that is less susceptible to the volatility often seen with exotic heterocyclic building blocks. Furthermore, the elimination of precious metal catalysts like palladium in favor of abundant nickel and zinc significantly reduces the bill of materials. This shift not only lowers the direct cost of goods sold but also simplifies the regulatory compliance landscape regarding heavy metal residues in electronic components. The robust nature of the reaction conditions, which tolerate standard laboratory temperatures and pressures, further implies that scaling this process from grams to kilograms requires minimal re-engineering of existing reactor infrastructure.
- Cost Reduction in Manufacturing: The substitution of expensive palladium catalysts with a nickel-zinc system represents a major economic advantage. Palladium prices are historically volatile and high, whereas nickel and zinc are abundant and cost-effective industrial metals. By removing the need for precious metals, the overall catalyst cost per kilogram of product is drastically simplified and reduced. Additionally, the high yields observed in the initial cyclization step minimize waste generation, contributing to a more lean and efficient manufacturing process that maximizes atom economy and reduces the burden on waste treatment facilities.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including 4-bromobenzaldehyde and 9,10-phenanthrenequinone, are widely produced intermediates available from multiple global suppliers. This diversity in sourcing options mitigates the risk of supply disruptions that can occur when relying on single-source specialty reagents. The synthetic route itself is robust, with reaction times and temperatures that are easily controlled in standard batch reactors. This reliability translates to predictable production schedules and reducing lead time for high-purity display materials, allowing downstream device manufacturers to plan their inventory with greater confidence and accuracy.
- Scalability and Environmental Compliance: The purification methods described, primarily involving filtration and column chromatography with common solvents like petroleum ether and dichloromethane, are well-established techniques in the fine chemical industry. These methods are easily adaptable to large-scale preparative chromatography or recrystallization processes. Moreover, the absence of toxic heavy metals in the final catalyst system simplifies the environmental impact assessment. The process generates less hazardous waste compared to traditional cross-coupling methods, aligning with increasingly strict global environmental regulations and supporting the industry's move towards greener chemical manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the synthesis and application of these diphenyloxyphosphorus-substituted compounds. Understanding these details is crucial for R&D teams evaluating the material for specific device architectures. The answers are derived directly from the experimental data and mechanistic explanations provided in the patent literature, ensuring accuracy and relevance for technical decision-making.
Q: How does the diphenyl phosphine oxide group improve OLED performance?
A: The diphenyl phosphine oxide group acts as an electron acceptor that balances the electron-donating phenanthroimidazole unit. This donor-pi-acceptor structure prevents excessive charge transfer red-shifting, maintaining high blue light color purity while improving carrier transport balance and preventing fluorescence quenching caused by pi-pi stacking.
Q: What are the critical reaction conditions for the phosphorylation step?
A: The phosphorylation requires a nickel-catalyzed system using Nickel Chloride (NiCl2) and Zinc powder (Zn) often with a ligand like bipyridine. The reaction is typically conducted in polar aprotic solvents like DMF or THF at temperatures between 80-90°C for extended periods (18-36 hours) to ensure complete substitution of the bromine atoms.
Q: Why is this synthesis route preferred for commercial scale-up?
A: This route utilizes readily available starting materials such as 4-bromobenzaldehyde and 9,10-phenanthrenequinone. Furthermore, the substitution of expensive palladium catalysts with a nickel-zinc system significantly reduces raw material costs, and the purification via standard column chromatography allows for scalable production of high-purity electronic chemicals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diphenyloxyphosphorus-Substituted Phenanthroimidazole Supplier
As the demand for high-performance blue emitters continues to surge, partnering with an experienced CDMO is critical for success. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications required for electronic grade materials. We understand that trace impurities can devastate OLED device lifetime, which is why our quality control protocols are designed to detect and eliminate sub-ppm level contaminants that could compromise performance.
We invite you to collaborate with us to optimize this synthetic route for your specific production volumes. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project requirements, identifying further opportunities for efficiency gains. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your development timeline and bring next-generation display technologies to market faster and more economically.
