Advanced Manufacturing of Eribulin Intermediates via Chromium-Reduced Catalysis
Advanced Manufacturing of Eribulin Intermediates via Chromium-Reduced Catalysis
The global demand for high-purity oncology therapeutics continues to surge, with Eribulin Mesylate standing as a cornerstone treatment for metastatic breast cancer. As a tubulin polymerization inhibitor with a unique mechanism of action, the supply chain integrity for its complex intermediates is paramount for pharmaceutical manufacturers worldwide. A significant technological breakthrough in this domain is detailed in patent CN113024589A, which discloses a novel preparation method for Eribulin and its key intermediates, specifically targeting the optimization of the synthetic route to Compound C. This innovation addresses critical bottlenecks in the legacy Kishi synthesis, offering a pathway that is not only chemically superior but also environmentally sustainable. By fundamentally re-engineering the hydroxyl protection strategy and refining the organometallic coupling steps, this technology provides a robust foundation for the commercial scale-up of complex pharmaceutical intermediates.
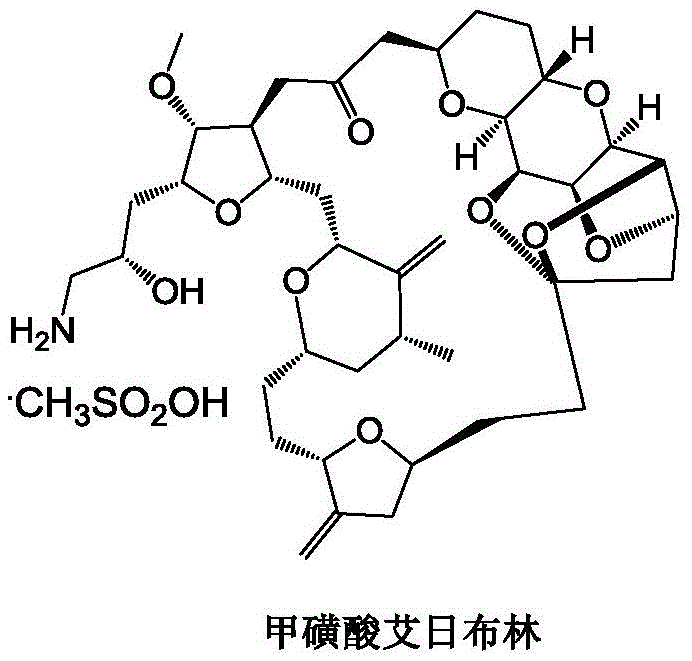
The strategic value of this patent lies in its ability to enhance the reliability of the eribulin intermediate supplier network. Traditional methods have long struggled with low overall yields and excessive reliance on toxic heavy metals, creating supply chain vulnerabilities and escalating waste management costs. The disclosed methodology introduces a cyclopentanone-based protection group to replace the conventional cyclohexanone, coupled with a Lewis acid-catalyzed condensation that dramatically improves reaction efficiency. Furthermore, the optimization of the Nozaki-Hiyama-Kishi (NHK) reaction through the addition of manganese powder and precise modulation of nickel and chromium catalysts represents a paradigm shift in green chemistry for fine pharmaceutical synthesis. For procurement leaders and R&D directors, adopting this route translates directly into cost reduction in pharmaceutical intermediates manufacturing while ensuring the stringent purity specifications required for clinical-grade active ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The historical benchmark for synthesizing the Eribulin C-ring fragment, often attributed to the Kishi group, relies heavily on a cyclohexanone-based protection strategy for the D-gulonolactone starting material. While chemically valid, this legacy approach suffers from severe inefficiencies that hinder modern large-scale production. The total yield for synthesizing the key intermediate Compound B via this traditional route is reported to be merely 19.2 percent, indicating substantial material loss across multiple steps. More critically, the Nozaki-Hiyama-Kishi (NHK) coupling step in the conventional process demands an exorbitant dosage of chromium reagents, typically ranging from 400 to 1600 mol percent. This excessive use of hexavalent chromium not only inflates raw material costs but also generates hazardous heavy metal waste, complicating environmental compliance and disposal logistics. The difficulty in purifying intermediates and the harsh conditions required further exacerbate the operational risks, making the conventional method economically and ecologically unsustainable for high-volume API intermediate manufacturing.
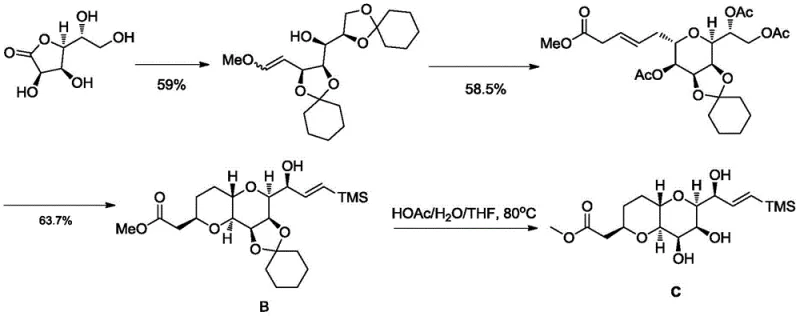
The Novel Approach
In stark contrast, the novel methodology disclosed in CN113024589A introduces a series of transformative improvements that dismantle these historical barriers. The process initiates with a condensation reaction between D-gulonolactone and cyclopentanone (or its derivatives), catalyzed by anhydrous zinc chloride, which serves as a benign Lewis acid. This switch in protecting group chemistry alone boosts the yield of the initial intermediate, Compound C01, to over 85 percent, a massive leap from previous standards. The route proceeds through a meticulously optimized sequence involving reduction, Wittig olefination, and highly stereoselective dihydroxylation. The crown jewel of this new approach is the revamped NHK reaction for converting Compound C08 to C01a. By incorporating manganese powder as an assimilation agent alongside a tuned mixture of NiCl2, CrCl2, aluminum scraps, and trimethylchlorosilane, the process slashes chromium usage to negligible levels (0.05-0.1 molar equivalent) while simultaneously boosting reaction yield. This holistic redesign ensures that the high-purity eribulin intermediates produced are both economically viable and environmentally responsible.
Mechanistic Insights into Optimized NHK Coupling and Stereoselective Oxidation
The mechanistic elegance of this new synthesis is best exemplified in the critical carbon-carbon bond-forming steps and the control of stereochemistry. The conversion of Compound C03 to C04 utilizes osmium tetroxide (OsO4) catalysis in an isopropanol-water solvent system at strictly controlled temperatures of 0-5°C. This specific condition set is crucial for achieving a diastereomeric ratio (d.r.) of greater than 25:1, effectively suppressing the formation of unwanted isomers that would otherwise complicate downstream purification. The high chiral selectivity ensures that the resulting polyol framework possesses the exact spatial configuration required for biological activity, thereby reducing the burden on chromatographic separation units. Furthermore, the use of N-methylmorpholine N-oxide as a co-oxidant allows for the catalytic turnover of osmium, minimizing the load of this toxic and expensive metal in the final product stream.
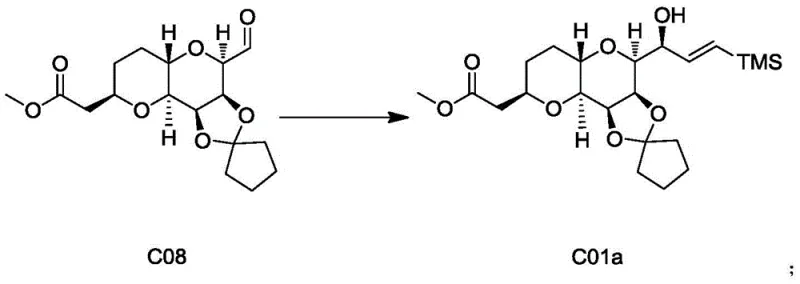
Equally significant is the mechanistic refinement of the Nozaki-Hiyama-Kishi (NHK) coupling reaction, which links the side chain to the core macrocycle precursor. In the traditional mechanism, chromium(II) acts as the primary reductant, but it is prone to rapid oxidation and requires vast excesses to drive the reaction to completion. The patented innovation introduces manganese powder into the reaction matrix, which acts as a sacrificial reductant to regenerate the active low-valent metal species in situ. This synergistic effect between Nickel, Chromium, Aluminum, and Manganese creates a highly active catalytic cycle that operates efficiently with only 0.01-0.05 molar equivalent of Nickel and 0.05-0.1 molar equivalent of Chromium. This drastic reduction in stoichiometric metal usage not only lowers the impurity profile of the crude product—facilitating easier workup and crystallization—but also aligns the process with strict ICH guidelines for elemental impurities in drug substances. The result is a robust, scalable transformation that delivers Compound C01a with high fidelity.
How to Synthesize Eribulin Intermediate Compound C Efficiently
The synthesis of Compound C via this novel route involves a sequence of nine distinct chemical transformations, each optimized for yield and purity. The process begins with the Lewis acid-catalyzed protection of D-gulonolactone, followed by reduction and olefination to establish the carbon backbone. Subsequent steps involve ring closure, selective acetylation, and the pivotal NHK coupling described above. The final stages include cyclization, deacetylation, oxidation to the aldehyde, and a final deprotection step using aqueous acetic acid to reveal the free hydroxyl groups of Compound C. Each step has been engineered to avoid extreme conditions, utilizing solvents like toluene, THF, and acetonitrile which are easily recoverable. For a detailed breakdown of the specific reagents, molar ratios, and temperature profiles required to execute this synthesis in a GMP environment, please refer to the standardized protocol below.
- Protect D-gulonolactone with cyclopentanone using ZnCl2 catalyst to form Compound C01, followed by reduction to C02.
- Perform Wittig olefination to generate C03, followed by OsO4-catalyzed dihydroxylation to achieve high stereoselectivity in Compound C04.
- Execute the critical Nozaki-Hiyama-Kishi (NHK) coupling of aldehyde C08 with vinyl bromide using a Ni/Cr/Mn/Al system to yield intermediate C01a, finally deprotecting to Compound C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers compelling strategic advantages that extend beyond simple chemistry. The primary value proposition lies in the drastic simplification of the waste management profile. By eliminating the need for massive excesses of chromium reagents, the facility requirements for heavy metal scrubbing and hazardous waste disposal are significantly reduced, leading to lower operational overheads. Additionally, the higher overall yield of the route—improved by approximately 20 percent compared to the prior art—means that less raw material is required to produce the same amount of finished intermediate. This efficiency gain directly translates to substantial cost savings in raw material procurement and inventory holding costs, making the final API more price-competitive in the global market.
- Cost Reduction in Manufacturing: The elimination of expensive transition metals in stoichiometric quantities and the reduction of reaction steps through higher yields create a leaner cost structure. The use of zinc chloride as a catalyst in the initial step is far more economical than the specialized reagents required in older routes. Furthermore, the simplified purification processes, driven by higher stereoselectivity and cleaner reaction profiles, reduce the consumption of chromatography media and solvents. These factors combine to lower the cost of goods sold (COGS) without compromising on quality, allowing for more aggressive pricing strategies in tender negotiations.
- Enhanced Supply Chain Reliability: The robustness of the new chemical route ensures consistent batch-to-batch quality, which is critical for maintaining regulatory filings and avoiding production delays. The starting materials, such as D-gulonolactone and cyclopentanone, are commodity chemicals with stable supply lines, reducing the risk of raw material shortages. Moreover, the mild reaction conditions (e.g., 0-5°C for sensitive steps rather than cryogenic temperatures) reduce the energy load on manufacturing plants and decrease the likelihood of thermal runaways or equipment failures. This operational stability guarantees reducing lead time for high-purity pharmaceutical intermediates and ensures uninterrupted supply to downstream API manufacturers.
- Scalability and Environmental Compliance: As regulatory scrutiny on environmental, social, and governance (ESG) metrics intensifies, this green chemistry approach positions the supply chain for long-term viability. The significant reduction in chromium waste aligns with global initiatives to minimize heavy metal pollution, facilitating easier permitting for plant expansions. The process is inherently scalable, having been designed with kilogram-to-tonnage production in mind, ensuring that supply can ramp up rapidly to meet market surges without the need for complex process re-validation. This scalability ensures that partners can rely on a steady flow of materials even during periods of high global demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy techniques in terms of yield, purity, and environmental impact.
Q: How does the new process reduce environmental impact compared to the Kishi route?
A: The novel method replaces the traditional cyclohexanone protection with a cyclopentanone-based strategy and significantly optimizes the Nozaki-Hiyama-Kishi (NHK) reaction. By introducing manganese powder and optimizing the Ni/Cr/Al catalyst system, the usage of toxic chromium reagents is drastically reduced from 400-1600 mol% in prior art to approximately 5-10 mol%, minimizing heavy metal waste.
Q: What is the stereochemical purity of the key intermediate C04?
A: The process achieves exceptional chiral control during the dihydroxylation step. Using OsO4 catalysis under controlled low-temperature conditions (0-5°C), the reaction yields Compound C04 with a diastereomeric ratio (d.r.) exceeding 25:1, ensuring high optical purity for downstream synthesis.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route is designed for scalability. It utilizes robust Lewis acid catalysis (ZnCl2) for the initial protection step with yields over 85% and employs mild reaction conditions for subsequent transformations. The total yield of the route to Compound C is improved by approximately 20% compared to conventional methods, enhancing overall process efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eribulin Mesylate Supplier
The technological advancements detailed in patent CN113024589A represent the cutting edge of oncology intermediate manufacturing, yet realizing their full potential requires a partner with deep process engineering expertise. NINGBO INNO PHARMCHEM stands at the forefront of this industry, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific nuances of this chromium-reduced route, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We understand that the transition to a new synthetic route involves careful validation, and our technical team is dedicated to ensuring a seamless technology transfer that maintains the highest standards of safety and quality.
We invite global pharmaceutical partners to collaborate with us to leverage this efficient synthesis for your Eribulin supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener, higher-yielding process. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample availability. Let us help you secure a sustainable, cost-effective, and reliable supply of high-quality Eribulin intermediates for your critical drug development programs.
