Scalable Synthesis of Fluorescent Imidazole Ligands for Advanced Biological Detection Applications
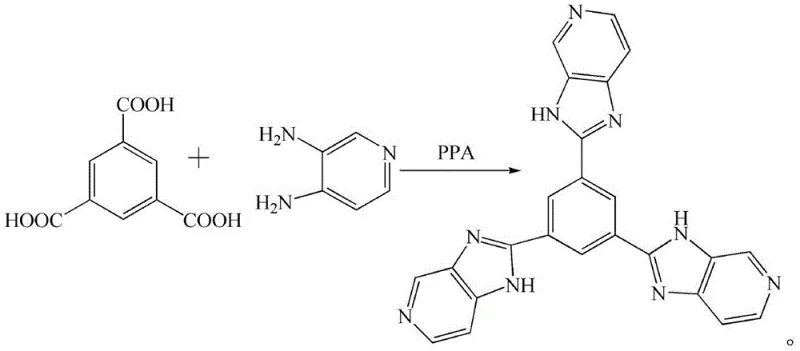
The pharmaceutical and biotechnology sectors are constantly seeking advanced molecular tools for precise biological detection, driving the demand for specialized fluorescent ligands. Patent CN110330511B introduces a robust synthetic methodology for producing a novel organic ligand, specifically 5-(1H-imidazo[4,5-c]pyridin-2-yl)-1,3-bis(3H-imidazo[4,5-c]pyridin-2-yl)benzene, referred to herein as Compound 3. This molecule represents a significant advancement in the field of bio-detection reagents due to its unique structural architecture featuring a central benzene ring flanked by imidazo-pyridine moieties. The patent outlines a streamlined process that utilizes polyphosphoric acid (PPA) as both a solvent and a dehydrating agent to facilitate the condensation reaction between trimesic acid and 3,4-diaminopyridine. For R&D directors and procurement specialists evaluating new supply chains, this technology offers a compelling value proposition by simplifying the production of high-value fluorescent intermediates. The method addresses common challenges in heterocyclic synthesis, such as incomplete cyclization and difficult purification, providing a clear pathway to high-purity materials essential for sensitive biological assays.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for complex imidazole-fused heterocycles often suffer from multi-step procedures that require harsh reaction conditions and expensive transition metal catalysts. Conventional methods frequently rely on sequential bond formations that introduce significant opportunities for side reactions, leading to complex impurity profiles that are difficult to separate. The use of volatile organic solvents and sensitive catalysts not only increases the environmental footprint but also complicates the scale-up process due to safety concerns regarding exothermic events. Furthermore, older methodologies often struggle with regioselectivity, resulting in isomeric mixtures that compromise the fluorescence quantum yield and binding affinity of the final ligand. These inefficiencies translate directly into higher manufacturing costs and longer lead times, creating bottlenecks for companies requiring reliable supplies of specialized chemical intermediates for diagnostic applications.
The Novel Approach
The methodology described in Patent CN110330511B circumvents these historical limitations by employing a direct condensation strategy mediated by polyphosphoric acid. This approach leverages the dual functionality of PPA to activate the carboxylic acid groups of trimesic acid while simultaneously promoting the cyclization with the diamine functionality of 3,4-diaminopyridine. By operating at elevated temperatures between 150-170°C under reflux, the reaction drives the equilibrium towards the formation of the stable imidazo[4,5-c]pyridine ring system with high efficiency. The process eliminates the need for external coupling agents or precious metal catalysts, significantly reducing the raw material cost and simplifying the downstream purification workflow. This direct one-pot style transformation ensures that the structural integrity of the conjugated system is maintained, which is critical for the optical properties required in biological detection applications.
Mechanistic Insights into Polyphosphoric Acid Mediated Cyclization
The core of this synthetic innovation lies in the mechanistic role of polyphosphoric acid (PPA) as a powerful dehydrating condensing agent. In this reaction system, PPA protonates the carbonyl oxygen of the trimesic acid, increasing its electrophilicity and facilitating nucleophilic attack by the amino groups of the 3,4-diaminopyridine. This initial amidation is followed by an intramolecular cyclodehydration step where the PPA assists in the elimination of water molecules to close the imidazole ring. The high viscosity and acidity of the PPA medium create a unique microenvironment that stabilizes the transition states and suppresses competing hydrolysis reactions. This mechanism ensures that the three arms of the trimesic acid core are uniformly functionalized, resulting in the symmetric C3-symmetric structure of Compound 3. Understanding this mechanism is vital for process chemists aiming to optimize reaction kinetics and minimize the formation of mono- or di-substituted byproducts.
Impurity control in this system is achieved through precise stoichiometric management and thermal regulation. The patent specifies a molar ratio of trimesic acid to 3,4-diaminopyridine between 1:3 and 1:3.4, ensuring a slight excess of the amine to drive the reaction to completion without overwhelming the system with unreacted starting material that could co-precipitate. The subsequent workup involves a carefully controlled neutralization step using sodium carbonate, which converts the acidic PPA matrix into water-soluble phosphates while precipitating the organic product. This pH-switch isolation technique is highly effective at removing inorganic salts and polar impurities. Finally, the triple recrystallization in methanol serves as a critical purification barrier, leveraging the differential solubility of the target ligand versus potential oligomeric impurities to deliver a product with the stringent purity specifications required for fluorescence-based assays.
How to Synthesize 5-(1H-imidazo[4,5-c]pyridin-2-yl)-1,3-bis(3H-imidazo[4,5-c]pyridin-2-yl)benzene Efficiently
Executing this synthesis requires strict adherence to the thermal and stoichiometric parameters defined in the patent to ensure reproducibility and high yield. The process begins with the uniform mixing of the solid reactants before introducing the polyphosphoric acid medium to prevent localized hot spots. Maintaining the reflux temperature within the 150-170°C window is crucial; temperatures below this range may result in incomplete cyclization, while excessive heat could lead to decomposition of the sensitive heterocyclic rings. The extended reaction time of 12 to 15 hours allows for the slow equilibration necessary to form the thermodynamically stable tris-substituted product. Detailed standardized synthesis steps, including specific mass ratios and workup volumes, are outlined in the technical guide below to assist process engineers in scaling this route from laboratory to pilot plant.
- Mix trimesic acid and 3,4-diaminopyridine in a molar ratio of 1: 3 to 1:3.4 within a polyphosphoric acid system.
- Conduct a reflux reaction at temperatures between 150-170°C for 12 to 15 hours to ensure complete cyclization.
- Dilute the reaction mixture with deionized water, neutralize with sodium carbonate, and purify the crude product via triple recrystallization in methanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this PPA-mediated synthesis route offers substantial strategic advantages over traditional multi-step pathways. The primary benefit lies in the drastic simplification of the bill of materials, as the process relies on commodity chemicals like trimesic acid and diaminopyridine rather than exotic, supply-constrained catalysts. This shift significantly reduces the risk of supply chain disruptions caused by the scarcity of specialized reagents. Additionally, the elimination of transition metals removes the need for expensive and time-consuming heavy metal scavenging steps, which are often a bottleneck in API intermediate manufacturing. The use of methanol and water for purification further aligns with green chemistry principles, lowering waste disposal costs and simplifying regulatory compliance regarding solvent residues in the final product.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the high atom economy of the condensation reaction and the recyclability of the solvent systems. By avoiding precious metal catalysts, the direct material costs are significantly lowered, and the burden on wastewater treatment facilities is reduced due to the absence of toxic metal ions. The straightforward isolation procedure via precipitation and recrystallization minimizes energy consumption associated with complex chromatographic separations. These factors combine to create a manufacturing profile that supports substantial cost savings, making the final fluorescent ligand more accessible for large-scale biological screening programs without compromising on quality.
- Enhanced Supply Chain Reliability: The reliance on bulk chemical feedstocks ensures a stable and continuous supply chain, as trimesic acid and pyridine derivatives are produced globally at industrial scales. The robustness of the PPA reaction conditions means that the process is less sensitive to minor fluctuations in raw material quality, enhancing batch-to-b consistency. This reliability is critical for long-term contracts where consistent delivery schedules are paramount. Furthermore, the simplified process flow reduces the overall cycle time from raw material intake to finished goods, allowing suppliers to respond more agilely to changes in market demand and reducing lead times for high-purity fluorescent intermediates.
- Scalability and Environmental Compliance: Scaling this reaction is facilitated by the homogeneous nature of the PPA medium, which handles heat transfer efficiently even in larger reactors. The workup procedure generates primarily inorganic phosphate salts and aqueous waste streams that are easier to treat compared to halogenated organic waste. This environmental compatibility simplifies the permitting process for new manufacturing lines and reduces the long-term liability associated with hazardous waste storage. The ability to scale from kilogram to multi-ton production while maintaining the same purification protocol ensures that the supply can grow in tandem with the commercial success of the downstream biological detection applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this imidazole ligand. These answers are derived directly from the experimental data and claims within Patent CN110330511B, providing a factual basis for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating this material into their existing workflows.
Q: What are the critical reaction conditions for synthesizing this imidazole ligand?
A: The synthesis requires a polyphosphoric acid (PPA) medium with a reflux temperature maintained between 150-170°C for 12-15 hours. The molar ratio of trimesic acid to 3,4-diaminopyridine is strictly controlled between 1:3 and 1:3.4 to maximize yield and minimize impurities.
Q: How is the purity of the final ligand ensured during workup?
A: Purity is achieved through a rigorous workup process involving dilution with deionized water, neutralization using a 10% sodium carbonate solution, and critically, three successive recrystallizations in methanol. This removes residual acids and unreacted starting materials effectively.
Q: What is the primary application of this synthesized compound?
A: The compound exhibits strong yellow-green fluorescence and acts as a static quencher for Bovine Serum Albumin (BSA). It is primarily designed for use in biological detection systems and as a building block for novel fluorescent materials with multiple coordination sites.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazole Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance fluorescent ligands play in the advancement of biological detection and diagnostic technologies. Our team of expert process chemists has extensively analyzed the synthetic route detailed in Patent CN110330511B and is fully prepared to execute this chemistry at commercial scales. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including detailed impurity profiling and fluorescence characterization, to guarantee that every batch meets the exacting standards required for research and development applications.
We invite you to collaborate with us to optimize this synthesis for your specific volume requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that evaluates the economic benefits of switching to this efficient manufacturing route. Please contact us to request specific COA data and route feasibility assessments tailored to your project timelines. By partnering with NINGBO INNO PHARMCHEM, you secure a supply chain partner dedicated to delivering quality, reliability, and innovation in the field of specialty chemical intermediates.
