Advanced Synthesis of Ranolazine Intermediate: A Breakthrough in Purity and Scalability
Advanced Synthesis of Ranolazine Intermediate: A Breakthrough in Purity and Scalability
The pharmaceutical landscape for anti-anginal medications has been significantly shaped by the introduction of Ranolazine, a late sodium ion current inhibitor approved for treating chronic angina. Central to the efficient manufacturing of this critical active pharmaceutical ingredient (API) is the availability of high-quality intermediates, specifically N-(2,6-dimethylphenyl)-2-(1-piperazinyl)acetamide. Patent CN103570645A discloses a revolutionary method for preparing this key intermediate, addressing long-standing challenges regarding impurity profiles and process safety that have plagued previous synthetic routes. By leveraging a sophisticated in situ salt formation strategy, this technology enables the production of the target compound with exceptional purity levels exceeding 99.5% and molar yields surpassing 95%. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic advantages of this patent is crucial for securing a robust supply chain. The following analysis delves into the technical nuances of this innovation, highlighting its potential to redefine cost structures and quality standards in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-(2,6-xylyl)-2-(1-piperazinyl)ethanamide has been fraught with significant chemical and operational hurdles that impede large-scale industrial adoption. Traditional Route 1, often favored for its conceptual simplicity, relies on the direct reaction of 2-chloro-N-(2,6-xylyl)acetamide with excess anhydrous piperazine to suppress the formation of disubstituted byproducts. However, due to the symmetrical configuration and identical reactivity of the two secondary amine groups in piperazine, achieving perfect selectivity is chemically improbable, inevitably leading to a mixture containing substantial amounts of the undesired disubstituted piperazine derivative. Purification of the target mono-substituted product from this mixture typically necessitates the use of diethyl ether for recrystallization, a solvent characterized by its extremely low boiling point and high volatility, posing severe explosion risks and environmental hazards in a factory setting. Furthermore, alternative methods attempting to use piperazine monohydrochloride directly often suffer from low yields due to product loss during extensive aqueous washing steps required to remove unreacted starting materials, as the target amide possesses non-negligible solubility in water. These cumulative inefficiencies result in elevated production costs, compromised safety profiles, and inconsistent batch-to-batch quality, rendering many conventional processes unsuitable for modern, high-volume API manufacturing demands.
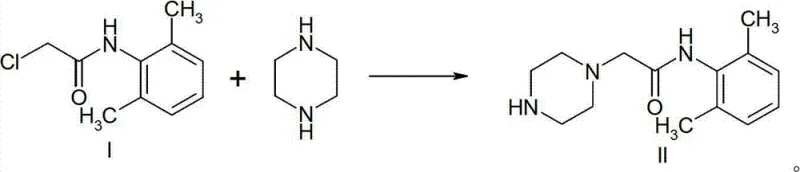
The Novel Approach
In stark contrast to these legacy techniques, the methodology outlined in CN103570645A introduces a paradigm shift by ingeniously controlling the nucleophilicity of the piperazine ring through precise stoichiometric manipulation. Instead of relying on hazardous ether purification or suffering from poor selectivity with free base piperazine, this novel approach generates piperazine monohydrochloride in situ by reacting piperazine dihydrochloride with anhydrous piperazine in a controlled molar ratio ranging from 2:1 to 4:1. This strategic pre-formation ensures that one nitrogen atom of the piperazine ring is permanently protonated and thus deactivated towards nucleophilic attack, effectively forcing the reaction to occur exclusively at the remaining free secondary amine. Consequently, the formation of the problematic disubstituted impurity is theoretically and practically eliminated at the source, rather than being removed downstream. The process utilizes absolute ethanol as a benign and easily recoverable solvent, replacing dangerous ethers and simplifying the workup procedure. By integrating a smart acidification step post-reaction, unreacted monohydrochloride is converted back into insoluble piperazine dihydrochloride, which can be filtered off and recycled, thereby maximizing atom economy and minimizing waste generation. This holistic redesign of the synthetic pathway offers a compelling solution for cost reduction in pharmaceutical intermediate manufacturing while simultaneously elevating product quality.
Mechanistic Insights into Controlled Mono-Substitution Strategy
The core chemical innovation driving the success of this patent lies in the thermodynamic and kinetic control of the piperazine species present in the reaction medium. In a standard nucleophilic substitution involving a diamine like piperazine, the statistical probability of double alkylation is high because the mono-alkylated product often retains similar or even enhanced nucleophilicity compared to the starting material. The patented method circumvents this by establishing an equilibrium where the dominant species is the piperazine monohydrochloride salt. The protonation of one nitrogen atom creates a strong electron-withdrawing effect via induction, significantly reducing the electron density on the adjacent nitrogen atom, but more importantly, the positive charge on the protonated nitrogen renders it completely inert to electrophilic attack by the chloroacetamide derivative. This acts as a temporary protecting group that requires no additional synthetic steps to install or remove. As the reaction proceeds under reflux in ethanol, the free amine attacks the alpha-carbon of the chloroacetamide, displacing the chloride ion to form the desired amide bond. Because the second nitrogen remains protonated throughout the majority of the reaction timeline, the likelihood of a second substitution event is negligible. This mechanistic certainty translates directly into a cleaner crude reaction profile, reducing the burden on downstream purification units and allowing for simpler crystallization protocols to achieve the reported >99.5% purity.
Furthermore, the post-reaction processing mechanism is equally critical for maintaining high yield and economic viability. Upon completion of the substitution, the reaction mixture contains the target product, solvent, and potentially unreacted piperazine monohydrochloride. The addition of a hydrochloric acid solution to adjust the pH to between 2 and 5 serves a dual purpose: it ensures any residual free amine is protonated, and crucially, it converts the soluble piperazine monohydrochloride into the less soluble piperazine dihydrochloride salt. This phase change allows for the physical separation of the excess piperazine reagent via simple filtration. The recovered piperazine dihydrochloride is not waste; it is a valuable raw material that can be fed back into the beginning of the process to generate more monohydrochloride, creating a closed-loop system for reagent usage. This recycling capability is a major factor in the process's overall efficiency, as it drastically reduces the consumption of piperazine, which is a significant cost driver. The filtrate, now enriched with the target amide and stripped of bulk amine impurities, undergoes standard basification and extraction, yielding a product of such high quality that extensive chromatographic purification is rendered unnecessary, further streamlining the manufacturing workflow.
How to Synthesize N-(2,6-dimethylphenyl)-2-(1-piperazinyl)acetamide Efficiently
Implementing this advanced synthesis route requires careful attention to stoichiometry and temperature control to fully realize the benefits of the in situ salt formation strategy. The process begins with the preparation of the reactive amine species in absolute ethanol, followed by the controlled addition of the electrophile and a specialized acidic workup to facilitate reagent recovery. Operators must adhere strictly to the specified molar ratios and pH targets to ensure the selective formation of the mono-substituted product and the efficient precipitation of the dihydrochloride byproduct. The following guide outlines the critical operational phases derived from the patent examples, providing a framework for scaling this chemistry from the laboratory to commercial production volumes. Detailed standard operating procedures and specific parameter optimizations should be consulted to adapt this general method to specific reactor configurations.
- React piperazine dihydrochloride and anhydrous piperazine in absolute ethanol at a molar ratio of 2: 1 to 4:1 to generate piperazine monohydrochloride in situ.
- Add 2-chloro-N-(2,6-xylyl)acetamide to the mixture and heat under reflux conditions to facilitate the nucleophilic substitution reaction.
- Acidify the reaction system to pH 2-5 with hydrochloric acid to convert unreacted monohydrochloride back to dihydrochloride for filtration and recycling.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route represents a significant opportunity to optimize both the cost base and the reliability of the supply chain for Ranolazine intermediates. The elimination of hazardous solvents like diethyl ether not only improves workplace safety but also reduces the capital expenditure associated with explosion-proof infrastructure and specialized solvent recovery systems. Moreover, the ability to recycle piperazine dihydrochloride directly from the reaction mother liquor transforms a potential waste stream into a reusable asset, substantially lowering the raw material cost per kilogram of the final product. The high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, which in turn reduces the complexity and duration of the purification process, leading to faster batch cycle times and increased throughput capacity without the need for additional equipment. These factors combine to create a manufacturing process that is not only more economically attractive but also more resilient to fluctuations in raw material pricing and regulatory scrutiny regarding solvent residues and environmental emissions.
- Cost Reduction in Manufacturing: The implementation of this method drives down production costs through multiple mechanisms, primarily by maximizing the utilization efficiency of piperazine reagents. By converting unreacted intermediates back into a recyclable salt form, the process minimizes raw material waste, which is a critical factor given the volatility of chemical commodity prices. Additionally, the replacement of low-boiling, high-loss solvents like ether with ethanol significantly improves solvent recovery rates, further contributing to overall cost savings. The high yield and purity achieved reduce the need for expensive reprocessing or discard of off-spec batches, ensuring that a higher percentage of input costs are converted into saleable product. These efficiencies collectively result in a more competitive cost structure for the intermediate, allowing for better margin management in the final API production.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex purification steps and reliance on hazardous materials that may face transportation or storage restrictions. This streamlined process mitigates such risks by utilizing common, non-hazardous solvents and straightforward unit operations like filtration and crystallization. The robustness of the chemistry, evidenced by the high tolerance for variation and consistent purity outcomes, ensures that production schedules can be met with greater predictability. Furthermore, the ability to recycle key reagents internally reduces dependency on external suppliers for every single batch, providing a buffer against supply chain disruptions. This stability is essential for maintaining uninterrupted API production lines, particularly for chronic medications like Ranolazine where consistent patient access is paramount.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this method is inherently designed for industrial suitability. The use of ethanol aligns with green chemistry principles, reducing the environmental footprint associated with volatile organic compound (VOC) emissions. The simplified workup, which avoids extensive aqueous washing that generates large volumes of wastewater containing dissolved product, significantly lowers the load on effluent treatment plants. The high atom economy resulting from the recycling loop means less solid waste is generated per unit of product. These attributes make the process highly scalable, allowing manufacturers to increase capacity from pilot scale to multi-ton production with minimal regulatory friction and lower capital investment in environmental control systems.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, drawing directly from the data and claims presented in the patent documentation. Understanding these details is vital for technical teams evaluating the feasibility of adopting this route for their own manufacturing operations or for procurement professionals assessing supplier capabilities. The answers provided reflect the specific advantages and operational parameters defined in the intellectual property, ensuring accuracy and relevance for decision-makers.
Q: How does this method prevent the formation of disubstituted piperazine impurities?
A: The method utilizes a precise molar ratio of piperazine dihydrochloride to anhydrous piperazine to generate piperazine monohydrochloride in situ. The protonated nitrogen is blocked, ensuring only one nitrogen atom participates in the nucleophilic attack, thereby drastically reducing disubstitution byproducts.
Q: What is the expected purity and yield of the target intermediate?
A: According to the patent data, this optimized process achieves a molar yield exceeding 95% and an HPLC purity greater than 99.5%, significantly outperforming traditional ether recrystallization methods.
Q: Is the solvent system safe for large-scale industrial production?
A: Yes, the process uses absolute ethanol as the primary solvent, eliminating the need for highly volatile and explosive solvents like ether, which enhances operational safety and simplifies solvent recovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(2,6-dimethylphenyl)-2-(1-piperazinyl)acetamide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceuticals like Ranolazine depends on the availability of intermediates produced with uncompromising quality and efficiency. Our technical team has extensively analyzed the breakthroughs detailed in CN103570645A and possesses the expertise to implement this advanced synthesis strategy at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of high yield and purity are realized in actual manufacturing runs. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the detection of trace disubstituted impurities, guaranteeing that every batch meets the exacting standards required for global API registration.
We invite pharmaceutical partners to collaborate with us to leverage this superior technology for their supply chains. By choosing NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized process can lower your total cost of ownership. We encourage interested parties to contact our technical procurement team to request specific COA data from our recent pilot batches and to discuss route feasibility assessments for your long-term projects. Let us be your strategic partner in delivering high-performance intermediates that drive the success of your final drug products.
