Advanced One-Step Synthesis of Watermelon Ketone for Commercial Flavor Production
Advanced One-Step Synthesis of Watermelon Ketone for Commercial Flavor Production
The global demand for high-quality olfactory ingredients continues to surge, driven by the premiumization of personal care and fine fragrance markets. At the forefront of this sector is Watermelon Ketone, chemically known as 7-methyl-3,4-dihydro-2H-1,5-benzodioxepin-3-one, a molecule renowned for its fresh, marine, and fruity olfactory profile. Recent technological advancements have significantly optimized the production of this valuable compound. Specifically, the methodology disclosed in Chinese Patent CN114456146B represents a paradigm shift in synthetic efficiency. This patent introduces a robust, one-step catalytic process utilizing macroporous anion exchange resins, which eliminates the complexities of traditional multi-step syntheses. By leveraging heterogeneous catalysis and high-boiling point solvents, this innovation delivers exceptional reaction selectivity and yield, addressing critical pain points for industrial manufacturers seeking reliable flavor & fragrance intermediate suppliers.
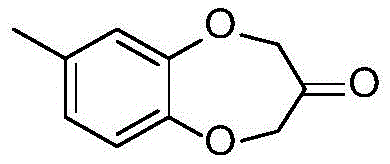
The structural integrity and purity of Watermelon Ketone are paramount for its application in high-end perfumery and functional fragrances. The molecule, depicted above, possesses a unique benzodioxepine ring system that is sensitive to harsh reaction conditions. Conventional synthetic routes often struggle to maintain the delicate aromatic profile of the final product due to the formation of oxidative byproducts or polymeric tars. The novel approach detailed in the patent data utilizes 4-methyl catechol and 1,3-dichloroacetone as primary feedstocks. Unlike previous iterations that relied on stoichiometric amounts of inorganic bases or phase transfer catalysts which were difficult to recover, this new method employs a solid-phase anion exchange resin. This not only simplifies the downstream processing but also ensures that the final organoleptic properties remain pristine, free from the metallic or amine-like off-notes associated with older catalytic systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Watermelon Ketone has been plagued by inefficiencies that drive up costs and compromise supply chain stability. Prior art, such as the methods described in academic literature by Zhang Jing, typically involves a two-step condensation and rearrangement sequence. These legacy processes often utilize sodium carbonate in an alkaline environment, resulting in mediocre yields hovering around 60 percent. Furthermore, the reliance on stepwise synthesis necessitates intermediate isolation, which increases solvent consumption and labor intensity. Other patented methods, such as those disclosed in CN101429188A, attempt a one-pot synthesis but suffer from even lower yields, approximately 46 percent, due to poor control over side reactions. Additionally, processes involving organic amines and iodinated salts, as seen in CN103058984B, introduce significant environmental hazards and purification challenges, as these additives are notoriously difficult to remove completely from the final product matrix.
The Novel Approach
In stark contrast, the methodology defined in CN114456146B streamlines the entire production workflow into a single, continuous operation. By substituting traditional homogeneous bases with macroporous basic styrene or acrylic anion exchange resins (such as models D201, D301, or D311), the reaction achieves a remarkable leap in selectivity. The resin acts as a solid base catalyst, facilitating the nucleophilic attack of the catechol hydroxyl groups on the dichloroacetone without promoting excessive polymerization. As illustrated in the reaction pathway below, the cyclization proceeds smoothly to form the seven-membered dioxepine ring. This mechanistic clarity allows for reaction yields to consistently exceed 92 percent, with some embodiments reaching 92.4 percent. The elimination of soluble inorganic salts and difficult-to-remove organic amines drastically simplifies the work-up procedure, transforming a cumbersome batch process into a streamlined operation suitable for continuous fixed-bed reactors.
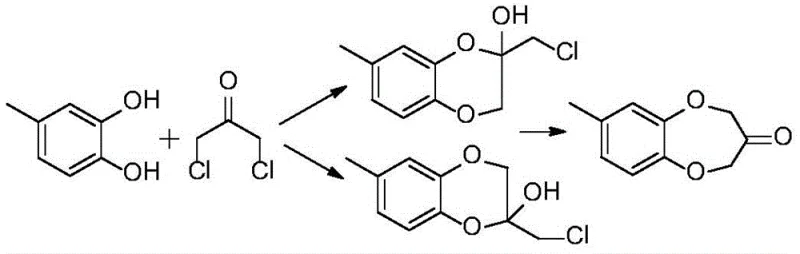
Mechanistic Insights into Anion Exchange Resin-Catalyzed Cyclization
The core innovation of this synthesis lies in the specific interaction between the anion exchange resin and the reactants within a high-boiling organic solvent medium. The resin, functioning as a heterogeneous base, deprotonates the phenolic hydroxyl groups of 4-methyl catechol, generating a phenoxide species that is highly nucleophilic. This activated species then attacks the electrophilic carbon centers of 1,3-dichloroacetone. The macro-porous structure of resins like D301 provides a large surface area and accessible active sites, ensuring efficient mass transfer even in viscous reaction media. Crucially, the solid nature of the catalyst prevents the localized high pH environments often seen with soluble bases, which are responsible for the degradation of the sensitive ketone functionality. This controlled basicity is the key to suppressing side reactions such as the formation of chlorohydrin intermediates that fail to cyclize, thereby maximizing the atom economy of the transformation.
Furthermore, the choice of solvent plays a pivotal role in the thermodynamic and kinetic profile of the reaction. The patent specifies the use of organic solvents with boiling points ranging from 300 to 350 degrees Celsius, such as benzophenone, octadecane, or eicosane. This is a critical deviation from standard protocols that utilize lower boiling solvents like methanol or toluene. In traditional low-boiling systems, the reaction temperature is capped by the solvent's reflux point, often necessitating prolonged reaction times to reach completion. Conversely, the high-boiling solvents allow the reaction to proceed at elevated temperatures, typically between 105 and 120 degrees Celsius, significantly accelerating the reaction kinetics. Moreover, during the post-reaction vacuum rectification, the high boiling point of the solvent ensures that the Watermelon Ketone product (boiling point approx. 305 degrees Celsius) distills over before the solvent residue becomes too concentrated, effectively minimizing thermal degradation and product loss in the still bottoms.
How to Synthesize Watermelon Ketone Efficiently
Implementing this advanced synthesis route requires precise control over reaction parameters and purification steps to replicate the high yields reported in the patent data. The process is versatile, capable of being executed in both batch kettle systems and continuous fixed-bed reactors, offering flexibility for different production scales. The key to success lies in the preparation of the feed solution, maintaining the correct molar ratio of 1,3-dichloroacetone to 4-methyl catechol (optimally between 0.95 and 1.05), and strictly adhering to the temperature profiles during the subsequent crystallization phases. The following guide outlines the standardized operational procedure derived from the patent examples, designed to ensure consistent product quality and maximize throughput for commercial manufacturing.
- Load anion exchange resin (e.g., D301) into a fixed-bed reactor or batch kettle under inert gas protection.
- React 4-methyl catechol and 1,3-dichloroacetone in a high-boiling organic solvent (300-350°C) at 105-120°C.
- Purify the crude product via vacuum rectification followed by a combined dynamic and static recrystallization process using ethyl acetate and n-hexane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this resin-catalyzed process offers tangible strategic benefits beyond mere chemical yield. The transition from a multi-step, salt-generating process to a one-step catalytic cycle fundamentally alters the cost structure of Watermelon Ketone manufacturing. By eliminating the need for stoichiometric amounts of inorganic bases and the subsequent neutralization and washing steps, the process significantly reduces the volume of wastewater generated. This reduction in effluent load translates directly into lower environmental compliance costs and reduced burden on waste treatment facilities. Furthermore, the ability to use the anion exchange resin in a fixed-bed configuration enables true continuous production, which drastically improves equipment utilization rates and reduces the labor intensity associated with batch charging and discharging.
- Cost Reduction in Manufacturing: The economic implications of this technology are profound, primarily driven by the drastic simplification of the downstream processing. In conventional methods, the removal of inorganic salts and organic amines often requires multiple extraction and washing cycles, consuming vast quantities of water and organic solvents. The new method, by virtue of using a solid, insoluble catalyst, allows for simple filtration or, in the case of fixed-bed reactors, no catalyst separation at all. This elimination of unit operations leads to substantial cost savings in terms of solvent recovery energy and labor hours. Additionally, the high selectivity of the resin minimizes the formation of hard-to-separate impurities, reducing the loss of valuable raw materials to waste streams and improving the overall material balance of the plant.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for any reliable flavor & fragrance intermediate supplier. The robustness of the anion exchange resin catalyst, which demonstrates stability over extended operation periods (e.g., 1000 hours with minimal activity loss), ensures consistent production output without frequent catalyst change-outs. This stability mitigates the risk of unplanned downtime that often plagues processes using sensitive homogeneous catalysts. Moreover, the raw materials—4-methyl catechol and 1,3-dichloroacetone—are commodity chemicals with stable global supply chains. The process tolerance for slight variations in feed ratios further enhances operational resilience, allowing manufacturers to maintain steady output even when facing minor fluctuations in raw material quality.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production is often fraught with heat transfer and mixing challenges. However, the exothermic nature of this cyclization is well-managed by the high-boiling solvent system, which acts as a thermal buffer. The process is inherently safer and easier to scale than reactions requiring cryogenic conditions or hazardous reagents. From an environmental perspective, the absence of heavy metal catalysts or persistent organic pollutants aligns perfectly with the increasingly stringent global regulations on chemical manufacturing. The ability to produce high-purity product without generating halogenated waste streams positions this technology as a sustainable choice for long-term manufacturing strategies.
Frequently Asked Questions (FAQ)
The technical nuances of this synthesis method often raise questions regarding its practical implementation and comparative advantages over established technologies. The following answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing clarity on the operational parameters and quality outcomes expected from this advanced catalytic system. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing production infrastructure.
Q: Why is anion exchange resin preferred over traditional inorganic bases for Watermelon Ketone synthesis?
A: Traditional methods using inorganic bases often require multi-step processes and result in lower yields (around 60%) due to side reactions. Anion exchange resins offer high selectivity, enabling a one-step reaction with yields exceeding 92% and preventing aroma degradation.
Q: What is the advantage of using high-boiling solvents like benzophenone in this process?
A: Using solvents with boiling points between 300-350°C allows for higher reaction temperatures which accelerate the reaction rate. Furthermore, during post-treatment vacuum rectification, the product distills off before the solvent, minimizing product loss and thermal degradation compared to low-boiling solvents.
Q: How does the combined recrystallization method improve product quality?
A: The patent specifies a unique combination of dynamic recrystallization (under stirring) followed by static recrystallization at low temperatures (-10°C to 0°C). This dual approach effectively removes impurities and isomers, achieving product purities up to 99.5% without the need for repeated, solvent-intensive purification cycles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Watermelon Ketone Supplier
The synthesis of Watermelon Ketone via anion exchange resin catalysis represents a significant milestone in the evolution of fine chemical manufacturing, offering a blend of high efficiency and environmental stewardship. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative patent technologies into commercial reality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of this process, including the operation of fixed-bed reactors and the management of high-boiling solvent systems. We adhere to stringent purity specifications and utilize rigorous QC labs to ensure that every batch of Watermelon Ketone meets the exacting standards required by the global fragrance industry.
We invite procurement leaders and R&D directors to collaborate with us to leverage this superior manufacturing technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us directly to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability. Let us help you secure a competitive edge in the market with high-purity, sustainably produced Watermelon Ketone.
