Optimizing Flumioxazin Intermediate Production: A Technical Deep Dive into Novel Nitration and Etherification Pathways
The global demand for high-performance herbicides continues to drive innovation in the synthesis of key agrochemical intermediates. Patent CN101948389A introduces a robust and economically viable method for preparing 2-(5-fluoro-2,4-dinitrophenoxy) acetic acid and its esters, which serve as critical precursors for the widely used herbicide Flumioxazin. This technical disclosure marks a significant departure from traditional synthetic routes that have long plagued manufacturers with high costs and safety concerns. By shifting the starting material strategy to utilize 2,4-difluoronitrobenzene, the inventors have unlocked a pathway that offers superior regioselectivity and operational simplicity. For R&D directors and procurement specialists alike, understanding this shift is crucial for securing a stable supply chain of high-purity agrochemical intermediates. The methodology not only addresses the scarcity of traditional raw materials but also streamlines the purification process, ensuring that the final product meets the stringent quality specifications required for downstream pharmaceutical and agricultural applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this vital intermediate has been fraught with significant technical and economic hurdles that hinder efficient commercial scale-up. Traditional methods often relied on 3-fluorophenol as a starting material, a reagent that is notoriously expensive and difficult to source in bulk quantities, creating a bottleneck for reliable agrochemical intermediate suppliers. Furthermore, the nitration of 3-fluorophenol suffers from poor regioselectivity, generating unwanted isomers that drastically reduce the overall yield to approximately 30%, thereby inflating production costs and waste disposal burdens. Alternative routes utilizing 1,5-dichloro-2,4-dinitrobenzene require harsh fluorination conditions and strict anhydrous environments to prevent hydrolysis, adding complexity to the reactor setup and increasing energy consumption. Other approaches involving methyl bromoacetate introduce severe safety risks due to the compound's strong irritant effects on the respiratory tract and skin, necessitating expensive containment systems and posing liability issues for manufacturing facilities.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages the direct nitration of 2,4-difluoronitrobenzene, a readily available and cost-effective starting material that fundamentally alters the economic landscape of this synthesis. This method achieves high conversion rates through a controlled nitration process using concentrated nitric acid or a mixture with sulfuric acid at moderate temperatures ranging from 60 to 90°C. The subsequent etherification step is equally optimized, utilizing glycolic acid or its esters in the presence of an organic acid binding agent like triethylamine. This combination allows the condensation reaction to proceed smoothly at mild temperatures between -10 and 50°C, eliminating the need for extreme thermal conditions. The result is a streamlined two-step process that delivers yields exceeding 90% in many embodiments, representing a massive improvement over legacy technologies and offering a clear path toward cost reduction in agrochemical intermediate manufacturing.
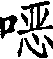
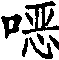
Mechanistic Insights into Selective Nitration and Nucleophilic Substitution
The success of this synthetic route lies in the precise manipulation of electronic effects within the aromatic ring to favor the desired substitution pattern. In the initial nitration step, the presence of the existing nitro group and fluorine atoms on the 2,4-difluoronitrobenzene ring directs the incoming nitronium ion to the 5-position with high fidelity. This regioselectivity is driven by the strong electron-withdrawing nature of the substituents, which deactivates the ring towards electrophilic attack except at the specific position para to the amino-equivalent or meta to the nitro group, depending on the resonance contributors. By carefully controlling the acidity and temperature of the nitrating mixture, the process minimizes the formation of poly-nitrated byproducts or isomers, ensuring that the resulting 1,5-difluoro-2,4-dinitrobenzene is obtained with a purity greater than 95%. This high level of control at the molecular level is essential for maintaining the integrity of the final herbicide active ingredient.
Following nitration, the etherification proceeds via a nucleophilic aromatic substitution (SnAr) mechanism, where the glycolic acid derivative acts as the nucleophile attacking the electron-deficient aromatic ring. The presence of two powerful electron-withdrawing nitro groups ortho and para to the leaving fluorine atom significantly activates the ring for this displacement. The choice of acid binding agent plays a pivotal role here; organic bases like triethylamine effectively scavenge the generated hydrogen fluoride without promoting side reactions such as hydrolysis, which is a common pitfall when using inorganic bases in protic environments. This mechanistic advantage explains why embodiments using triethylamine consistently achieve yields around 90%, whereas those using potassium carbonate often struggle below 50%. Understanding these kinetic and thermodynamic nuances allows process chemists to fine-tune reaction parameters for maximum efficiency and minimal impurity generation.
How to Synthesize 2-(5-Fluoro-2,4-Dinitrophenoxy) Acetic Acid Efficiently
Implementing this synthesis requires careful attention to stoichiometry and temperature control to replicate the high yields reported in the patent literature. The process begins with the preparation of the dinitro intermediate, followed by the coupling with the glycolic acid moiety under basic conditions. Operators must ensure that the molar ratio of the acid binding agent is sufficient to neutralize the acid byproduct without causing excessive exotherms. Detailed standard operating procedures regarding solvent selection, addition rates, and workup protocols are critical for transferring this laboratory success to pilot and commercial scales. For a comprehensive breakdown of the specific operational parameters and safety guidelines, please refer to the standardized synthesis steps outlined below.
- Perform nitration on 2,4-difluoronitrobenzene using concentrated nitric acid or a nitric/sulfuric acid mixture at controlled temperatures between 60-90°C to obtain 1,5-difluoro-2,4-dinitrobenzene.
- Conduct etherification by reacting the dinitro intermediate with glycolic acid or its esters in an organic solvent such as tetrahydrofuran.
- Utilize an organic acid binding agent like triethylamine at mild temperatures ranging from -10 to 50°C to maximize yield and purity before final filtration and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers transformative benefits that extend far beyond simple yield improvements. By transitioning away from scarce and volatile raw materials like 3-fluorophenol, manufacturers can secure a more stable and predictable supply of inputs, mitigating the risk of production stoppages due to raw material shortages. The use of common industrial solvents such as tetrahydrofuran and dichloromethane further simplifies logistics, as these chemicals are widely available and easy to recycle, contributing to a more circular and sustainable manufacturing model. Additionally, the elimination of hazardous reagents like methyl bromoacetate reduces the regulatory burden and insurance costs associated with handling toxic substances, thereby lowering the total cost of ownership for the production facility.
- Cost Reduction in Manufacturing: The shift to 2,4-difluoronitrobenzene as a starting material represents a significant strategic advantage in terms of raw material expenditure. This commodity chemical is produced on a massive scale for other industries, ensuring a competitive price point that is substantially lower than specialized fluorinated phenols. Furthermore, the high selectivity of the nitration step minimizes the loss of valuable intermediates to isomeric byproducts, effectively maximizing the atom economy of the process. When combined with the superior yields achieved through organic base catalysis, the overall cost per kilogram of the final intermediate is drastically reduced, allowing for better margin management in a competitive agrochemical market.
- Enhanced Supply Chain Reliability: Reliability is the cornerstone of a resilient supply chain, and this process enhances it by decoupling production from the bottlenecks of complex multi-step syntheses. The simplified two-step workflow reduces the number of unit operations required, shortening the overall cycle time from raw material intake to finished goods. This agility enables manufacturers to respond more quickly to fluctuations in market demand for Flumioxazin and related herbicides. Moreover, the robustness of the reaction conditions means that production is less susceptible to minor variations in utility supply or environmental conditions, ensuring consistent output quality and delivery schedules for downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but the mild operating conditions of this method facilitate a smooth transition from benchtop to commercial reactors. The ability to run the etherification at temperatures as low as -10°C without requiring cryogenic cooling infrastructure simplifies equipment requirements. From an environmental perspective, the reduction in waste generation due to higher selectivity aligns with increasingly strict global regulations on industrial effluent. The process avoids the generation of heavy metal waste or persistent organic pollutants, making it easier to obtain environmental permits and maintain a positive corporate sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this intermediate. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation. They are intended to provide clarity on the feasibility and advantages of adopting this specific synthetic methodology for your supply chain needs.
Q: What are the primary advantages of using 2,4-difluoronitrobenzene over 3-fluorophenol for this synthesis?
A: Using 2,4-difluoronitrobenzene avoids the high cost and scarcity associated with 3-fluorophenol. Furthermore, it eliminates the low-yield nitration isomer issues found in phenol-based routes, significantly improving overall process efficiency and reducing waste generation.
Q: How does the choice of acid binding agent impact the reaction yield?
A: Experimental data indicates that organic bases like triethylamine provide superior results compared to inorganic bases like potassium carbonate. Triethylamine facilitates smoother reaction kinetics at lower temperatures, resulting in yields exceeding 90%, whereas inorganic bases often struggle to reach 50% yield under similar conditions.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability. It utilizes common solvents like tetrahydrofuran and dichloromethane, operates at mild temperatures (-10 to 50°C), and avoids hazardous reagents like methyl bromoacetate, making it safer and more economically viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(5-Fluoro-2,4-Dinitrophenoxy) Acetic Acid Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition to a new synthetic route requires a partner with deep technical expertise and proven execution capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from development to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific solvent systems and temperature controls required for this nitration and etherification process, guaranteeing adherence to stringent purity specifications. With our rigorous QC labs and commitment to quality assurance, we deliver high-purity agrochemical intermediates that meet the exacting standards of the global herbicide market.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a competitive edge in the agrochemical industry through superior chemistry and reliable supply.
