Advanced One-Step Fluorination for 2-Chloro-5-Trifluoromethylpyrazine Commercial Production
The landscape of fluorinated heterocycle synthesis is undergoing a significant transformation, driven by the urgent need for more efficient and environmentally sustainable manufacturing processes. A pivotal advancement in this domain is documented in patent CN107840828B, which details a novel synthetic method for 2-chloro-5-trifluoromethylpyrazine, a critical building block in the development of advanced pharmaceuticals and agrochemicals. This patent introduces a groundbreaking one-step deoxofluorination strategy that utilizes sulfur tetrafluoride (SF4) and anhydrous hydrogen fluoride (HF) to directly convert 5-chloropyrazine-2-carboxylic acid into the target trifluoromethyl derivative. Unlike traditional multi-step condensations that suffer from cumulative yield losses, this innovative approach achieves a remarkable isolated yield of up to 90% with product purity reaching 99%. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology represents a paradigm shift, offering a robust pathway to secure high-purity pyrazine derivatives while simultaneously addressing the economic and environmental pressures inherent in modern fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2-chloro-5-trifluoromethylpyrazine has been plagued by inefficient multi-step sequences that impose severe bottlenecks on supply chains and inflate production costs. As illustrated in the prior art reaction scheme, the conventional route typically initiates with the condensation of 5,6-diamino-4-hydroxypyrimidine sulfate and 1,1-dibromo-2,2,2-trifluoroacetone, a reaction that notoriously delivers a meager yield of merely 15% for the intermediate 2-amino-5-trifluoromethylpyrazine. This initial inefficiency is compounded by subsequent processing steps, including a diazotization and hydrolysis sequence to generate the hydroxy-pyrazine, followed by a final chlorination using phosphorus oxychloride. Even under optimized conditions, these downstream steps achieve yields of only 70% and 75% respectively, resulting in a catastrophic overall total yield of less than 8%. Furthermore, this lengthy synthetic trajectory generates substantial quantities of hazardous three wastes, creating significant disposal liabilities and complicating regulatory compliance for manufacturers aiming for cost reduction in fine chemical manufacturing.
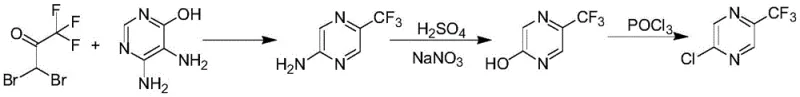
The Novel Approach
In stark contrast to the cumbersome legacy methods, the novel approach disclosed in the patent streamlines the entire synthesis into a single, highly efficient transformation. By leveraging the potent fluorinating capability of the SF4/HF system, the process bypasses the need for complex ring construction and functional group interconversions, directly modifying the carboxylic acid moiety of the readily available 5-chloropyrazine-2-carboxylic acid. This direct conversion not only eliminates the accumulation of impurities associated with isolation of multiple intermediates but also dramatically improves atom economy. The reaction proceeds smoothly in a sealed autoclave environment, where precise control over temperature and pressure facilitates the complete replacement of the carbonyl oxygen atoms with fluorine, followed by rearrangement to the stable trifluoromethyl group. This technological leap transforms a previously marginal process into a commercially viable operation, ensuring that commercial scale-up of complex fluorinated intermediates can be achieved with unprecedented efficiency and reliability.
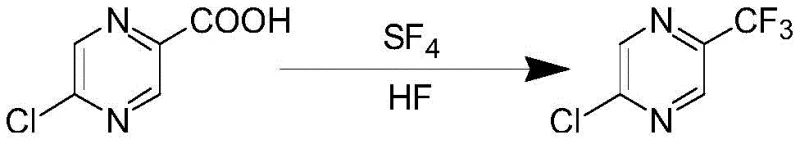
Mechanistic Insights into SF4-Mediated Deoxofluorination
The core of this synthetic breakthrough lies in the intricate mechanism of deoxofluorination mediated by sulfur tetrafluoride in the presence of anhydrous hydrogen fluoride. The reaction initiates with the nucleophilic attack of the carboxylic acid oxygen on the electrophilic sulfur center of SF4, forming a reactive acyl fluoride intermediate. Under the acidic conditions provided by the excess HF, this intermediate undergoes further fluorination to generate a gem-difluoro species, which subsequently eliminates sulfur dioxide and hydrogen fluoride to yield the final trifluoromethyl group. The presence of HF is critical not only as a solvent but also as a catalyst that stabilizes ionic intermediates and enhances the solubility of the organic substrate in the fluorinating medium. Crucially, the patent data indicates that the reaction temperature must be meticulously maintained between 75-80°C; deviations above this range lead to decomposition or side reactions that hinder product yield, while lower temperatures result in incomplete conversion. This delicate thermal balance underscores the importance of precise process engineering in harnessing the full potential of this chemistry for industrial applications.
From an impurity control perspective, this mechanism offers distinct advantages over the traditional diazotization route. The conventional method involves the formation of unstable diazonium salts, which are prone to generating tar-like byproducts and regio-isomers that are difficult to separate. In the SF4-mediated pathway, the reaction specificity is inherently higher because the transformation occurs at the exocyclic carboxyl group without disturbing the aromatic pyrazine ring or the chloro substituent. The use of a closed autoclave system prevents the ingress of moisture, which is a common source of hydrolysis byproducts in fluorination chemistry. Consequently, the crude reaction mixture obtained after neutralization and steam distillation exhibits a purity profile that is significantly cleaner than that of the multi-step alternative. This inherent purity simplifies the downstream rectification process, allowing manufacturers to consistently achieve the stringent purity specifications required for API synthesis without resorting to expensive chromatographic purification techniques.
How to Synthesize 2-Chloro-5-Trifluoromethylpyrazine Efficiently
Implementing this high-yield synthesis requires strict adherence to the operational parameters defined in the patent to ensure safety and reproducibility. The process begins with the careful charging of 5-chloropyrazine-2-carboxylic acid into a corrosion-resistant autoclave, followed by cooling to 0-10°C before the introduction of anhydrous hydrogen fluoride. The subsequent addition of sulfur tetrafluoride gas must be conducted at cryogenic temperatures (-40°C) to manage the exotherm and prevent premature reaction. Once the reagents are charged, the system is heated to the optimal window of 75-80°C for a duration of approximately 6 hours. The detailed standardized synthetic steps, including specific workup procedures involving neutralization with sodium carbonate and final purification via rectification, are outlined in the guide below to assist technical teams in replicating this success.
- Charge 5-chloropyrazine-2-carboxylic acid into an autoclave and cool to 0-10°C, followed by the addition of anhydrous hydrogen fluoride.
- Cool the mixture further to -40°C and introduce sulfur tetrafluoride gas while maintaining a molar ratio of approximately 1: 12:2.2.
- Heat the reaction mixture to 75-80°C for 6 hours, then neutralize with sodium carbonate solution and purify via steam distillation and rectification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates into tangible strategic benefits that extend far beyond simple yield improvements. The transition from a three-step sequence to a single-step process fundamentally alters the cost structure of production by eliminating the labor, energy, and equipment time associated with isolating and handling two distinct intermediates. This step economy results in a drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures and a smaller physical footprint required for production. Moreover, the use of 5-chloropyrazine-2-carboxylic acid as a starting material leverages a supply chain that is generally more robust and accessible than the specialized diamino-pyrimidine precursors required by the old method, thereby enhancing supply chain reliability and reducing the risk of raw material shortages.
- Cost Reduction in Manufacturing: The elimination of two entire synthetic steps removes the need for multiple rounds of solvent recovery, filtration, and drying, which are traditionally major cost drivers in batch processing. By consolidating the synthesis into a single reactor charge, the consumption of utilities such as steam and cooling water is significantly lowered, leading to substantial cost savings in energy-intensive fluorination processes. Additionally, the dramatic increase in yield from less than 8% to 90% means that the effective cost per kilogram of the final active ingredient is reduced exponentially, as the fixed costs of production are amortized over a much larger output of saleable product.
- Enhanced Supply Chain Reliability: Relying on a shorter synthesis route minimizes the number of potential failure points in the production schedule, ensuring more predictable lead times for customers. The starting material, 5-chloropyrazine-2-carboxylic acid, is a stable solid that is easier to transport and store compared to the sensitive liquid reagents and unstable intermediates of the conventional route. This stability allows for larger batch sizes to be produced with greater confidence, enabling suppliers to maintain healthy inventory levels and respond rapidly to fluctuations in market demand without compromising on quality or delivery schedules.
- Scalability and Environmental Compliance: The process is designed for scalability, utilizing standard autoclave equipment that is widely available in fine chemical facilities, facilitating the commercial scale-up of complex fluorinated intermediates from pilot to full production. From an environmental standpoint, the reduction in waste generation is profound; avoiding the heavy metal salts and acidic sludge associated with diazotization and phosphorus oxychloride chlorination simplifies wastewater treatment and lowers disposal costs. This alignment with green chemistry principles not only reduces the environmental burden but also future-proofs the supply chain against increasingly stringent global regulations regarding hazardous waste emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorination technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the primary advantage of the SF4/HF method over traditional condensation routes?
A: The SF4/HF method reduces the synthesis from three steps to a single step, increasing the total yield from less than 8% to over 90% while drastically reducing waste generation.
Q: What are the critical reaction parameters for maximizing yield in this fluorination?
A: Optimal yield is achieved at a reaction temperature of 75-80°C with a molar ratio of acid:HF:SF4 at 1:12:2.2; temperatures exceeding this range can hinder product formation.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes standard autoclave equipment and readily available raw materials, making it highly scalable for industrial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-5-Trifluoromethylpyrazine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced pharmaceutical intermediates requires more than just a patent; it demands deep process expertise and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2-chloro-5-trifluoromethylpyrazine meets the exacting standards required for drug substance manufacturing. Our infrastructure is specifically designed to handle hazardous fluorination chemistries safely and efficiently, providing a secure foundation for your long-term supply needs.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this high-yield route for your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your supply chain resilience and your bottom line.
