Advanced Continuous Flow Synthesis of 4, 6-Dichloro-5-Methoxypyrimidine for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking more efficient pathways for synthesizing critical heterocyclic intermediates, and the recent technological advancements detailed in patent CN108658871B represent a significant leap forward in this domain. This patent introduces a novel preparation method for 4, 6-dichloro-5-methoxypyrimidine, a pivotal intermediate used in the synthesis of the broad-spectrum antibacterial drug sulfadoxine and various pyrimidine salicylate herbicides. Unlike traditional batch processes that suffer from low yields and complex purification requirements, this invention leverages a continuous flow production strategy that starts from readily available malonic diester. The process is designed to be environmentally friendly and safe, eliminating the need for intermediate discharge and significantly streamlining the operational workflow. By integrating chlorination, methoxylation, cyclization, and secondary chlorination into a cohesive sequence, the technology addresses long-standing inefficiencies in heterocyclic chemistry, offering a robust solution for manufacturers aiming to enhance their production capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4, 6-dichloro-5-methoxypyrimidine has been plagued by significant technical and economic hurdles that hinder industrial scalability. One prevalent conventional method involves using 2-methoxy-dimethyl malonate condensed with thiourea to form a mercaptopyrimidine ring, followed by desulfurization with Raney nickel and subsequent chlorination. However, this route is fundamentally flawed because the intermediate, 4, 6-dihydroxy-5-methoxypyrimidine, exhibits high solubility in water, making extraction extremely difficult and resulting in a disappointingly low total yield. Another common approach utilizes expensive formamidino salts as cyclization agents under sodium methoxide catalysis. While chemically feasible, the high cost of formamidinyl salts renders this route economically unviable for large-scale industrial production. Furthermore, existing methods often require multiple discrete purification steps between reactions, which not only increases solvent consumption and waste generation but also extends the overall production cycle time, thereby reducing throughput and increasing the carbon footprint of the manufacturing process.
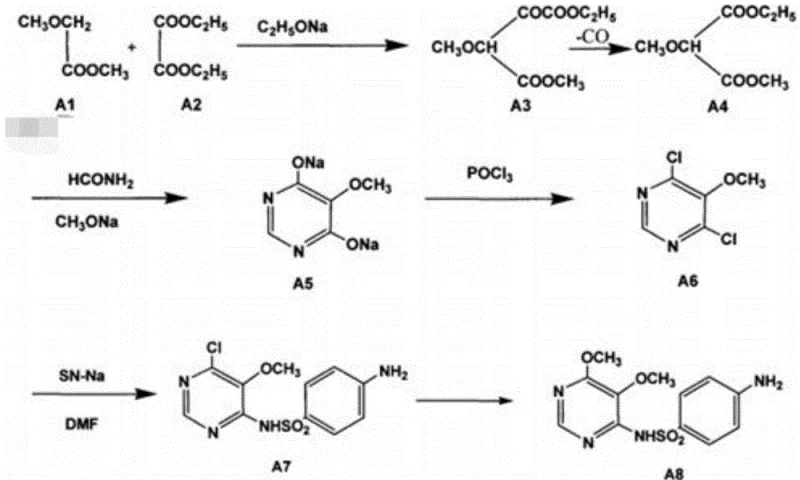
The Novel Approach
In stark contrast to these legacy methods, the innovative process disclosed in CN108658871B utilizes a streamlined, continuous flow architecture that begins with the inexpensive and abundant raw material, malonic diester. This new methodology sequentially executes a chlorination reaction to prepare 2-chloro-malonic diester, followed immediately by a methoxylation reaction to obtain 2-methoxy-malonic diester. The process then advances to a cyclization reaction to generate the 4, 6-dihydroxy-5-methoxy pyrimidine disodium salt, which is directly subjected to a secondary chlorination reaction to yield the final 4, 6-dichloro-5-methoxy pyrimidine. By avoiding the isolation of unstable or difficult-to-handle intermediates and minimizing purification steps, this approach realizes a continuous flow of the production process without discharging waste between stages. This not only enhances the safety profile by containing hazardous intermediates within closed systems but also drastically improves industrial production efficiency, saving at least two hours per batch compared to traditional discontinuous methods.
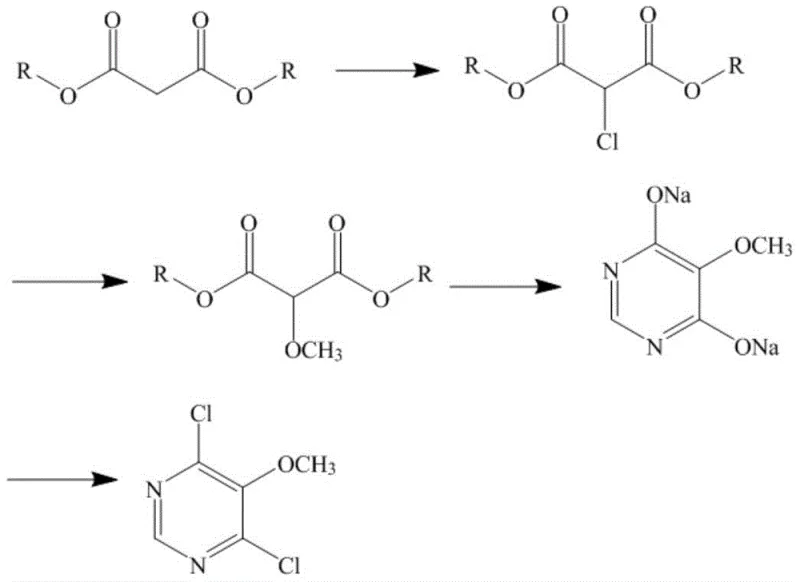
Mechanistic Insights into Malonic Diester-Based Cyclization
The core of this technological breakthrough lies in the precise control of reaction conditions during the transformation of malonic diester into the pyrimidine scaffold. The process initiates with a highly controlled chlorination step where chlorine gas is introduced into a solution of malonic diester (preferably dimethyl malonate) in a solvent like dichloromethane at a strictly maintained temperature of 5 to 15°C. This low-temperature control is critical to prevent over-chlorination and ensure the selective formation of 2-chloro-malonic diester. Following this, the methoxylation reaction is conducted by adding sodium methoxide at temperatures between 10 and 25°C, converting the chloro-ester into 2-methoxy-malonic diester with high conversion rates, indicated by residual starting material levels dropping below 0.1%. The subsequent cyclization step is particularly elegant; by reacting the methoxy-diester with formamide and additional sodium methoxide under reflux, the system forms the 4, 6-dihydroxy-5-methoxy pyrimidine disodium salt. This salt form is advantageous because it has a lower boiling point and reduced skin sensitization risks compared to the free acid, facilitating safer handling in a continuous flow environment.
Furthermore, the mechanism ensures exceptional impurity control through the strategic use of in-process monitoring and direct transfer of reaction mixtures. High-performance gas chromatography is employed to monitor reaction endpoints, ensuring that the residual quantity of precursors like 2-chloro-malonic diester or 2-methoxy-malonic diester is less than 1% before proceeding to the next stage. This rigorous control prevents the carryover of impurities that could complicate the final crystallization. The final secondary chlorination using phosphorus oxychloride (POCl3) converts the disodium salt into the target dichloro product. The workup procedure involves recovering excess POCl3 under reduced pressure and extracting the product with dichloromethane, followed by washing with saturated sodium bicarbonate. This sequence effectively removes acidic byproducts and inorganic salts, resulting in a final product with a purity of more than 97%, often exceeding 99%, and a moisture content of less than 0.5%, demonstrating the robustness of the chemical design.
How to Synthesize 4, 6-Dichloro-5-Methoxypyrimidine Efficiently
Implementing this synthesis route requires careful attention to the sequential addition of reagents and temperature management to maintain the integrity of the continuous flow. The process is designed to be scalable, moving seamlessly from laboratory validation to industrial production by utilizing standard reactor setups equipped for gas absorption and vacuum concentration. Operators must ensure that the molar ratios of key reagents, such as chlorine gas to malonic diester (1:0.8-1) and phosphorus oxychloride to the pyrimidine salt (1.539-2.015:1), are strictly adhered to for optimal yield. The detailed standardized synthesis steps, including specific solvent volumes, stirring rates, and crystallization times, are outlined in the comprehensive guide below to assist technical teams in replicating this high-efficiency protocol.
- Perform chlorination of malonic diester with chlorine gas at 5-15°C to form 2-chloro-malonic diester.
- Conduct methoxylation using sodium methoxide at 10-25°C to obtain 2-methoxy-malonic diester.
- Execute cyclization with formamide and sodium methoxide to generate 4, 6-dihydroxy-5-methoxy pyrimidine disodium salt.
- Complete secondary chlorination with phosphorus oxychloride to yield the final 4, 6-dichloro-5-methoxy pyrimidine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented continuous flow technology offers substantial strategic benefits that extend beyond mere chemical efficiency. The shift from expensive, specialized starting materials like formamidino salts to commodity chemicals like malonic diester fundamentally alters the cost structure of the supply chain, reducing dependency on niche suppliers and mitigating price volatility. Additionally, the elimination of intermediate isolation and purification steps translates directly into reduced labor costs, lower solvent procurement requirements, and decreased waste disposal fees. The ability to save at least two hours per production cycle significantly increases asset utilization rates, allowing manufacturers to meet tight delivery deadlines and respond more agilely to market demand fluctuations without the need for capital-intensive capacity expansion.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of high-cost raw materials with inexpensive malonic diesters, which are widely available in the global chemical market. By removing the need for complex extraction procedures associated with water-soluble intermediates in older methods, the process significantly reduces solvent consumption and energy usage for distillation. The continuous nature of the reaction minimizes downtime between batches, leading to a drastic simplification of the operational workflow and a corresponding decrease in overall manufacturing overheads.
- Enhanced Supply Chain Reliability: Utilizing a continuous flow process inherently improves supply chain resilience by reducing the number of discrete unit operations where bottlenecks can occur. The method's reliance on stable, storable intermediates like the disodium salt allows for better inventory management and production scheduling. Furthermore, the high conversion rates and minimal side reactions ensure consistent output quality, reducing the risk of batch failures that could disrupt downstream API synthesis schedules for pharmaceutical partners.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, the "no discharge" characteristic of this continuous flow process is a major advantage, aligning with increasingly stringent global environmental standards. The containment of hazardous reagents like chlorine gas and phosphorus oxychloride within closed systems enhances workplace safety and reduces the burden on effluent treatment plants. This green chemistry approach facilitates easier regulatory approval for new manufacturing sites and supports corporate sustainability goals by minimizing the ecological footprint of chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis method. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on yield improvements, purity standards, and operational safety. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for commercial scale-up.
Q: Why is the continuous flow method superior for 4, 6-dichloro-5-methoxypyrimidine production?
A: The continuous flow method eliminates intermediate isolation steps, significantly reducing purification time and solvent usage while improving overall yield compared to batch processes.
Q: What are the purity specifications achievable with this new synthesis route?
A: The patented process consistently achieves product purity greater than 97%, often exceeding 99%, with moisture content controlled below 0.5%.
Q: How does this method address the cost issues of traditional formamidino salt routes?
A: By utilizing cheap malonic diester as the starting material instead of expensive formamidino salts, the method drastically lowers raw material costs and simplifies the supply chain.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4, 6-Dichloro-5-Methoxypyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving pharmaceuticals and effective agrochemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 4, 6-dichloro-5-methoxypyrimidine meets the highest international standards, providing our partners with the reliability they need to advance their own product pipelines with confidence.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific applications. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized continuous flow process can drive value and efficiency in your supply chain.
