Scalable Production of High-Purity (E)-Alpha-Asaronum via Novel Catalytic Routes
Scalable Production of High-Purity (E)-Alpha-Asaronum via Novel Catalytic Routes
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for synthesizing bioactive intermediates that ensure both safety and economic viability. Patent CN102199077B introduces a groundbreaking production method for (E)-2,4,5-trimethoxy-1-propenylbenzene, commonly known as Alpha-Asaronum, a compound with significant pharmacological potential including anti-inflammatory and anticonvulsant properties. Unlike traditional extraction methods from natural sources which often suffer from the co-extraction of carcinogenic impurities like beta-asarone, this synthetic approach offers a pristine alternative with purity levels exceeding 99%. The process utilizes 1,2,4-trimethoxybenzene as a key starting material, derived efficiently from p-benzoquinone, and proceeds through a streamlined sequence of propionylation, reduction, and dehydration reactions. This technological advancement represents a paradigm shift in manufacturing this critical intermediate, addressing long-standing issues regarding toxicity, yield, and operational complexity.
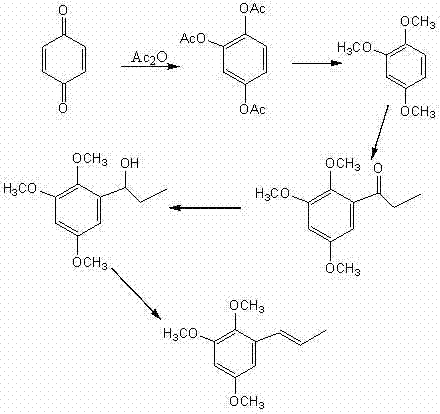
By leveraging readily available raw materials and avoiding toxic organic solvents in critical steps, this method significantly lowers the barrier to entry for large-scale production. The strategic design of the reaction pathway ensures that waste generation is minimized, aligning with modern green chemistry principles while maintaining high reaction yields. For R&D directors and procurement managers alike, this patent offers a compelling solution that balances technical sophistication with commercial pragmatism, ensuring a reliable supply chain for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Alpha-Asaronum has been plagued by significant technical and safety hurdles that hindered its widespread industrial adoption. Traditional methods often relied on the direct extraction from plants, a process inherently fraught with the risk of contaminating the final product with beta-asarone, a known carcinogen that poses severe regulatory and health challenges. Alternative synthetic routes reported in prior art frequently utilized Grignard reagents, such as ethylmagnesium bromide, which necessitate strictly anhydrous conditions and complex handling procedures that are difficult to maintain on a large scale. Furthermore, many existing processes required the use of expensive chiral substrates or sophisticated separation techniques like column chromatography to isolate the desired (E)-isomer from cis-isomer mixtures. These limitations not only inflated production costs but also introduced substantial operational risks, making the consistent supply of high-purity material unreliable for commercial pharmaceutical manufacturing.
The Novel Approach
The methodology outlined in patent CN102199077B fundamentally reengineers the synthesis pathway to overcome these historical bottlenecks through a clever integration of catalytic efficiency and simplified downstream processing. Instead of relying on moisture-sensitive organometallic reagents, the new process employs a robust Friedel-Crafts acylation strategy using propionic anhydride and a novel binary composite catalyst system. This approach allows the reaction to proceed under milder conditions with superior regioselectivity, directly yielding the desired ketone intermediate without the formation of problematic byproducts. Crucially, the final dehydration step utilizes a specific combination of anhydrous cupric sulfate and copper powder, which exhibits exceptional selectivity for the trans-configuration, effectively eliminating the need for difficult geometric isomer separations. By replacing column chromatography with straightforward crystallization and extraction techniques, the process achieves a level of operational simplicity that is rarely seen in complex aromatic syntheses.
Mechanistic Insights into Binary Catalyst Propionylation and Selective Dehydration
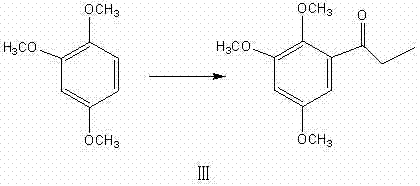
A deep dive into the reaction mechanism reveals the sophistication behind the high yields and purity achieved in this process, particularly in the propionylation and dehydration stages. The propionylation of 1,2,4-trimethoxybenzene is catalyzed by a binary composite system consisting of anhydrous aluminum chloride and zinc chloride in a specific mass ratio, typically around 9:1. This dual-catalyst system creates a synergistic Lewis acid environment that activates the propionic anhydride more effectively than single-component catalysts, facilitating a smoother electrophilic aromatic substitution. The presence of zinc chloride likely modulates the acidity of the aluminum chloride, preventing over-reaction or polymerization while ensuring high conversion rates at temperatures ranging from 15°C to 90°C. This precise control over the catalytic activity is essential for minimizing side reactions and maximizing the yield of 2,4,5-trimethoxypropiophenone, which serves as the pivotal precursor for the subsequent reduction step.
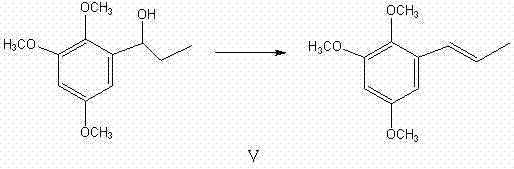
Following the reduction of the ketone to the corresponding alcohol, the final transformation involves a highly selective dehydration reaction that dictates the stereochemistry of the final product. The use of anhydrous cupric sulfate mixed with copper powder as a dehydrating agent is a critical innovation that drives the elimination of water to form the double bond with high (E)-selectivity. Unlike acidic dehydration methods that often produce mixtures of cis and trans isomers requiring tedious purification, this solid-state catalytic dehydration favors the thermodynamically more stable trans-alkene configuration. The mechanism likely involves the coordination of the hydroxyl group to the copper species, facilitating a concerted elimination pathway that preserves the geometric integrity of the molecule. This mechanistic advantage ensures that the final product, (E)-Alpha-Asaronum, is obtained with a purity greater than 99% directly after recrystallization, bypassing the need for further chromatographic purification.
How to Synthesize (E)-Alpha-Asaronum Efficiently
The synthesis of (E)-Alpha-Asaronum via this patented route is designed for seamless translation from laboratory bench to industrial reactor, offering a clear roadmap for process chemists. The procedure begins with the preparation of the 1,2,4-trimethoxybenzene core, followed by the critical propionylation step where temperature control and catalyst loading are paramount for success. Subsequent reduction using borohydride reagents in solvents like tetrahydrofuran or ethyl acetate provides the alcohol intermediate in high yield, setting the stage for the final dehydration. The operational simplicity of each step, characterized by standard workup procedures such as aqueous washing and crystallization, makes this route exceptionally attractive for scale-up. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results.
- Perform acetylation of p-benzoquinone followed by methylation to obtain 1,2,4-trimethoxybenzene.
- Conduct propionylation using a binary composite catalyst (AlCl3/ZnCl2) to form 2,4,5-trimethoxypropiophenone.
- Execute reduction and subsequent dehydration using anhydrous cupric sulfate to yield the final (E)-isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic methodology translates into tangible strategic benefits that extend far beyond simple unit cost metrics. The elimination of expensive and hazardous reagents like Grignard reagents removes a significant layer of complexity from the supply chain, reducing the dependency on specialized raw material vendors who often have long lead times. Furthermore, the avoidance of column chromatography, a technique notoriously difficult to scale and resource-intensive in terms of silica gel and solvent consumption, drastically simplifies the manufacturing infrastructure requirements. This simplification allows for the utilization of standard stainless steel reactors and filtration equipment, thereby lowering capital expenditure barriers for contract manufacturing organizations. The robustness of the process also implies a higher reliability of supply, as the risk of batch failure due to moisture sensitivity or difficult purifications is significantly mitigated.
- Cost Reduction in Manufacturing: The implementation of this process leads to substantial cost savings primarily through the optimization of raw material usage and the reduction of waste disposal expenses. By utilizing a binary catalyst system that is both effective and relatively inexpensive compared to precious metal catalysts, the overall material cost per kilogram of product is significantly lowered. Additionally, the ability to recycle solvents and the reduction in solvent volume required due to the absence of chromatography contribute to a leaner cost structure. The high yield of each step ensures that raw material attrition is minimized, further enhancing the economic efficiency of the production line without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as p-benzoquinone, propionic anhydride, and common inorganic salts ensures a stable and resilient supply chain that is less susceptible to market volatility. Unlike processes dependent on niche chiral building blocks or unstable organometallics, this route leverages widely available feedstocks that can be sourced from multiple global suppliers. This diversification of supply sources reduces the risk of production stoppages due to raw material shortages, ensuring consistent delivery schedules for downstream pharmaceutical customers. The operational safety of the process, devoid of pyrophoric reagents, also minimizes the risk of unplanned shutdowns due to safety incidents.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method offers a cleaner production profile that aligns with increasingly stringent global environmental standards. The reduction in hazardous waste generation, particularly the elimination of silica gel waste from chromatography and the minimization of toxic solvent use, simplifies waste treatment protocols and lowers compliance costs. The process is inherently scalable, allowing for production volumes to be increased from pilot scale to multi-ton commercial quantities without the need for fundamental process re-engineering. This scalability ensures that the supply can grow in tandem with market demand, supporting long-term commercial partnerships and product lifecycle management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of (E)-Alpha-Asaronum, based on the specific innovations detailed in the patent literature. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific advantages of the catalytic systems and purification strategies employed in this method.
Q: What are the advantages of this synthetic method over plant extraction?
A: Unlike plant extraction which risks contamination with carcinogenic beta-asarone, this synthetic route guarantees high purity (>99%) and eliminates toxic impurities, ensuring safety for pharmaceutical applications.
Q: How does the binary catalyst improve the propionylation step?
A: The use of a binary composite catalyst comprising anhydrous aluminum chloride and zinc chloride significantly enhances reaction efficiency and selectivity while reducing overall catalyst costs compared to traditional single-component Lewis acids.
Q: Is column chromatography required for purification in this process?
A: No, the process is designed for industrial scalability by eliminating the need for column chromatography. Purification is achieved through crystallization and standard extraction methods, drastically simplifying downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (E)-Alpha-Asaronum Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable supply of high-quality intermediates for your pharmaceutical development pipelines. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of (E)-Alpha-Asaronum delivered meets the highest industry standards for safety and efficacy. We are committed to supporting your growth with a supply chain that is both resilient and responsive to your evolving requirements.
We invite you to engage with our technical procurement team to discuss how this advanced synthetic route can be tailored to your specific production goals. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this methodology for your specific application. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your manufacturing operations.
