Advanced Synthesis of Isothiocyanate Liquid Crystal Monomers for Commercial Display Applications
Advanced Synthesis of Isothiocyanate Liquid Crystal Monomers for Commercial Display Applications
The rapid evolution of optoelectronic display technologies, ranging from high-resolution touch screens to advanced beam deflectors and spatial light modulators, has created an unprecedented demand for liquid crystal materials with superior physical and chemical stability. Patent CN112279794A, published in early 2021, addresses this critical market need by disclosing a highly optimized synthetic method for isothiocyanate liquid crystal monomers. These specific monomers are renowned for their wide working temperature ranges, low viscosity, and rapid response times, making them indispensable components in next-generation infrared scene generators and field sequential displays. The patent outlines a robust three-stage process involving salt formation, isothiocyanation, and coupling, which collectively overcome the historical challenges of low yields and complex purification associated with traditional alkynyl isothiocyanate synthesis. By leveraging a novel active intermediate strategy, this technology enables the production of a diverse series of liquid crystal monomers with exceptional purity profiles, directly addressing the stringent quality requirements of modern electronic material manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of isothiocyanate-based liquid crystal materials, particularly those incorporating alkynyl linkages, has been plagued by significant technical bottlenecks that hinder efficient commercial production. Traditional routes often suffer from inherently low reaction yields due to the instability of intermediate species and the propensity for side reactions during the introduction of the isothiocyanate (-NCS) functional group. Furthermore, conventional post-treatment protocols are frequently cumbersome, requiring complex extraction and purification sequences to remove stubborn impurities and residual catalysts that can degrade the electro-optical performance of the final liquid crystal mixture. The difficulty in extracting the final product, especially when varying halogen substituents are present, often leads to substantial material loss and increased production costs. Additionally, the direct use of hazardous reagents like thiophosgene without proper stabilization steps poses significant safety risks and environmental compliance challenges in an industrial setting, necessitating a safer and more controlled approach to isothiocyanate group installation.
The Novel Approach
The methodology described in patent CN112279794A introduces a paradigm shift by utilizing a stable dithiocarbamate salt as a pivotal active intermediate, effectively decoupling the isothiocyanation step from the subsequent coupling reactions. This strategic design allows for the isolation and characterization of the isothiocyanate intermediate before it undergoes further transformation, thereby ensuring that only high-quality substrates enter the final coupling stage. By employing triphosgene as a safer alternative to gaseous phosgene derivatives under controlled low-temperature conditions, the process achieves a highly efficient conversion of the amine precursor to the isothiocyanate group with minimal byproduct formation. The subsequent palladium-catalyzed coupling reactions, whether Suzuki or Sonogashira types, proceed with enhanced activity and selectivity when initiated from this purified intermediate. This modular approach not only simplifies the overall workflow but also dramatically improves the final product purity, as evidenced by the high-performance liquid chromatography (HPLC) and gas chromatography (GC) data showing purity levels consistently above 99 percent in the provided examples.
Mechanistic Insights into Palladium-Catalyzed Coupling and Isothiocyanation
The core of this synthetic innovation lies in the precise control of reaction kinetics during the isothiocyanation and coupling phases. The process begins with the formation of a dithiocarbamate salt through the reaction of halogenated aniline with carbon disulfide and triethylene diamine in an ethanol solvent system. This salt acts as a protected reservoir of the nitrogen functionality, which is then selectively converted to the isothiocyanate group using triphosgene in dichloromethane (DCM) at temperatures strictly maintained between -5 and 5°C. This low-temperature regime is critical for suppressing the decomposition of the reactive isothiocyanate species and preventing polymerization or oligomerization side reactions. Following the generation of the active intermediate, such as 2-fluoro-4-bromo-1-isothiocyanatobenzene, the system transitions to a palladium-catalyzed cross-coupling mechanism. Whether utilizing arylboronic acids for biphenyl structures or terminal alkynes for alkynyl derivatives, the presence of the electron-withdrawing isothiocyanate group and halogen substituents requires a carefully tuned catalytic cycle to ensure high turnover numbers.
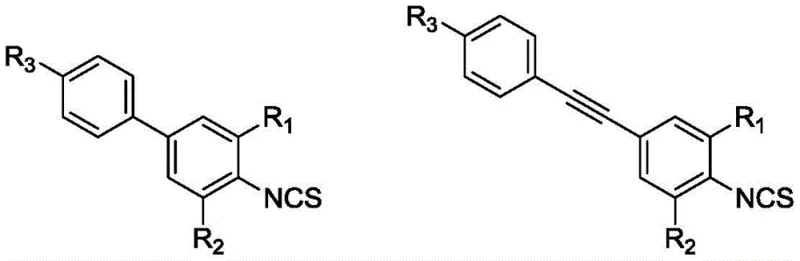
The versatility of this mechanism is demonstrated by its ability to accommodate various substituents, including fluorine, chlorine, bromine, and iodine, although the reactivity order of I > Br > Cl dictates the specific coupling conditions required. For instance, when iodine is present as the leaving group, the oxidative addition step in the palladium cycle proceeds with significantly higher activity, facilitating milder reaction conditions. Conversely, chloro-substituted intermediates may require more vigorous reflux conditions or specialized ligand systems to achieve comparable conversion rates. The patent explicitly details the use of catalysts such as tetrakis(triphenylphosphine)palladium(0) or bis(triphenylphosphine)palladium(II) dichloride, which are effective in mediating the bond formation between the isothiocyanate-bearing aromatic ring and the coupling partner. This mechanistic robustness ensures that a wide library of liquid crystal monomers can be accessed from a common set of intermediates, providing a scalable platform for material discovery and optimization.
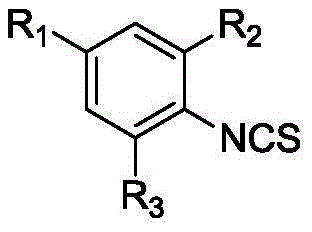
Impurity control is another critical aspect of the mechanistic design, achieved through a combination of selective reactivity and rigorous workup procedures. The use of triphosgene allows for the stoichiometric generation of the isothiocyanate group, minimizing the presence of unreacted amines which could otherwise act as impurities in the final liquid crystal mixture. Post-reaction quenching with water effectively decomposes excess triphosgene, while subsequent washing steps with dilute hydrochloric acid remove basic impurities and residual metal catalysts. The final purification via flash column chromatography or recrystallization ensures that the resulting monomers meet the stringent purity specifications required for electronic applications, where even trace impurities can adversely affect voltage holding ratios and response times. This comprehensive approach to impurity management underscores the suitability of the process for the manufacture of high-value specialty chemicals where quality is paramount.
How to Synthesize Isothiocyanate Liquid Crystal Monomers Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing the importance of precise stoichiometry and temperature control at each stage. The process initiates with the salifying reaction, where halogenated aniline is dissolved in ethanol and treated with carbon disulfide and triethylene diamine in a molar ratio of approximately 1:3-5:3-5, stirring overnight to ensure complete conversion to the salt. This solid intermediate is then isolated and subjected to isothiocyanation in DCM using a triphosgene solution added dropwise at low temperatures, followed by a controlled warm-up to reflux to drive the reaction to completion. The resulting active isothiocyanate intermediate is purified and then immediately utilized in the coupling step, where it reacts with substrates like p-tolylboronic acid or p-n-propylphenylacetylene in the presence of a base and palladium catalyst. Detailed standardized synthesis steps follow below.
- Perform salifying reaction using halogenated aniline, carbon disulfide, and triethylene diamine in ethanol to form a stable dithiocarbamate salt intermediate.
- Execute isothiocyanate conversion by reacting the salt with triphosgene or thiophosgene in DCM at low temperatures (-5 to 5°C) to generate the active isothiocyanate intermediate.
- Conduct palladium-catalyzed coupling (Suzuki or Sonogashira) between the active intermediate and coupling substrates (e.g., boronic acids or alkynes) followed by rigorous purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic methodology offers substantial strategic benefits by streamlining the manufacturing workflow and enhancing overall process reliability. The ability to generate a stable, isolable intermediate decouples the production timeline, allowing for the stockpiling of high-quality precursors that can be rapidly converted into final products based on fluctuating market demand. This flexibility significantly mitigates the risk of supply chain disruptions, as the bottleneck of complex, multi-step synthesis is broken down into manageable, high-yield unit operations. Furthermore, the use of commodity chemicals such as ethanol, DCM, and commercially available palladium catalysts ensures that raw material sourcing remains straightforward and cost-effective, avoiding the dependencies on exotic or proprietary reagents that often plague niche chemical syntheses. The robustness of the process also translates to reduced batch-to-batch variability, a key factor in maintaining long-term supplier qualifications with major electronics manufacturers.
- Cost Reduction in Manufacturing: The optimized reaction pathway eliminates the need for extensive and costly purification steps typically associated with low-yield isothiocyanate syntheses, directly lowering the cost of goods sold. By achieving high conversion rates in the salt formation and isothiocyanation steps, the process minimizes the consumption of expensive starting materials and reduces the volume of waste solvent generated per kilogram of product. The avoidance of hazardous gaseous reagents in favor of solid triphosgene also reduces the capital expenditure required for specialized containment and scrubbing systems, further contributing to overall cost efficiency. Additionally, the high purity of the crude product prior to final chromatography reduces the load on purification columns, extending their lifespan and reducing silica gel consumption.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis, which relies on widely available halogenated anilines and coupling partners, ensures a resilient supply chain that is less susceptible to raw material shortages. The ability to synthesize a wide variety of monomers from a common intermediate platform allows manufacturers to respond quickly to custom requests for specific liquid crystal formulations without retooling entire production lines. This agility is crucial in the fast-paced display industry, where time-to-market for new panel technologies is a critical competitive advantage. Moreover, the scalability of the reaction conditions, which do not require extreme pressures or cryogenic temperatures beyond standard industrial capabilities, facilitates seamless technology transfer from laboratory to commercial scale.
- Scalability and Environmental Compliance: The process is designed with environmental sustainability in mind, utilizing solvents that can be readily recovered and recycled, thereby minimizing the environmental footprint of the manufacturing operation. The replacement of highly toxic thiophosgene gas with solid triphosgene significantly enhances operational safety and simplifies regulatory compliance regarding hazardous air pollutants. The high atom economy of the coupling reactions, combined with efficient post-treatment protocols that reduce aqueous waste loads, aligns with modern green chemistry principles. This commitment to sustainable manufacturing practices not only reduces disposal costs but also strengthens the brand reputation of suppliers among environmentally conscious global clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this isothiocyanate synthesis technology, drawing directly from the experimental data and process descriptions provided in the patent documentation. These insights are intended to clarify the operational parameters and quality benchmarks associated with the production of these high-performance liquid crystal monomers. Understanding these details is essential for technical teams evaluating the feasibility of integrating this supply source into their existing material qualification pipelines.
Q: What are the key advantages of the salt-formation method in patent CN112279794A?
A: The salt-formation method stabilizes the reactive amine group, allowing for a controlled and high-yield conversion to the isothiocyanate group using triphosgene, significantly reducing side reactions compared to direct methods.
Q: How does this synthesis ensure high purity for electronic applications?
A: The process incorporates specific post-treatment steps, including washing with dilute hydrochloric acid and flash column chromatography, which effectively remove palladium residues and unreacted starting materials, achieving purities exceeding 99%.
Q: Can this method be scaled for industrial production of liquid crystal materials?
A: Yes, the method utilizes common solvents like ethanol and DCM and standard catalysts like tetrakis(triphenylphosphine)palladium(0), making it highly adaptable for large-scale commercial manufacturing without requiring exotic reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isothiocyanate Liquid Crystal Monomer Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing the technical expertise and infrastructure necessary to translate complex patent methodologies like CN112279794A into reliable commercial supply chains. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We understand that the production of electronic grade chemicals demands stringent purity specifications and rigorous QC labs capable of detecting trace impurities that could compromise device performance. Our state-of-the-art facilities are equipped to handle the specific solvent systems and catalyst handling requirements of this isothiocyanate synthesis, guaranteeing a consistent supply of high-quality monomers for your display manufacturing needs.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific volume and purity requirements. By leveraging our process optimization capabilities, we can provide a Customized Cost-Saving Analysis that demonstrates the economic benefits of switching to this high-yield methodology. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to validate the quality and compatibility of our isothiocyanate liquid crystal monomers with your current formulation strategies. Together, we can accelerate the development of next-generation display technologies through superior material science and supply chain excellence.
