Scalable Metal-Free Synthesis of N-Substituted Benzothiazin-4-ones for Commercial Production
Scalable Metal-Free Synthesis of N-Substituted Benzothiazin-4-ones for Commercial Production
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign synthetic routes for high-value heterocyclic scaffolds. A recent breakthrough detailed in patent CN114181169A introduces a highly efficient, metal-free methodology for the preparation of N-(substituent) benzothiazin-4-ones, a class of compounds renowned for their potent antitumor, antimalarial, and myocardial protecting activities. This novel approach fundamentally shifts the paradigm from traditional metal-dependent cyclizations to a greener, iodide-mediated process that utilizes readily available reagents such as Selectfluor and sodium iodide. By eliminating the reliance on stoichiometric heavy metals like silver or zinc, this technology not only addresses critical environmental compliance standards but also drastically simplifies the downstream purification processes required for high-purity pharmaceutical intermediates. For global supply chain leaders and R&D directors, this represents a significant opportunity to optimize the manufacturing of complex heterocycles while reducing the overall carbon footprint of the production lifecycle.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-substituted benzothiazin-4-one derivatives has been plagued by significant technical and economic hurdles that hinder large-scale adoption. The first generation of synthetic routes relied heavily on the use of tert-butyl dimethyl (1-methoxy ethyleneoxy) silane in conjunction with zinc iodide, a combination that introduces severe safety and cost liabilities due to the toxicity and high expense of the silicon reagents. Furthermore, these legacy methods often necessitate multi-step preparations of the starting thioether materials, compounding the operational complexity and reducing the overall atom economy of the process. Another prevalent conventional technique involves the reflux of o-carboxyl thiophenol with formaldehyde and amines in toluene, a procedure that is notoriously inefficient, yielding poor quantities of the target product while exposing workers to the pungent and harmful vapors of thiophenol. Perhaps most critically for modern GMP manufacturing, the third common method employs silver oxide as a promoter, which not only inflates raw material costs but also creates a substantial burden for heavy metal removal to meet stringent regulatory limits for API intermediates.
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the innovative method disclosed in CN114181169A offers a streamlined, one-pot solution that operates under metal-free conditions with exceptional efficiency. This novel approach leverages the synergistic effects of 1-chloromethyl-4-fluoro-1,4-diazabicyclo[2.2.2]octane bis(tetrafluoroborate) salt (Selectfluor), sodium iodide, and hydroiodic acid in an acetonitrile solvent system to drive the cyclization forward. The process is remarkably robust, tolerating a wide variety of N-substituents including n-butyl, methyl, benzyl, and cyclohexyl groups, thereby demonstrating superior substrate adaptability compared to the rigid constraints of silver-mediated pathways. By operating at a moderate temperature of 120°C in a sealed vessel, the reaction achieves high conversion rates without the need for exotic catalysts or hazardous reagents. This transition to a metal-free protocol effectively removes the bottleneck of heavy metal scavenging, facilitating a much smoother path to commercial scale-up of complex heterocycles and ensuring a cleaner impurity profile for the final active pharmaceutical ingredients.
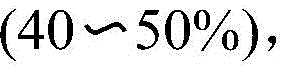
Mechanistic Insights into Metal-Free C-S and C-N Bond Construction
The mechanistic elegance of this transformation lies in its unique ability to orchestrate simultaneous bond formation and cleavage events without the assistance of transition metals. The reaction initiates with the activation of the sulfanyl group on the N-(substituted)-2-sulfanylbenzamide substrate, where the C-S bond undergoes selective cleavage facilitated by the iodide species generated in situ. Following this activation, the chloromethyl moiety from the Selectfluor additive is transferred to the substrate, serving as the crucial methylene bridge that links the nitrogen and sulfur atoms. This dual functionality allows for the concurrent construction of both C-S and C-N bonds, effectively closing the seven-membered benzothiazine ring in a single operational step. The role of the sodium iodide and hydroiodic acid combination is pivotal, acting as a catalytic system that promotes the nucleophilic substitution and subsequent cyclization, a mechanism that is far more atom-economical than the stoichiometric consumption of silver salts seen in prior art. Understanding this mechanism is vital for process chemists aiming to replicate the cost reduction in fine chemical manufacturing, as it highlights the importance of maintaining strict anhydrous conditions and precise reagent ratios to maximize the efficiency of the chloromethyl transfer.
From an impurity control perspective, this metal-free pathway offers distinct advantages by minimizing the formation of metal-complexed byproducts that are difficult to separate. In traditional silver-mediated reactions, trace amounts of silver can persist in the final product, requiring additional purification steps such as specialized resin treatment or recrystallization, which inevitably lead to yield losses. The absence of such metals in the new protocol ensures that the primary impurities are organic in nature, which are generally easier to remove via standard chromatographic techniques or crystallization. Moreover, the use of Selectfluor as a stable, solid reagent enhances the reproducibility of the reaction, reducing batch-to-batch variability that often plagues processes relying on sensitive organometallic catalysts. This level of control is essential for producing high-purity benzothiazin-4-one derivatives that meet the rigorous quality specifications demanded by top-tier pharmaceutical companies, ensuring that the final drug substance is free from genotoxic or cytotoxic metal contaminants.
How to Synthesize N-(N-butyl) benzothiazin-4-one Efficiently
To implement this groundbreaking synthesis in a laboratory or pilot plant setting, operators must adhere to a precise sequence of reagent addition and thermal control to ensure optimal yields. The process begins by charging a sealed reaction vessel with acetonitrile as the solvent, followed by the sequential addition of the N-(substituted)-2-sulfanylbenzamide substrate, the Selectfluor oxidant, sodium iodide, and hydroiodic acid. It is critical that the molar ratios are maintained at approximately 1:1:3:1 for the substrate, Selectfluor, sodium iodide, and hydroiodic acid respectively, as deviations can lead to incomplete conversion or the formation of side products. Once the reagents are combined, the mixture is heated to 120°C and subjected to vigorous stirring for a period of 24 hours to drive the reaction to completion. Detailed standardized synthesis steps see the guide below.
- Charge a sealed tube with acetonitrile, N-(N-butyl)-2-thioethylbenzamide, Selectfluor, sodium iodide, and hydroiodic acid.
- Heat the reaction mixture to 120°C and stir vigorously for 24 hours to ensure complete conversion.
- Concentrate the reaction liquid and purify the crude product via column chromatography to isolate the target ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis route translates into tangible strategic benefits that extend far beyond simple yield improvements. The elimination of expensive and volatile reagents like silver oxide and organosilanes results in a significantly reduced raw material cost base, allowing for more competitive pricing structures in the global market. Furthermore, the simplified workflow reduces the number of unit operations required, which directly correlates to lower energy consumption and reduced labor hours per kilogram of product produced. This efficiency gain is particularly valuable in the current economic climate, where optimizing the cost reduction in fine chemical manufacturing is a top priority for maintaining margin integrity. Additionally, the use of common, commercially available reagents like Selectfluor and sodium iodide mitigates supply chain risks associated with sourcing specialized or regulated catalysts, ensuring a more reliable and continuous supply of critical intermediates for downstream drug production.
- Cost Reduction in Manufacturing: The most immediate financial impact of this technology is the removal of stoichiometric silver oxide, a high-cost reagent that historically inflated the bill of materials for benzothiazinone synthesis. By replacing this with inexpensive sodium iodide and hydroiodic acid, the direct material costs are drastically lowered, while the avoidance of toxic silane reagents further reduces waste disposal expenses. This shift allows manufacturers to allocate resources more effectively, potentially passing savings on to clients or reinvesting in capacity expansion. The qualitative improvement in cost structure makes this route highly attractive for large-volume production campaigns where even marginal savings per kilogram compound into substantial annual financial benefits.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on commodity chemicals rather than bespoke catalysts that may have long lead times or single-source dependencies. Sodium iodide and acetonitrile are widely produced globally, ensuring that production schedules are not disrupted by raw material shortages. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor variations in reagent quality, reducing the risk of batch failures that can delay deliveries to key customers. This reliability is crucial for maintaining the trust of pharmaceutical partners who depend on just-in-time delivery models for their own clinical and commercial manufacturing timelines.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this metal-free process aligns perfectly with modern green chemistry principles, facilitating easier regulatory approval and permitting for new manufacturing lines. The absence of heavy metals simplifies wastewater treatment protocols and reduces the hazard classification of the final product, lowering transportation and storage costs. Scalability is further enhanced by the straightforward workup procedure, which involves simple concentration and chromatography, avoiding complex extraction or filtration steps required to remove metal precipitates. This ease of scale-up ensures that the transition from gram-scale R&D to tonnage-level commercial production can be achieved with minimal technical friction.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology, providing clarity for potential partners evaluating its suitability for their specific projects. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, ensuring accuracy and relevance for industrial application. Understanding these nuances is key to leveraging the full potential of this synthetic route for your specific supply chain needs.
Q: Does this synthesis method involve heavy metal catalysts?
A: No, the method described in CN114181169A is explicitly metal-free, avoiding expensive and toxic reagents like silver oxide or zinc iodide used in prior art.
Q: What is the substrate scope for the sulfur-alkyl group?
A: The process demonstrates excellent adaptability, successfully converting substrates with methyl, ethyl, propyl, butyl, and benzyl thio-groups into the target heterocycle.
Q: How does the yield compare to traditional silver-mediated methods?
A: The new method achieves isolated yields up to 80% for n-butyl derivatives, significantly outperforming older methods that often suffer from low yields or require harsh conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(substituent) benzothiazin-4-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis route and are fully equipped to bring this technology to commercial reality for our global partners. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are designed to handle complex heterocyclic chemistry with precision, supported by rigorous QC labs that enforce stringent purity specifications to meet the highest industry standards. We understand that consistency is key in the pharmaceutical supply chain, and our dedicated process development team is ready to optimize this route specifically for your volume requirements, guaranteeing a reliable supply of high-quality intermediates.
We invite you to collaborate with us to explore how this innovative chemistry can enhance your product portfolio and reduce your overall manufacturing costs. Our technical procurement team is standing by to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating exactly how switching to this metal-free protocol can improve your bottom line. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this advanced synthesis method into your supply chain. Let us be your partner in driving efficiency and innovation in the production of next-generation therapeutic agents.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
