Scalable Green Manufacturing of 3,5-Dichloro-2-Pyridyloxyacetic Acid for Global Agrochemical Markets
Introduction to Patent CN110240560B: A Paradigm Shift in Herbicide Intermediate Synthesis
The global demand for high-efficiency pyridyloxy herbicides, such as triclopyr and fluroxypyr, necessitates robust and environmentally sustainable supply chains for their critical intermediates. Patent CN110240560B introduces a groundbreaking preparation method for 3,5-dichloro-2-pyridyloxyacetic acid derivatives that fundamentally alters the manufacturing landscape. This technology bypasses the historical reliance on hazardous cyanide reagents and expensive glycolic acid precursors, opting instead for a direct etherification with ethylene glycol followed by a selective catalytic oxidation. For R&D directors and procurement strategists, this patent represents a significant opportunity to optimize cost structures while adhering to increasingly stringent environmental regulations. The disclosed methodology not only achieves superior product purity but also streamlines the post-reaction workup, addressing long-standing pain points in the commercial production of these vital agrochemical building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
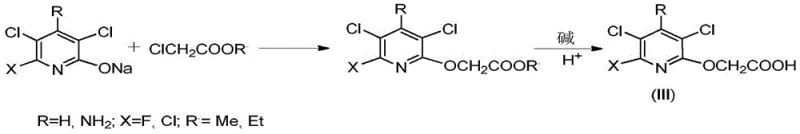
Historically, the industrial synthesis of 3,5-dichloro-2-pyridyloxyacetic acid has been plagued by significant technical and economic inefficiencies. One prevalent route involves the reaction of 3,5-dichloro-2-pyridinol sodium salts with chloroacetic acid esters, as illustrated in the reaction scheme below. While this method can achieve moderate yields, it suffers from severe drawbacks during the hydrolysis and purification stages. The hydrolysis process often leads to the cleavage of the ether bond, regenerating the starting phenol and drastically reducing the overall yield. Furthermore, this pathway generates substantial volumes of alcohol-containing wastewater, creating a heavy burden on waste treatment facilities and inflating operational expenditures.
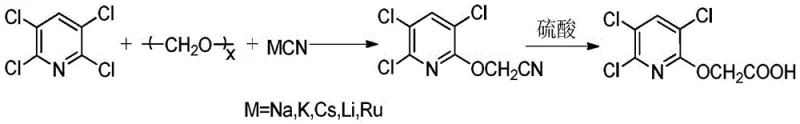
Another traditional approach utilizes 2,3,5,6-tetrachloropyridine reacting with paraformaldehyde and alkali metal cyanides. As depicted in the following diagram, this cyanide-based route produces tetrachloro-pyridoxyacetonitrile which is subsequently hydrolyzed. Although chemically feasible, this method is increasingly untenable in modern manufacturing due to the extreme toxicity of cyanide reagents. The generation of highly toxic cyanide-containing wastewater requires complex and costly treatment protocols to meet environmental discharge standards. Additionally, the overall yield of this process is often limited to around 84%, indicating significant material loss and inefficiency in atom economy.
A third reported method involves the direct reaction of tetrachloropyridine with glycolic acid under phase transfer catalysis. While this avoids cyanide, it introduces other economic barriers. Glycolic acid is a relatively expensive raw material compared to simple diols, and the process requires large quantities of dimethyl sulfoxide (DMSO) as a solvent to dissolve the glycolic acid salts. The recovery of DMSO is energy-intensive, and the phase transfer catalysts often suffer from deactivation or difficult recovery, leading to inconsistent batch quality and higher production costs. These cumulative factors render many conventional routes suboptimal for competitive, large-scale manufacturing.

The Novel Approach
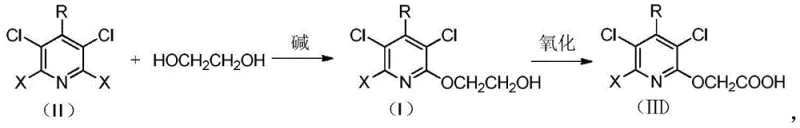
In stark contrast to these legacy methods, the technology disclosed in CN110240560B offers a streamlined, two-step sequence that maximizes efficiency and minimizes environmental impact. The process initiates with the nucleophilic substitution of the chloropyridine substrate using ethylene glycol, a commodity chemical that is abundant and low-cost. This etherification step proceeds smoothly under alkaline conditions without the need for exotic catalysts, yielding the intermediate 3,5-dichloro-2-pyridyloxyethanol with exceptional purity. The subsequent oxidation step utilizes a sophisticated yet practical catalyst system comprising TEMPO, metal nitrates, and metal halides. This allows for the selective oxidation of the primary alcohol to the carboxylic acid using benign oxidants like air or oxygen. By eliminating toxic cyanides and expensive acids, this novel approach provides a clear pathway for cost reduction in herbicide manufacturing while ensuring a safer working environment.
Mechanistic Insights into TEMPO-Catalyzed Oxidation and Etherification
The core innovation of this patent lies in the strategic combination of a robust etherification followed by a highly selective aerobic oxidation. In the first stage, the electron-deficient pyridine ring, activated by multiple chlorine substituents, undergoes nucleophilic aromatic substitution with the alkoxide derived from ethylene glycol. The use of inorganic bases like potassium carbonate or hydroxides facilitates the generation of the reactive alkoxide species in situ. This reaction is remarkably clean, avoiding the formation of poly-ether byproducts that often complicate similar syntheses. The resulting hydroxyethyl ether intermediate serves as a stable precursor that can be isolated or carried forward directly, offering flexibility in process design.
The second stage involves the oxidation of the primary alcohol moiety to the corresponding acetic acid derivative. This transformation is mediated by a catalytic system featuring 2,2,6,6-tetramethylpiperidinyloxy (TEMPO) in conjunction with ferric nitrate and alkali metal halides. Mechanistically, the TEMPO radical acts as the active oxidant, cycling between its nitroxyl, oxoammonium, and hydroxylamine forms to abstract hydrogen from the alcohol substrate. The metal nitrate serves as a co-catalyst to regenerate the active TEMPO species using molecular oxygen or air as the terminal oxidant. This catalytic cycle ensures that the oxidation proceeds with high selectivity, preventing over-oxidation or degradation of the sensitive pyridine ring. The inclusion of metal halides further enhances the reaction rate and stability of the catalyst system, resulting in yields exceeding 90% in many embodiments.
From an impurity control perspective, this mechanism offers distinct advantages. Traditional hydrolysis routes often struggle with ether bond cleavage, leading to the reformation of the starting phenol. However, the oxidative pathway employed here targets the side-chain alcohol specifically, leaving the aryl-oxygen bond intact. The mild reaction conditions, typically conducted at room temperature or slightly elevated temperatures in solvents like toluene or dichloroethane, further suppress thermal degradation pathways. This results in a crude product profile that is significantly cleaner than those obtained from cyanide or ester hydrolysis routes, thereby reducing the load on downstream purification units and enhancing the overall process mass intensity.
How to Synthesize 3,5-Dichloro-2-Pyridyloxyacetic Acid Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and catalyst loading to maximize the benefits of the patented method. The process begins with the preparation of the ether intermediate by heating the chloropyridine substrate with excess ethylene glycol and a base such as triethylamine or potassium hydroxide. Once the etherification is complete, the reaction mixture is worked up to isolate the alcohol intermediate. The subsequent oxidation is performed by dissolving the alcohol in an organic solvent and adding the TEMPO/metal nitrate/halide catalyst cocktail. The reaction is then stirred under an atmosphere of air or oxygen until conversion is complete. For detailed operational parameters and specific embodiment data, please refer to the standardized synthesis guide below.
- React 2,3,5,6-tetrachloropyridine derivatives with ethylene glycol under alkaline conditions to form the pyridyloxyethanol intermediate.
- Oxidize the resulting alcohol intermediate using a TEMPO/metal nitrate catalyst system with air or oxygen as the terminal oxidant.
- Isolate the final 3,5-dichloro-2-pyridyloxyacetic acid derivative through standard workup and crystallization procedures.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this technology translates into tangible strategic advantages beyond mere chemical yield. The shift away from specialized, high-cost reagents like glycolic acid and phase transfer catalysts towards commodity chemicals like ethylene glycol creates a more resilient and cost-effective supply base. Ethylene glycol is produced on a massive global scale for the polyester industry, ensuring consistent availability and price stability even during market fluctuations. Furthermore, the elimination of toxic cyanide reagents removes a significant regulatory hurdle, simplifying the permitting process for new manufacturing lines and reducing the liability associated with hazardous material storage and transport.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by raw material substitution and process simplification. By replacing expensive glycolic acid with low-cost ethylene glycol, the direct material cost per kilogram of product is significantly lowered. Additionally, the avoidance of complex phase transfer catalysts and the use of air as a stoichiometric oxidant eliminate the need for purchasing and recovering costly chemical oxidants. The simplified workup procedure, which avoids energy-intensive distillation steps required in ester hydrolysis routes, further reduces utility consumption and labor costs, leading to a substantially improved gross margin profile for the final intermediate.
- Enhanced Supply Chain Reliability: Reliance on a diverse range of hazardous or specialty chemicals often introduces fragility into the supply chain. This new method relies on robust, widely available inputs that are less susceptible to supply disruptions. The reaction conditions are mild and do not require specialized high-pressure or cryogenic equipment, allowing for production in a wider range of standard chemical reactors. This flexibility enables manufacturers to diversify their production footprint across different facilities without significant capital investment, thereby mitigating the risk of single-site failures and ensuring continuous supply to downstream herbicide formulators.
- Scalability and Environmental Compliance: As environmental regulations tighten globally, the ability to demonstrate a "green" manufacturing process is a critical competitive differentiator. This method generates minimal hazardous waste, particularly by avoiding cyanide-laden effluents and reducing the volume of organic solvent waste through higher yields and simpler purification. The use of air or oxygen as the oxidant means the only byproduct is water, aligning with principles of green chemistry. This environmental profile not only reduces waste disposal costs but also enhances the brand reputation of the supplier, making it easier to qualify as a vendor for multinational agrochemical companies with strict sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a reliable basis for feasibility assessments. Understanding these details is crucial for R&D teams evaluating the technology for pilot plant trials and for procurement teams negotiating supply agreements.
Q: How does this new method improve upon traditional cyanide-based synthesis routes?
A: Unlike traditional methods utilizing toxic alkali metal cyanides which generate hazardous wastewater, this novel approach employs ethylene glycol and catalytic oxidation, eliminating cyanide waste and significantly reducing environmental compliance costs.
Q: What represents the key cost advantage in this preparation method?
A: The process replaces expensive glycolic acid and complex phase transfer catalysts with commodity-grade ethylene glycol and inexpensive inorganic bases, drastically lowering raw material expenditure while simplifying the purification workflow.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the method operates under mild conditions without high temperature or pressure requirements, utilizes air or oxygen as a green oxidant, and demonstrates high yields in pilot examples, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Dichloro-2-Pyridyloxyacetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis methods disclosed in CN110240560B for the global agrochemical sector. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial operations. Our state-of-the-art facilities are equipped to handle the specific catalytic oxidation chemistry described in this patent, maintaining stringent purity specifications through our rigorous QC labs. We are committed to delivering high-quality intermediates that meet the exacting standards required for the synthesis of next-generation herbicides.
We invite potential partners to engage with our technical team to explore how this advanced manufacturing route can be integrated into your supply chain. By leveraging our expertise, you can achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring a seamless transition to this more efficient and sustainable production method.
