Advanced Manufacturing of High-Purity Gadobutrol for Next-Gen MRI Contrast Agents
The landscape of magnetic resonance imaging (MRI) diagnostics has been revolutionized by the advent of gadolinium-based contrast agents, with Gadobutrol standing out as a premier non-ionic macrocyclic agent known commercially under brands such as Gadavist®. Its superior relaxivity and high concentration capabilities make it indispensable for central nervous system and whole-body imaging. However, the manufacturing of this complex chelate has historically faced significant hurdles regarding yield, purity, and the presence of microscopic particulates. The recent patent CN108047151B discloses a breakthrough preparation method that addresses these critical bottlenecks. By utilizing cyclen mono-formaldehyde as a strategic starting material and employing tert-butyl bromoacetate for selective alkylation, this novel route achieves exceptional purity levels exceeding 99.8% while mitigating the formation of hazardous by-products. For pharmaceutical manufacturers seeking a reliable gadobutrol supplier, understanding the technical nuances of this patented pathway is essential for securing a stable supply of high-quality MRI intermediates.
This structural integrity is paramount, as even trace impurities can lead to nephrogenic systemic fibrosis or other adverse reactions. The patent outlines a robust synthetic strategy that not only enhances chemical yield but also solves the persistent industry challenge of flocculent matter in aqueous solutions, a defect that previously rendered batches unsuitable for injectable formulations. By integrating this advanced chemistry into production pipelines, companies can achieve cost reduction in MRI contrast agent manufacturing through improved process efficiency and reduced waste disposal costs associated with toxic reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Gadobutrol has been plagued by selectivity issues and harsh reaction conditions that hinder commercial scalability. Early methods, such as those described in Inorg. Chem. 1997, utilized unprotected cyclen reacting directly with epoxide derivatives. This approach suffered from poor regioselectivity because all four secondary amine groups on the cyclen ring were equally reactive, leading to a complex mixture of di-substituted and tetra-substituted impurities that were notoriously difficult to separate. Alternative routes employed protecting groups like p-toluenesulfonyl (tosyl) to mask three of the amines; however, these processes required extreme temperatures upwards of 170°C, creating significant energy burdens and safety risks during scale-up. Furthermore, the removal of tosyl groups necessitated strong basic reflux conditions, often resulting in incomplete hydrolysis and the generation of persistent mono- or di-deprotected impurities. Other pathways relying on DMF acetals for protection introduced instability, as the protected intermediates were prone to hydrolysis, complicating inventory management and quality control. Perhaps most critically, many legacy processes relied on chloroacetic acid for alkylation, a compound classified as highly toxic and carcinogenic, imposing severe regulatory and environmental compliance costs on manufacturers.
The Novel Approach
The methodology presented in patent CN108047151B represents a paradigm shift towards greener and more controllable chemistry. The core innovation lies in the use of cyclen mono-formaldehyde, where a single nitrogen is temporarily masked by a formyl group. This electronic and steric modification directs the subsequent alkylation exclusively to the remaining three nitrogen atoms when reacted with tert-butyl bromoacetate. This selectivity eliminates the formation of unwanted poly-alkylated side products at the source. Unlike the liquid intermediates of prior art, this process generates a tricarboxylate salt intermediate that precipitates as a solid. This physical property is a game-changer for high-purity gadobutrol production, as solids can be recrystallized to remove soluble impurities before the final, expensive gadolinium complexation step. Additionally, replacing chloroacetic acid with tert-butyl bromoacetate drastically lowers the toxicity profile of the synthesis, simplifying worker safety protocols and waste treatment requirements. The reaction conditions are notably mild, typically proceeding between 40°C and 80°C, which facilitates easier thermal management and reduces the risk of thermal runaway in large reactors.
Mechanistic Insights into Selective Alkylation and Complexation
The chemical elegance of this synthesis is rooted in the precise orchestration of protecting group chemistry and coordination dynamics. The process initiates with the alkylation of cyclen mono-formaldehyde. In a mixed solvent system of water and THF, typically with a volume ratio of 1:1, the presence of an inorganic base like potassium carbonate deprotonates the three free secondary amines. These nucleophiles then attack the electrophilic carbon of tert-butyl bromoacetate via an SN2 mechanism. The use of the bulky tert-butyl ester serves a dual purpose: it prevents intramolecular cyclization side reactions and provides a protecting group that is stable enough to survive the reaction conditions yet labile enough to be removed under controlled hydrolysis. Following alkylation, the tert-butyl esters are hydrolyzed. A key mechanistic advantage here is the ability to perform this hydrolysis in a water-THF mixture without needing to concentrate vast volumes of water, a common energy sink in traditional processes. The result is the tricarboxylate intermediate (Compound 3), which exists as a stable sodium, potassium, or lithium salt.
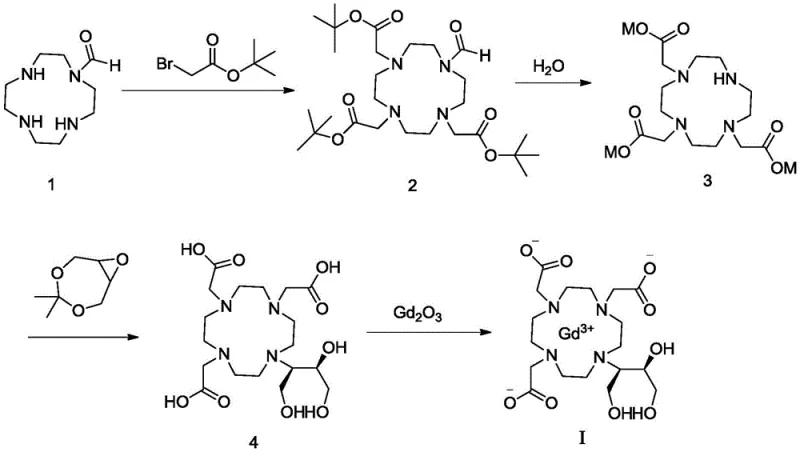
The subsequent step involves the ring-opening of 4,4-dimethyl-3,5,8-trioxabicyclo[5.1.0]octane by the remaining free amine on the macrocycle. This reaction, conducted under reflux in alcohols like isopropanol, installs the crucial hydroxypropyl side chain that defines the Gadobutrol structure. Finally, the free ligand is complexed with a gadolinium source, preferably gadolinium oxide (Gd2O3), in an aqueous environment at 80-100°C. The thermodynamic stability of the Gd(III)-macrocyclic complex drives this reaction to completion. A critical mechanistic insight from this patent is the resolution of the "floc" issue. It was discovered that microscopic insoluble particulates, likely polymeric aggregates or uncomplexed salts, persist even after standard recrystallization. The patent mandates a specific dissolution-filtration-reconcentration cycle, where the final product is dissolved in purified water, passed through a fine filter (e.g., 20μm), and lyophilized or dried. This physical purification step ensures that the final commercial scale-up of complex pharmaceutical intermediates meets the stringent clarity requirements for parenteral drugs.
How to Synthesize Gadobutrol Efficiently
The synthesis of Gadobutrol via this patented route requires strict adherence to stoichiometric ratios and temperature controls to maximize the benefits of the solid intermediate purification. The process is designed to be telescoped where possible, yet the isolation of the tricarboxylate salt is recommended for optimal quality assurance. Operators must ensure that the alkylation step maintains a pH conducive to amine deprotonation without promoting ester hydrolysis prematurely. The hydrolysis step should be monitored to ensure complete conversion to the carboxylate salt, verified by NMR or HPLC, before proceeding to the ring-opening reaction. For detailed operational parameters, including specific solvent volumes, reaction times, and workup procedures, please refer to the standardized protocol below.
- Alkylation of cyclen mono-formaldehyde with tert-butyl bromoacetate to form the protected intermediate.
- Hydrolysis of the tert-butyl esters to obtain the solid tricarboxylate salt intermediate for purification.
- Reaction with 4,4-dimethyl-3,5,8-trioxabicyclo[5.1.0]octane followed by gadolinium complexation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible strategic benefits beyond mere chemical yield. The transition away from hazardous reagents like chloroacetic acid significantly de-risks the manufacturing process, reducing the likelihood of regulatory shutdowns or costly safety incidents that can disrupt supply continuity. The ability to isolate a solid intermediate provides a crucial buffer in the supply chain; this intermediate can be stockpiled and quality-tested independently before committing valuable gadolinium resources to the final complexation step. This modularity enhances reducing lead time for high-purity pharmaceutical intermediates by allowing parallel processing and batch decoupling. Furthermore, the mild reaction temperatures reduce the energy load on manufacturing facilities, contributing to lower operational expenditures and a smaller carbon footprint, which is increasingly important for corporate sustainability goals.
- Cost Reduction in Manufacturing: The elimination of toxic chloroacetic acid removes the need for specialized containment systems and expensive hazardous waste disposal protocols, leading to substantial cost savings. Additionally, the avoidance of high-temperature reactions (170°C) reduces energy consumption significantly. The solid-state purification of the intermediate minimizes the loss of expensive starting materials compared to liquid-liquid extraction methods used in older protocols, thereby improving the overall mass balance and economic efficiency of the production line.
- Enhanced Supply Chain Reliability: By utilizing stable, commercially available starting materials like tert-butyl bromoacetate and cyclen mono-formaldehyde, the process reduces dependency on custom-synthesized, unstable reagents. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in utility supplies or environmental conditions, ensuring consistent output. The ability to produce a high-purity intermediate that can be stored creates a strategic inventory buffer, safeguarding against upstream supply shocks and ensuring continuous availability for downstream formulation.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing common solvents like THF, water, and isopropanol which are easily recovered and recycled in industrial distillation columns. The avoidance of heavy metal catalysts or toxic alkylating agents simplifies the environmental permitting process for new manufacturing sites. The final filtration step to remove flocs ensures that the product meets global pharmacopeia standards without requiring reprocessing, which streamlines the release timeline and accelerates time-to-market for finished diagnostic agents.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented Gadobutrol synthesis. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on quality control and process safety.
Q: How does this patent address the issue of flocculent impurities in Gadobutrol?
A: The patent introduces a specific post-synthesis purification step where the final product is dissolved in purified water, filtered through a membrane (e.g., 20μm), and then re-concentrated. This effectively removes insoluble particulates that cause turbidity in injectable solutions.
Q: Why is tert-butyl bromoacetate preferred over chloroacetic acid in this synthesis?
A: Chloroacetic acid is highly toxic and poses significant safety risks during large-scale manufacturing. Tert-butyl bromoacetate is safer to handle, and the resulting tert-butyl ester groups can be easily removed under mild hydrolysis conditions, improving the overall environmental profile of the process.
Q: What represents the key quality control advantage of the tricarboxylate intermediate?
A: Unlike previous methods where intermediates were oils or unstable liquids, this process yields the tricarboxylate intermediate as a solid salt. This physical state allows for crystallization and rigorous purification before the final complexation step, ensuring higher final purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gadobutrol Supplier
As the global demand for high-resolution MRI diagnostics continues to surge, the need for a dependable source of ultra-pure Gadobutrol has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced synthetic methodologies like the one described in CN108047151B to deliver superior pharmaceutical intermediates. Our facility is equipped with state-of-the-art reactor trains capable of handling complex macrocyclic chemistry under strictly controlled GMP conditions. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet both clinical trial demands and full-scale commercial launch requirements. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which utilize HPLC and ICP-MS to verify that every batch meets the highest international standards for metal content and organic impurities.
We understand that cost efficiency and supply security are top priorities for your organization. That is why we invite you to engage with our technical procurement team for a Customized Cost-Saving Analysis tailored to your specific volume requirements. Whether you require specific COA data for regulatory filings or detailed route feasibility assessments for technology transfer, our experts are ready to provide the support you need. Partner with us to secure a sustainable, high-quality supply of Gadobutrol that empowers your diagnostic portfolio and drives patient care forward.
