Scaling High-Purity DTTA Production: A Technical Breakthrough in Quinacridone Precursor Synthesis
The global demand for high-performance organic pigments, particularly the quinacridone series known for their exceptional lightfastness and thermal stability, necessitates a reliable supply chain for critical intermediates like 2,5-diparamethylaniline terephthalic acid (DTTA). Patent CN101830820B introduces a transformative manufacturing methodology that addresses long-standing inefficiencies in DTTA production. By shifting from traditional volatile organic solvents to a novel ethylene glycol-water dispersion system, this technology enables the synthesis of DTTA with unprecedented purity and yield. For R&D directors and procurement specialists in the fine chemical sector, this patent represents a pivotal opportunity to optimize the commercial scale-up of complex pigment intermediates. The process leverages the unique solubility characteristics of dimethyl succinosuccinate (DMSS) in high-boiling glycol systems to overcome the thermodynamic barriers that have historically plagued this synthesis.
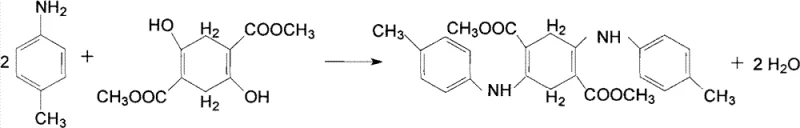
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of DTTA has been hindered by the reliance on low-boiling organic solvents such as methanol or ethanol. In these conventional systems, the solubility of the key raw material, DMSS, is severely limited at ambient pressures, leading to heterogeneous reaction conditions that impede efficient molecular collision. Because the boiling points of methanol and ethanol are relatively low, operators are forced to run reactions under elevated pressure to achieve temperatures sufficient for dissolution, introducing significant safety hazards related to flammability and explosion risks. Furthermore, the condensation by-product, water, complicates the recovery of these organic solvents, often resulting in azeotropic mixtures that are energy-intensive to separate. The resulting solid products are frequently difficult to filter and wash, trapping unreacted starting materials and lowering the overall purity of the final pigment intermediate.
The Novel Approach
The innovative process detailed in the patent data fundamentally re-engineers the reaction medium by employing a dispersion of ethylene glycol and water. This strategic substitution capitalizes on the high boiling point of ethylene glycol, allowing the reaction temperature to be safely elevated to the 110°C to 140°C range without requiring excessive pressurization. At these elevated temperatures, DMSS exhibits dramatically increased solubility, facilitating a homogeneous reaction phase prior to precipitation of the product. This transition from a difficult solid-solid or solid-liquid interface to a more manageable suspension system ensures that the condensation reaction proceeds to near-completion. Additionally, the use of an aqueous glycol system simplifies the downstream processing, as the product salts exhibit favorable solubility profiles that allow for effective purification via adsorption and recrystallization, ultimately delivering a superior quality high-purity fine chemical intermediate.
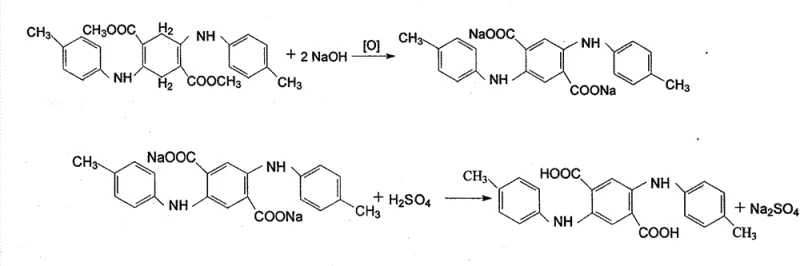
Mechanistic Insights into Condensation and Oxidative Aromatization
The core chemical transformation involves a two-stage cascade beginning with the acid-catalyzed condensation of two equivalents of p-toluidine with one equivalent of DMSS. In the presence of a strong acid catalyst such as sulfuric acid or hydrochloric acid, the amine groups of the p-toluidine nucleophilically attack the carbonyl carbons of the DMSS ester groups. This step is critical and is driven forward by the removal of methanol by-products and the precise control of temperature within the glycol dispersion. The result is the formation of 2,5-di-p-toluidino-3,6-dihydrodimethyl terephthalate, a yellow solid intermediate that precipitates out of the hot glycol solution as a fine suspension, preventing agglomeration and ensuring uniform particle size distribution which is vital for subsequent reaction kinetics.
Following condensation, the process moves to a simultaneous hydrolysis and oxidative aromatization step. A mineral alkali, typically sodium hydroxide or potassium hydroxide, is introduced to hydrolyze the ester groups, while a specialized oxidant such as m-nitrobenzenesulfonate sodium salt facilitates the dehydrogenation of the cyclohexadiene ring to form the aromatic terephthalic acid core. This oxidative step is exothermic and requires careful thermal management to prevent runaway reactions. The resulting disodium salt of DTTA is highly soluble in the hot aqueous glycol phase, allowing insoluble impurities to be filtered off. Subsequent acidification with sulfuric acid protonates the carboxylate groups, causing the final DTTA product to crystallize out of the solution with high structural integrity and minimal impurity inclusion, satisfying the rigorous specifications required for reliable dye intermediate supplier standards.
How to Synthesize 2,5-Diparamethylaniline Terephthalic Acid Efficiently
To implement this synthesis effectively, manufacturers must adhere to a precise sequence of unit operations that maximize the benefits of the glycol-water solvent system. The process begins with the careful batching of DMSS and p-toluidine in a reactor equipped with efficient agitation and heating capabilities. The detailed standardized synthetic steps, including specific molar ratios, temperature ramping rates, and pressure controls, are outlined in the guide below to ensure reproducibility and safety during scale-up.
- Mix DMSS and p-toluidine with ethylene glycol and water, then heat to 110-140°C to dissolve reactants into a biphasic system.
- Add acid catalyst (e.g., sulfuric acid) and perform condensation at 100-140°C under pressure to form the dihydro intermediate.
- Introduce mineral alkali and oxidant to the slurry, heating to 90-140°C for simultaneous hydrolysis and oxidation to form the sodium salt.
- Filter the mixture, treat the liquid phase with adsorbents, and acidify the filtrate to pH 2-6 to precipitate pure DTTA crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this glycol-based synthesis route offers profound logistical and economic benefits compared to legacy alcohol-based processes. The elimination of large volumes of volatile organic solvents reduces the regulatory burden associated with hazardous material storage and transport, while simultaneously lowering the capital expenditure required for explosion-proof infrastructure. The ability to recycle the mother liquor directly back into the batching stage significantly diminishes raw material waste, creating a closed-loop system that enhances overall process sustainability. These factors combine to create a robust supply chain capable of meeting the demanding volume requirements of the global pigment industry without compromising on delivery timelines or product consistency.
- Cost Reduction in Manufacturing: The shift to an ethylene glycol and water system eliminates the need for complex and energy-intensive solvent recovery distillation columns typically required for methanol or ethanol. By operating at atmospheric or near-atmospheric pressures with higher boiling solvents, the process reduces utility consumption for cooling and compression. Furthermore, the high conversion rate minimizes the loss of expensive p-toluidine and DMSS, leading to substantial cost savings in raw material procurement. The simplified filtration and washing steps also reduce labor and processing time, contributing to a lower overall cost of goods sold.
- Enhanced Supply Chain Reliability: The raw materials utilized in this process, specifically DMSS and p-toluidine, are commodity chemicals with stable global availability, reducing the risk of supply disruptions. The robustness of the reaction conditions means that production schedules are less susceptible to delays caused by equipment maintenance or safety shutdowns associated with high-pressure alcohol systems. This reliability ensures a consistent flow of high-purity pigment intermediates to downstream customers, allowing them to maintain their own production schedules for quinacridone pigments without interruption.
- Scalability and Environmental Compliance: The aqueous nature of the waste streams simplifies wastewater treatment, as the glycol content can be managed more effectively than mixed organic solvent wastes. The process generates significantly less hazardous waste, aligning with increasingly stringent environmental regulations in major manufacturing hubs. From a scalability perspective, the suspension chemistry translates seamlessly from pilot plant to multi-ton commercial reactors, as the heat transfer and mixing dynamics are easier to control in a high-boiling liquid medium than in a volatile gas-liquid system.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of DTTA using this advanced methodology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the operational parameters and quality outcomes. Understanding these details is essential for partners looking to integrate this intermediate into their high-performance pigment formulations.
Q: Why is ethylene glycol preferred over methanol in DTTA synthesis?
A: Ethylene glycol has a significantly higher boiling point than methanol, allowing reaction temperatures of 110-140°C. This ensures complete dissolution of DMSS and drives the condensation reaction to completion without the safety risks associated with high-pressure alcohol systems.
Q: What is the typical yield of this novel DTTA preparation method?
A: The patented process demonstrates exceptional efficiency, achieving yields exceeding 98% based on DMSS consumption, which is a substantial improvement over traditional methods that suffer from incomplete reactions and difficult product isolation.
Q: How does this process impact environmental compliance?
A: By utilizing a water-ethylene glycol system, the process eliminates the need for large volumes of volatile organic solvents. The aqueous mother liquor can be recycled directly back into the batching stage, drastically reducing wastewater generation and solvent recovery costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5-Diparamethylaniline Terephthalic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the performance of final pigment products. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of DTTA meets the exacting standards required for premium quinacridone pigment synthesis. Our facility is equipped to handle the specific thermal and mixing requirements of the glycol-dispersion process described in the patent data.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific application needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to our supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of technical confidence and supply security.
