Advanced Synthesis of 4,4'-Diphenyl Ether Dicarboxylic Acid for High-Performance Polymers
Advanced Synthesis of 4,4'-Diphenyl Ether Dicarboxylic Acid for High-Performance Polymers
The global demand for high-performance thermoplastic liquid crystal polymers (TLCP) and modified polyesters continues to surge, driven by the electronics and aerospace sectors requiring materials with superior heat resistance and mechanical strength. At the heart of these advanced materials lies 4,4'-diphenyl ether dicarboxylic acid, a critical monomer that imparts exceptional thermal stability and processability to polymer chains. A recent technological breakthrough detailed in patent CN111018706A introduces a robust, copper-catalyzed synthesis method that addresses longstanding inefficiencies in producing this vital chemical building block. By shifting away from harsh oxidation protocols and multi-step nitro-reductions, this novel approach leverages a sophisticated ligand-assisted coupling mechanism to achieve total reaction yields exceeding 80 percent. For R&D directors and procurement strategists, this represents a pivotal opportunity to secure a more reliable polymer additive supplier capable of delivering high-purity intermediates with a drastically simplified environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
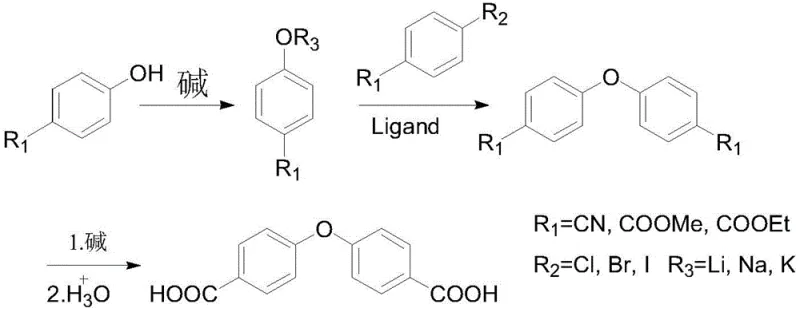
Historically, the industrial preparation of 4,4'-diphenyl ether dicarboxylic acid has been plagued by significant technical and economic hurdles that hinder scalable manufacturing. Early methodologies, such as those pioneered by the Tanhua research group, relied on the coupling of p-methylphenol and p-bromotoluene catalyzed by cuprous chloride, followed by a vigorous oxidation step using potassium permanganate to convert methyl groups into carboxylic acids. This two-step sequence suffers from extremely low overall yields due to the non-selective nature of the oxidation and the difficulty in controlling side reactions during the initial coupling. Furthermore, alternative routes utilizing p-nitrobenzoic acid as a starting material involve multiple synthetic transformations that generate substantial quantities of toxic nitrogen dioxide gas, creating severe environmental compliance challenges and necessitating expensive scrubbing infrastructure. These conventional pathways not only inflate production costs through excessive reagent consumption but also introduce complex purification burdens that compromise the final purity required for high-end electronic applications.
The Novel Approach
In stark contrast to these archaic processes, the methodology disclosed in CN111018706A streamlines production into a highly efficient sequence centered on a direct etherification coupling reaction. This innovative route bypasses the need for aggressive oxidants and toxic nitro-intermediates by directly coupling p-hydroxybenzoate derivatives with p-halobenzoates under optimized thermal conditions. The process initiates with a controlled dehydration step at 110-120°C to activate the phenolic substrate, followed by a high-temperature coupling phase between 145-195°C facilitated by a specialized copper-ligand catalyst system. This strategic modification not only simplifies the operational workflow by reducing the number of unit operations but also significantly enhances the atom economy of the synthesis. By eliminating the generation of hazardous nitrogen oxides and minimizing solvent usage through efficient recovery loops, this novel approach offers a sustainable pathway for cost reduction in polymer synthesis additives manufacturing while ensuring consistent product quality.
Mechanistic Insights into Copper-Ligand Catalyzed Etherification
The core innovation of this synthesis lies in the sophisticated modulation of the copper catalyst's electronic environment through the introduction of specific chelating ligands. In traditional Ullmann-type ether syntheses, simple cuprous halides often suffer from rapid deactivation via disproportionation or aggregation, leading to poor conversion rates and the formation of homocoupling byproducts. The patented method overcomes these kinetic barriers by complexing cuprous halides (such as CuCl, CuBr, or CuI) with bidentate or multidentate ligands like 8-hydroxyquinoline, triphenylphosphine, or diphosphines (dppp, dppe, dppm). This coordination stabilizes the copper center in its active +1 oxidation state, facilitating the oxidative addition of the aryl halide and the subsequent reductive elimination to form the diaryl ether bond. The result is a dramatic increase in catalytic turnover frequency and selectivity, allowing the coupling reaction yield to surpass 75 percent, a marked improvement over the 50-55 percent typical of uncatalyzed or poorly ligated systems.
Beyond the coupling mechanics, the process incorporates a meticulously controlled hydrolysis stage to ensure the final product meets stringent purity specifications required for optical and electronic grade polymers. Following the coupling reaction, the resulting diester intermediate is subjected to alkaline hydrolysis at moderate temperatures of 50-80°C, a condition gentle enough to prevent degradation of the sensitive ether linkage while ensuring complete saponification of the ester groups. The subsequent acidification and crystallization steps are designed to precipitate the 4,4'-diphenyl ether dicarboxylic acid with minimal inclusion of inorganic salts or unreacted starting materials. This precise control over the downstream processing parameters effectively suppresses the formation of colored impurities and regio-isomers, thereby delivering a white solid product with purity levels reaching 99.4 percent as demonstrated in the patent examples. Such rigorous impurity control is essential for maintaining the optical clarity and thermal stability of the resulting liquid crystal polymers.
How to Synthesize 4,4'-Diphenyl Ether Dicarboxylic Acid Efficiently
Implementing this advanced synthesis route requires strict adherence to the defined thermal profiles and stoichiometric ratios to maximize the benefits of the ligand-accelerated catalysis. The process begins with the activation of the phenolic precursor through dehydration, creating a nucleophilic phenoxide species ready for attack. This is immediately followed by the introduction of the electrophilic aryl halide and the pre-complexed copper catalyst, where temperature ramping must be managed carefully to balance reaction rate against potential side reactions. The final hydrolysis step serves as both a functional group transformation and a purification opportunity, leveraging solubility differences to isolate the target acid. For detailed operational parameters, including specific solvent choices and molar equivalents, please refer to the standardized protocol outlined below.
- Dehydrate p-hydroxybenzoate with alkali in solvent at 110-120°C under nitrogen protection to form a reactive mixed solution.
- Add copper catalyst with ligand and p-halobenzoate to the mixture, heating to 145-195°C for coupling to form the diester intermediate.
- Hydrolyze the diester in alkaline water solution at 50-80°C, adjust pH to acidic, and crystallize to obtain the final dicarboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis technology translates into tangible strategic advantages regarding cost structure and supply continuity. The elimination of expensive and hazardous oxidizing agents like potassium permanganate removes a significant variable cost component while simultaneously reducing the regulatory burden associated with handling strong oxidizers. Furthermore, the use of readily available commodity chemicals such as methyl p-hydroxybenzoate and methyl p-chlorobenzoate ensures a stable raw material supply base that is less susceptible to the volatility often seen with specialized nitro-compounds. The simplified workup procedure, which involves straightforward extraction and crystallization rather than complex chromatographic separations, drastically reduces processing time and solvent consumption, leading to substantial cost savings in utility and waste disposal expenditures.
- Cost Reduction in Manufacturing: The transition to a ligand-stabilized copper catalyst system fundamentally alters the economics of production by boosting overall yield from roughly 50 percent to over 80 percent. This efficiency gain means that for every ton of raw material input, manufacturers can extract significantly more saleable product, effectively lowering the cost of goods sold without compromising quality. Additionally, the ability to recover and recycle excess starting materials and solvents further compresses the operational budget, making this route highly competitive for large-scale commercial production of electronic chemical intermediates.
- Enhanced Supply Chain Reliability: By relying on a robust two-step sequence that avoids the generation of toxic gases and unstable intermediates, facilities can operate with higher uptime and fewer safety-related shutdowns. The process tolerance for various halogen substituents (chlorine, bromine, iodine) provides procurement teams with the flexibility to source the most economically viable halobenzoate available on the global market at any given time. This adaptability mitigates the risk of supply disruptions caused by regional shortages of specific reagents, ensuring a continuous flow of high-purity polymer monomers to downstream customers.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of heavy metal contamination in the final product simplify the scale-up process from pilot plant to full commercial production. Unlike methods requiring high-pressure hydrogenation or cryogenic conditions, this thermal coupling process can be executed in standard glass-lined or stainless steel reactors commonly found in fine chemical plants. The significant reduction in three-waste generation aligns perfectly with increasingly stringent global environmental regulations, future-proofing the supply chain against potential carbon taxes or waste disposal levies.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis pathway. Understanding these nuances is critical for R&D teams evaluating the feasibility of technology transfer and for quality assurance personnel establishing incoming inspection criteria. The answers provided are derived directly from the experimental data and comparative analysis presented in the underlying patent documentation, ensuring accuracy and relevance for industrial application.
Q: How does the new copper-ligand catalyst system improve yield compared to traditional methods?
A: Traditional methods using cuprous chloride alone often suffer from low selectivity and incomplete conversion, yielding only 50-55%. The novel method complexes copper salts with ligands like 8-hydroxyquinoline or triphenylphosphine, which stabilizes the catalyst and significantly increases reaction selectivity, pushing yields over 75% for the coupling step and over 80% total yield.
Q: What are the environmental advantages of this synthesis route?
A: Unlike previous routes involving p-nitrobenzoic acid which generate large amounts of toxic nitrogen dioxide gas, this method utilizes a direct coupling of esters or nitriles. It avoids harsh oxidation steps with potassium permanganate, resulting in significantly less three-waste generation and a more environmentally friendly production profile suitable for modern green chemistry standards.
Q: Can this process be adapted for different ester groups or nitrile precursors?
A: Yes, the process is highly flexible. The patent specifies that p-hydroxybenzoate and p-halobenzoate can be substituted with methyl, ethyl, or cyano variants. This allows manufacturers to produce dimethyl, diethyl, or dicyano intermediates depending on downstream processing needs, offering versatility in supply chain management for different polymer applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Diphenyl Ether Dicarboxylic Acid Supplier
As the demand for high-performance polyarylates and modified PET resins accelerates, securing a partner with deep technical expertise in monomer synthesis is paramount for maintaining product differentiation. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent quality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 4,4'-diphenyl ether dicarboxylic acid meets the exacting standards required for liquid crystal fiber and insulating material applications. We understand that in the fast-paced electronics and aerospace industries, supply consistency is just as critical as chemical performance.
We invite forward-thinking organizations to collaborate with us to optimize their material sourcing strategies and reduce overall manufacturing overhead. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage potential partners to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value across your entire supply chain. Let us help you bridge the gap between laboratory innovation and industrial reality with our proven synthesis capabilities.
