Scalable One-Pot Synthesis of 2-Hydroxy-4-Carboxyquinoline for Commercial Pharmaceutical Production
The pharmaceutical industry continuously seeks robust, scalable, and environmentally sustainable pathways for synthesizing critical heterocyclic intermediates. Patent CN108101842B introduces a groundbreaking preparation method for 2-hydroxy-4-carboxyquinoline, a pivotal building block in the manufacture of cinchocaine hydrochloride and various functional materials. This technology represents a significant departure from legacy synthetic routes by utilizing a direct one-pot condensation strategy involving isatin and malonic acid. By eliminating complex multi-step sequences and hazardous reagents, this innovation addresses the growing demand for greener chemistry while maintaining exceptional product quality. For global procurement and R&D teams, understanding the mechanistic elegance and operational simplicity of this patent is essential for securing a reliable supply chain of high-purity pharma intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-hydroxy-4-carboxyquinoline has been plagued by significant operational and environmental drawbacks that hinder large-scale efficiency. Traditional routes often involve the initial formation of N-acetylisatin through the reflux of isatin with acetic anhydride, a process notorious for generating intense, unpleasant odors and requiring rigorous containment measures. Subsequent steps typically necessitate hydrolysis and rearrangement in aqueous sodium hydroxide solutions, which not only consume vast quantities of water but also generate substantial volumes of alkaline wastewater that require costly treatment before discharge. Furthermore, alternative literature methods employing microwave irradiation, while effective on a small laboratory scale, fail to translate effectively to industrial reactors due to heat transfer limitations and equipment constraints. These conventional approaches collectively result in high production costs, extended lead times, and elevated safety risks associated with handling corrosive bases and volatile anhydrides.
The Novel Approach
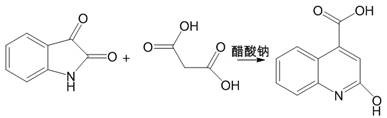
In stark contrast, the novel methodology disclosed in the patent streamlines the entire synthesis into a single, cohesive unit operation that maximizes atom economy and minimizes waste generation. By directly reacting isatin with malonic acid in the presence of sodium acetate within a glacial acetic acid medium, the process bypasses the need for isolated intermediate formation and harsh alkaline conditions. This one-pot strategy not only simplifies the equipment footprint required for production but also drastically reduces the labor intensity associated with multiple filtration and transfer steps. The use of glacial acetic acid serves a dual purpose as both a solvent and a reaction medium, which can be efficiently recovered and reused via reduced-pressure evaporation, thereby closing the loop on solvent consumption. This approach effectively resolves the safety hazards of previous methods, such as the use of explosive sodium hydride, offering a pathway that is inherently safer and more aligned with modern environmental, health, and safety (EHS) standards.
Mechanistic Insights into Sodium Acetate-Catalyzed Cyclization
The core of this technological advancement lies in the efficient condensation and cyclization mechanism facilitated by the mild basicity of sodium acetate. In this system, isatin acts as the electrophilic partner, while malonic acid provides the necessary carbon framework for ring expansion and functionalization. The sodium acetate catalyst activates the methylene group of the malonic acid, promoting nucleophilic attack on the carbonyl carbon of the isatin ring. This interaction initiates a cascade of dehydration and cyclization events that ultimately construct the quinoline backbone with high regioselectivity. The reaction environment provided by glacial acetic acid ensures that all reactants remain in solution during the critical reflux phase, facilitating optimal molecular collisions and reaction kinetics without the need for exotic or expensive transition metal catalysts.
Impurity control is intrinsically managed through the specific workup protocol designed to leverage the solubility differences of the target molecule versus byproducts. Following the reflux and solvent recovery, the addition of deionized water induces precise crystallization of the 2-hydroxy-4-carboxyquinoline, leaving soluble impurities in the mother liquor. The subsequent washing steps with cold glacial acetic acid and cold methanol are critical for removing residual starting materials and oligomeric side products that might co-precipitate. This rigorous purification sequence, integrated directly into the isolation process, ensures that the final solid product achieves a liquid phase purity of up to 99.6 percent without the need for energy-intensive recrystallization or chromatographic separation. Such high purity is paramount for downstream applications in pharmaceutical synthesis, where trace impurities can compromise the safety profile of the final active pharmaceutical ingredient.
How to Synthesize 2-Hydroxy-4-Carboxyquinoline Efficiently
The operational procedure for this synthesis is designed for seamless integration into standard multipurpose chemical reactors found in most fine chemical facilities. The process begins with the charging of glacial acetic acid followed by the sequential addition of isatin, malonic acid, and the sodium acetate catalyst under continuous agitation to ensure homogeneity. Once the mixture is heated to reflux and maintained for the specified duration, typically between 3 to 9 hours, the reaction is deemed complete, and the solvent is stripped under vacuum for recovery.
- Charge glacial acetic acid into a reactor, then add isatin, malonic acid, and sodium acetate under stirring to form a homogeneous mixture.
- Heat the reaction mixture to reflux temperature and maintain for 3 to 9 hours to ensure complete cyclization and conversion.
- Remove glacial acetic acid under reduced pressure for recovery, then add deionized water to the residue to induce crystallization, followed by filtration and washing.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible strategic benefits that extend beyond mere technical feasibility. The elimination of hazardous reagents like sodium hydride and the reduction of wastewater generation significantly lower the regulatory burden and insurance costs associated with chemical manufacturing. By simplifying the process flow to a one-pot reaction, manufacturers can achieve higher throughput rates with existing infrastructure, effectively increasing capacity without capital expenditure on new specialized equipment. The ability to recover and reuse the primary solvent, glacial acetic acid, creates a closed-loop system that insulates production costs from volatile raw material price fluctuations, ensuring more stable pricing for long-term contracts.
- Cost Reduction in Manufacturing: The economic model of this synthesis is heavily favored by the drastic reduction in raw material waste and solvent consumption. Since the glacial acetic acid is evaporated and recycled rather than discarded, the variable cost per kilogram of product is significantly lowered compared to processes requiring fresh solvent for every batch. Additionally, the avoidance of expensive purification technologies like column chromatography, relying instead on simple crystallization and filtration, reduces both utility costs and processing time. The use of commodity chemicals like isatin and malonic acid, which are widely available from multiple global suppliers, further mitigates supply risk and prevents vendor lock-in, fostering a competitive purchasing environment.
- Enhanced Supply Chain Reliability: The robustness of this one-pot method enhances supply continuity by minimizing the number of potential failure points in the production line. Traditional multi-step syntheses are prone to yield losses at each isolation stage, whereas this consolidated approach maximizes overall yield, ensuring that more finished product is generated from the same amount of input material. The mild reaction conditions reduce the likelihood of equipment corrosion or thermal runaway incidents, leading to fewer unplanned shutdowns and maintenance delays. Consequently, suppliers utilizing this technology can offer more reliable delivery schedules and shorter lead times, which is critical for pharmaceutical clients managing tight inventory windows and just-in-time manufacturing protocols.
- Scalability and Environmental Compliance: From an environmental compliance perspective, this process offers a clear advantage by substantially reducing the volume of organic waste and aqueous effluent requiring treatment. The absence of heavy metals and toxic reagents simplifies the waste disposal process, aligning with increasingly stringent global environmental regulations. The scalability of the reflux and crystallization steps is well-understood in chemical engineering, allowing for straightforward scale-up from pilot plants to multi-ton commercial production without encountering the mixing or heat transfer issues often seen in microwave-assisted or heterogeneous reactions. This ease of scale-up ensures that supply can be rapidly ramped up to meet surging market demand for cinchocaine intermediates without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on yield expectations, safety profiles, and operational parameters. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their broader supply networks.
Q: What are the primary safety advantages of this new synthesis route compared to traditional methods?
A: Unlike prior art methods that utilize hazardous sodium hydride or generate excessive wastewater through alkaline hydrolysis, this patented process employs mild sodium acetate catalysis in glacial acetic acid, significantly reducing explosion risks and environmental waste discharge.
Q: How does the solvent recovery process impact production costs?
A: The process allows for the evaporation and reuse of glacial acetic acid under reduced pressure, drastically lowering raw material consumption and waste treatment expenses associated with solvent disposal.
Q: What purity levels can be achieved with this one-pot method?
A: Experimental data from the patent indicates that the final product achieves a liquid phase purity of up to 99.6% with yields exceeding 96%, meeting stringent requirements for high-quality pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxy-4-Carboxyquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your pharmaceutical development pipelines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory concept to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify every batch against the highest industry standards. Our facility is equipped to handle the specific solvent recovery and crystallization requirements of this patented process, guaranteeing a consistent supply of 2-hydroxy-4-carboxyquinoline that supports your manufacturing goals.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages inherent in this technology. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on verified performance metrics and our proven capability to deliver excellence in fine chemical manufacturing.
