Advanced Synthesis of Spiro-Dihydropyrimidine Derivatives for Antitumor Drug Development
The pharmaceutical industry is constantly seeking novel heterocyclic scaffolds that offer enhanced biological profiles for oncology applications. Patent CN110041349B discloses a significant advancement in this field through the development of a new class of 5-aryl-4'-hydroxy-7-methyl-3-oxo-2'-aryl-3,4',5,5'-tetrahydro-2'H-spiro[thiazolo[3,2-a]pyrimidine-2,3'-thiophene]-6-carboxylic acid ethyl ester derivatives. These compounds represent a sophisticated fusion of dihydropyrimidinone and thiophene pharmacophores, designed to overcome the limitations of traditional calcium channel blockers and kinase inhibitors. The core innovation lies in the spiro-configuration, which restricts molecular conformation and potentially improves binding affinity to biological targets. As demonstrated in the structural formula below, the versatility of substituents R1 and R2 allows for extensive SAR optimization.
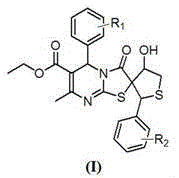
From a commercial perspective, the availability of such high-purity intermediates is critical for drug discovery pipelines. The patent highlights that these derivatives exhibit potent inhibitory activity against human tumor cell lines, including gastric cancer (MGC-803), prostate cancer (PC-3), and esophageal squamous carcinoma (EC-109). For R&D directors and procurement managers, understanding the synthetic accessibility of these complex molecules is paramount. The disclosed method provides a robust pathway that balances structural complexity with operational simplicity, utilizing readily available starting materials such as aromatic aldehydes, ethyl acetoacetate, and thiourea. This balance is essential for ensuring a reliable supply chain for preclinical and clinical material needs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of dihydropyrimidinones (DHPMs), often via the classic Biginelli reaction, frequently suffers from issues related to low yields, long reaction times, and the requirement for harsh acidic conditions or expensive Lewis acid catalysts. Furthermore, introducing a spiro-thiophene moiety onto this scaffold typically involves multi-step sequences with poor atom economy. Conventional methods for constructing sulfur-containing heterocycles often rely on volatile sulfur reagents or toxic solvents, creating significant environmental and safety burdens for manufacturing facilities. Additionally, the lack of stereocontrol in standard Biginelli protocols can lead to difficult-to-separate racemic mixtures, complicating downstream purification and increasing the cost of goods sold. These factors collectively hinder the rapid iteration required in modern medicinal chemistry programs.
The Novel Approach
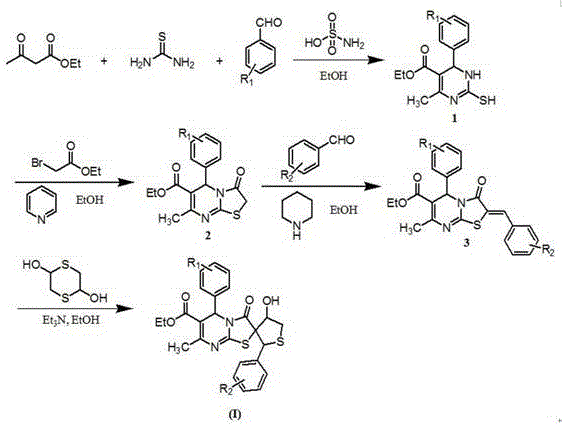
The methodology described in CN110041349B offers a streamlined solution by integrating the formation of the pyrimidine and thiophene rings into a cohesive sequence. The process begins with a catalyzed Biginelli condensation using sulfamic acid, a mild and inexpensive catalyst, which mitigates the corrosion and waste issues associated with strong mineral acids. Subsequent cyclization with ethyl bromoacetate efficiently constructs the thiazolo-pyrimidine core. The final spiro-cyclization step is particularly elegant, proceeding at ambient temperature (25°C) using 2,5-dihydroxy-1,4-dithiane. This mild condition preserves sensitive functional groups and reduces energy consumption. The overall route, depicted in the reaction scheme below, demonstrates high modularity, allowing for the rapid generation of diverse analogues by simply varying the aldehyde inputs.
Mechanistic Insights into Spiro-Cyclization and Ring Formation
The synthetic mechanism involves a cascade of well-defined organic transformations that ensure high regioselectivity. Initially, the condensation of the aldehyde, beta-keto ester, and thiourea forms the 2-mercapto-dihydropyrimidine intermediate. This step is crucial as it establishes the nitrogen-rich core essential for biological activity. The subsequent alkylation with ethyl bromoacetate triggers an intramolecular nucleophilic attack by the sulfur atom, closing the thiazole ring fused to the pyrimidine system. This creates the rigid tricyclic framework found in intermediate 2. The final transformation involves a Knoevenagel condensation with a second aromatic aldehyde to form a benzylidene intermediate, which then undergoes a Michael-type addition with the dithiane derivative. This final cyclization creates the unique spiro-junction at the 2-position of the thiazole ring, locking the molecule into a specific 3D geometry that is distinct from planar analogues.
Impurity control is inherently managed through the choice of reagents and reaction conditions. The use of column chromatography for purification, as specified in the patent examples, ensures the removal of unreacted starting materials and side products such as self-condensation polymers of the aldehydes. The mild basic conditions employed in the final step (using triethylamine) prevent the hydrolysis of the ethyl ester group, a common side reaction in alkaline environments. This stability is vital for maintaining the integrity of the carboxylic acid ethyl ester functionality, which often serves as a handle for further derivatization or prodrug design. The mechanistic clarity of this route provides confidence in its reproducibility across different batch scales.
How to Synthesize Spiro-Dihydropyrimidine Derivatives Efficiently
The synthesis protocol outlined in the patent is designed for operational ease, making it highly suitable for both laboratory scale-up and pilot plant production. The process relies on standard unit operations such as reflux, filtration, and chromatographic separation, which are universally available in chemical manufacturing facilities. The use of ethanol as the primary solvent throughout the sequence simplifies solvent recovery and reduces the environmental footprint of the process. Detailed standard operating procedures for each reaction stage, including specific molar ratios and temperature controls, are critical for achieving the reported yields of 46% to 75%. For a comprehensive guide on executing this synthesis with optimal parameters, please refer to the standardized protocol below.
- Perform Biginelli condensation of aromatic aldehyde, ethyl acetoacetate, and thiourea using sulfamic acid catalyst in ethanol under reflux to obtain the dihydropyrimidine intermediate.
- React the intermediate with ethyl bromoacetate and base to form the thiazolo-pyrimidine core, followed by Knoevenagel condensation with substituted benzaldehyde.
- Complete the spiro-cyclization by reacting the benzylidene intermediate with 2,5-dihydroxy-1,4-dithiane and triethylamine at 25°C to yield the final spiro derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic viability of a new intermediate is determined by raw material availability, process safety, and scalability. This synthetic route excels in all three areas by leveraging commodity chemicals. The starting materials, including various substituted benzaldehydes and ethyl acetoacetate, are produced globally in massive quantities, ensuring a stable supply base and protecting against price volatility. Furthermore, the elimination of precious metal catalysts removes the need for expensive scavenging steps to meet residual metal specifications, which is a significant cost driver in API manufacturing. The process safety is enhanced by avoiding high-pressure reactions or extremely low temperatures, reducing the capital expenditure required for specialized reactor equipment.
- Cost Reduction in Manufacturing: The synthetic strategy significantly lowers production costs by utilizing sulfamic acid and organic bases like piperidine and triethylamine instead of costly transition metal catalysts. This substitution not only reduces direct material costs but also simplifies the downstream purification process, as there is no need for complex heavy metal removal protocols. The high atom economy of the spiro-cyclization step minimizes waste generation, leading to substantial savings in waste disposal fees. Additionally, the ability to perform the final reaction at room temperature drastically cuts energy consumption compared to processes requiring cryogenic cooling or prolonged heating.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals ensures that production schedules are not disrupted by niche reagent shortages. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, providing a buffer against supply chain fluctuations. The modular nature of the synthesis allows manufacturers to quickly switch between different analogues by changing only the aldehyde feedstock, enabling a flexible response to changing R&D demands without retooling the entire production line.
- Scalability and Environmental Compliance: The use of ethanol as a green solvent aligns with increasingly stringent environmental regulations regarding VOC emissions and solvent residues. The process generates minimal hazardous waste, primarily consisting of aqueous washes and spent organic solvents that are easily recyclable. The straightforward workup procedures, involving simple filtration and crystallization in many steps, facilitate easy scale-up from grams to kilograms without encountering significant mixing or heat transfer issues. This scalability ensures that the transition from preclinical to clinical supply can be achieved rapidly and reliably.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these spiro-dihydropyrimidine derivatives. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation. Understanding these details is crucial for evaluating the feasibility of incorporating these intermediates into your drug development pipeline. We encourage technical teams to review these points to assess alignment with their specific project requirements.
Q: What represents the key structural advantage of these spiro-dihydropyrimidine derivatives?
A: The integration of a spiro-thiophene ring onto the dihydropyrimidine scaffold significantly enhances structural rigidity and lipophilicity, which correlates with improved inhibitory activity against tumor cell lines like MGC-803 and PC-3 compared to non-spiro analogs.
Q: Is the synthesis process suitable for large-scale manufacturing?
A: Yes, the process utilizes common solvents like ethanol and avoids expensive transition metal catalysts. The reactions proceed under mild conditions, with the final step occurring at room temperature (25°C), facilitating easier heat management and scale-up.
Q: How does the substituent variation affect biological activity?
A: Structure-activity relationship studies indicate that electron-donating or withdrawing groups at the ortho, meta, or para positions of the aryl rings modulate potency. Specifically, methyl and chloro substitutions have shown superior IC50 values in gastric and prostate cancer models.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro-Dihydropyrimidine Derivative Supplier
As a leading CDMO partner, NINGBO INNO PHARMCHEM possesses the technical expertise to translate complex laboratory routes like CN110041349B into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and quality. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the structural integrity and impurity profile of every batch. Our commitment to quality assurance guarantees that the spiro-dihydropyrimidine intermediates supplied meet the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to optimize this synthesis for your specific needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain that prioritizes efficiency, compliance, and innovation, empowering your R&D efforts to bring novel antitumor therapies to market faster.
