Scalable Synthesis of 2,4-Dichloro-3,5-Dinitrobenzotrifluoride for Global Supply Chains
The chemical industry is constantly evolving, driven by the need for more efficient and environmentally sustainable synthesis routes for critical intermediates. Patent CN111533660A introduces a groundbreaking preparation method for 2, 4-dichloro-3, 5-dinitrobenzotrifluoride, a vital building block in the synthesis of advanced pesticides, pharmaceuticals, and dyes. This technology addresses long-standing challenges in nitration chemistry by implementing a novel two-step reaction sequence that not only enhances product purity to over 98.5% but also integrates a circular economy model through waste acid recycling. For R&D directors and procurement specialists, this patent represents a significant leap forward in process chemistry, offering a pathway to reduce raw material consumption and minimize environmental impact without compromising on yield or quality standards. The ability to recycle semi-waste acid directly back into the initial reaction stage is a key differentiator that sets this method apart from traditional single-step or non-recycling processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2, 4-dichloro-3, 5-dinitrobenzotrifluoride has been plagued by inefficient reaction conditions and significant environmental burdens. Prior art methods, such as those described in earlier literature, often required prolonged reaction times extending up to 96 hours at elevated temperatures around 80°C, resulting in yields that hovered between 60% and 79%. These conventional approaches typically involved a one-pot nitration strategy that generated large volumes of difficult-to-treat waste acid, necessitating complex post-treatment procedures like neutralization which often led to product loss. Furthermore, the purity of the crude product in these older methods was frequently insufficient, often ranging between 85% and 90%, requiring extensive and costly purification steps to meet the stringent specifications demanded by the agrochemical and pharmaceutical industries. The energy consumption associated with maintaining high temperatures for such extended periods also contributed to higher operational costs and a larger carbon footprint.
The Novel Approach
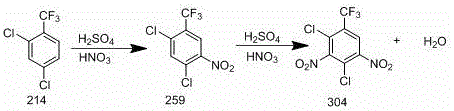
In stark contrast, the method disclosed in patent CN111533660A utilizes a strategic two-step nitration process that dramatically improves both efficiency and sustainability. By separating the nitration into two distinct stages, the process allows for precise control over reaction conditions, with the first step occurring at a mild 40-50°C and the second step optimized at 100-110°C. This staged approach ensures that the intermediate, 2, 4-dichloro-3-nitrobenzotrifluoride, is formed with high selectivity before undergoing the second nitration to achieve the desired 3, 5-dinitro substitution pattern. Crucially, the innovation lies in the treatment of the by-product acid; the semi-waste acid generated in the second step is not discarded but is instead directly recycled into the first-step reaction kettle. This closed-loop system not only reduces the discharge of hazardous waste but also leverages the residual acidity of the waste stream to catalyze the initial reaction, thereby lowering the consumption of fresh fuming sulfuric acid and driving down overall production costs significantly.
Mechanistic Insights into Two-Step Nitration and Acid Recycling
The core of this technological advancement lies in the precise manipulation of electrophilic aromatic substitution mechanisms under controlled acidic conditions. In the first step, 2, 4-dichlorobenzotrifluoride reacts with a mixture of fuming nitric acid and fuming sulfuric acid to introduce the first nitro group at the 3-position. The presence of the trifluoromethyl group and two chlorine atoms deactivates the benzene ring, requiring strong nitrating agents, yet the controlled temperature of 40-50°C prevents over-nitration and side reactions that could compromise the integrity of the trifluoromethyl moiety. The intermediate formed, 2, 4-dichloro-3-nitrobenzotrifluoride, is then isolated and dissolved in fuming sulfuric acid to ensure complete solvation before the second nitration. This solvation step is critical as it enhances the contact between the organic substrate and the nitrating species, facilitating the introduction of the second nitro group at the 5-position to yield the final 2, 4-dichloro-3, 5-dinitrobenzotrifluoride.
From an impurity control perspective, the mechanism benefits greatly from the avoidance of neutralization steps in the post-treatment phase. Traditional methods often require neutralizing the reaction mixture with base, which can lead to the formation of salts and emulsions that trap the product, reducing recovery rates. In this novel process, the crude product is separated via liquid-liquid phase separation while hot, followed by water washing and recrystallization using 1, 2-dichloroethane. This physical separation method preserves the product yield and ensures that the final purity exceeds 98.5% with a moisture content of less than 0.5%. The recycling of the semi-waste acid also plays a mechanistic role; by reintroducing acid containing residual nitronium ions and sulfuric acid into the first step, the reaction kinetics are maintained without the need for excessive fresh acid, creating a self-sustaining chemical environment that stabilizes the process over multiple batches.
How to Synthesize 2,4-Dichloro-3,5-Dinitrobenzotrifluoride Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry and temperature profiles outlined in the patent data to ensure reproducibility and safety. The process begins with the charging of 2, 4-dichlorobenzotrifluoride into a reactor along with fuming nitric and sulfuric acids, maintaining a molar ratio that favors the formation of the mono-nitro intermediate while minimizing by-products. Following the initial reaction and phase separation, the intermediate is subjected to the second nitration stage where temperature ramping from 100°C to 110°C is critical for driving the reaction to completion without degradation. The detailed standardized synthesis steps, including specific addition rates, stirring speeds, and crystallization parameters, are essential for achieving the reported yields of over 80% and purity levels suitable for high-end applications. For manufacturing teams looking to adopt this technology, adhering to these precise operational windows is key to unlocking the full commercial potential of this route.
- Conduct first-step nitration of 2,4-dichlorobenzotrifluoride with fuming nitric and sulfuric acid at 40-50°C to form the mono-nitro intermediate.
- Perform second-step nitration on the intermediate using mixed acid at 100-110°C, then separate the semi-waste acid for recycling.
- Recrystallize the crude product using dichloroethane and vacuum dry to obtain the final high-purity target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates into tangible strategic advantages regarding cost stability and supply continuity. The primary economic driver is the significant reduction in raw material costs achieved through the recycling of semi-waste acid. By reusing the acid stream from the second nitration step, the consumption of fresh fuming sulfuric acid is drastically lowered, which is a major cost component in nitration chemistry. Furthermore, the elimination of the neutralization step reduces the consumption of bases and the associated costs of wastewater treatment and disposal. This streamlined workflow not only lowers the variable cost per kilogram of the intermediate but also simplifies the supply chain by reducing the dependency on multiple auxiliary chemicals. The robustness of the process, characterized by its operation under normal pressure and relatively short reaction times, ensures high throughput and reliable delivery schedules for downstream customers.
- Cost Reduction in Manufacturing: The integration of waste acid recycling directly impacts the bottom line by minimizing the purchase volume of expensive fuming acids and reducing waste disposal fees. The process eliminates the need for neutralization agents, which further cuts down on chemical procurement costs and simplifies the inventory management of auxiliary materials. Additionally, the high yield and purity reduce the need for re-processing or discarding off-spec batches, ensuring that raw material input is converted into saleable product with maximum efficiency. These cumulative effects result in substantial cost savings that can be passed on to customers or reinvested into capacity expansion.
- Enhanced Supply Chain Reliability: The simplified operational procedure, which avoids complex post-treatment neutralization and extensive purification, reduces the risk of production bottlenecks and equipment downtime. The ability to operate under normal pressure enhances safety profiles, reducing the likelihood of regulatory interruptions or safety-related shutdowns. Moreover, the use of common and readily available raw materials like 2, 4-dichlorobenzotrifluoride ensures that supply is not constrained by exotic or scarce reagents. This reliability is crucial for maintaining consistent lead times and meeting the just-in-time delivery requirements of global agrochemical and pharmaceutical manufacturers.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction conditions that are easily transferable from laboratory to industrial reactors. The reduction in waste acid discharge aligns with increasingly stringent environmental regulations, mitigating the risk of compliance penalties and enhancing the corporate sustainability profile. By adopting a green chemistry approach that emphasizes circular economy principles, manufacturers can secure long-term operational licenses and appeal to environmentally conscious partners. The ease of scaling this complex nitro compound synthesis ensures that supply can be ramped up quickly to meet surging market demand without compromising on quality or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2, 4-dichloro-3, 5-dinitrobenzotrifluoride using this advanced nitration technology. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and benefits of this synthesis route for potential partners. Understanding these details is essential for evaluating the fit of this intermediate within your specific supply chain and product development pipelines. The answers provided reflect the technical realities of the process, ensuring transparency and trust in the proposed manufacturing capabilities.
Q: How does the new nitration process improve purity compared to conventional methods?
A: The novel two-step method avoids neutralization steps that typically cause product loss and achieves a purity exceeding 98.5% through optimized temperature control and recrystallization.
Q: What are the environmental benefits of the waste acid recycling technique?
A: By directly recycling semi-waste acid from the second nitration step into the first step, the process significantly reduces waste acid discharge and aligns with green chemistry principles.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process operates under normal pressure with simple operations and short reaction times, making it highly scalable for industrial manufacturing of agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-Dichloro-3,5-Dinitrobenzotrifluoride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final products, whether in crop protection or pharmaceutical formulations. Our technical team has extensively analyzed the pathway described in patent CN111533660A and possesses the expertise to scale this diverse pathway from 100 kgs to 100 MT/annual commercial production with precision. We are committed to delivering stringent purity specifications and maintaining rigorous QC labs to ensure that every batch of 2, 4-dichloro-3, 5-dinitrobenzotrifluoride meets the highest industry standards. Our CDMO capabilities allow us to adapt this efficient nitration process to your specific volume requirements, ensuring a seamless transition from development to full-scale manufacturing.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through this superior synthesis route. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific consumption needs. We encourage you to reach out for specific COA data and route feasibility assessments to verify how this technology can enhance your production efficiency. Partnering with us means gaining access to a reliable source of complex nitro compounds backed by a commitment to innovation, quality, and sustainable chemical manufacturing practices.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
