Advanced Toluene-Based Synthesis of 3,5-Di-Tert-Butyl-4-Hydroxybenzoic Acid for Commercial Scale-Up
The global demand for high-performance antioxidants and UV absorbers continues to drive innovation in the synthesis of key pharmaceutical and polymer intermediates. A significant breakthrough in this domain is detailed in patent CN109096099B, which discloses a novel production method for 3,5-di-tert-butyl-4-hydroxybenzoic acid. This compound serves as a critical precursor for synthesizing various esters used in plastics, rubber, and photo-curing coatings, offering resistance against inflammation, viruses, and tumors. The patented process represents a paradigm shift from traditional gas-solid heterogeneous reactions to a safer, more efficient liquid-phase Kolbe-Schmitt carboxylation. By optimizing solvent selection and reaction conditions, this technology addresses long-standing issues regarding yield, safety, and environmental impact, positioning it as a vital asset for any reliable pharmaceutical intermediate supplier seeking to enhance their portfolio with cost-effective manufacturing solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 3,5-di-tert-butyl-4-hydroxybenzoic acid has been plagued by significant technical and safety hurdles inherent to gas-solid processes. Traditional methods involve reacting dried powder of alkali metal salts of 2,6-di-tert-butylphenol with carbon dioxide, a procedure fraught with danger due to the propensity of these dry powders to undergo spontaneous combustion. Furthermore, gas-solid reactions necessitate specialized transmission equipment and unique stirring paddles to ensure adequate contact, driving up capital expenditure. Thermodynamically, these systems suffer from poor heat transfer, leading to local overheating, product coking, and ultimately, low yields. Alternative solvent-based approaches have also faced challenges; for instance, using octanol requires vacuum conditions for dehydration, increasing energy costs and solvent loss, while polar solvents like DMF complicate downstream processing and pose environmental risks due to nitrogenous waste.
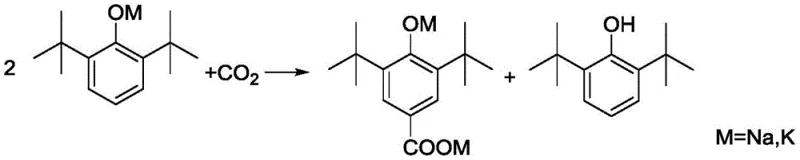
The Novel Approach
The innovative methodology described in the patent overcomes these deficiencies by employing toluene as a solvent and potassium hydroxide aqueous solution as the base. This liquid-phase approach allows for the in-situ formation of potassium 2,6-di-tert-butylphenolate under reflux, utilizing toluene's ability to azeotropically remove water without the need for vacuum equipment. The subsequent introduction of pressurized carbon dioxide facilitates a homogeneous Kolbe-Schmitt reaction that ensures uniform heating and prevents coking. Crucially, the non-polar nature of toluene enhances the solubility of carbon dioxide, significantly accelerating reaction kinetics compared to polar solvents. This strategic combination not only improves the single-pass conversion rate to over 90% but also simplifies the recovery of unreacted raw materials, creating a closed-loop system that drastically reduces waste and operational complexity.
Mechanistic Insights into Liquid-Phase Kolbe-Schmitt Carboxylation
The core of this technological advancement lies in the precise control of the carboxylation mechanism within a liquid medium. Unlike the chaotic interface of gas-solid reactions, the liquid phase allows for molecular-level interaction between the potassium phenolate and dissolved carbon dioxide. The reaction proceeds through the nucleophilic attack of the phenolate oxygen on the electrophilic carbon of CO2, followed by rearrangement to form the salicylate structure. The use of potassium ions is critical, as comparative data reveals that sodium ions fail to facilitate this transformation effectively under identical conditions. The reaction environment is maintained at pressures between 0.1-3.0 MPa and temperatures of 80-160°C, conditions that optimize the equilibrium towards the carboxylated product while minimizing thermal degradation. This controlled environment ensures that the regioselectivity favors the 4-position hydroxylation, yielding the desired 3,5-di-tert-butyl-4-hydroxybenzoic acid skeleton with high fidelity.
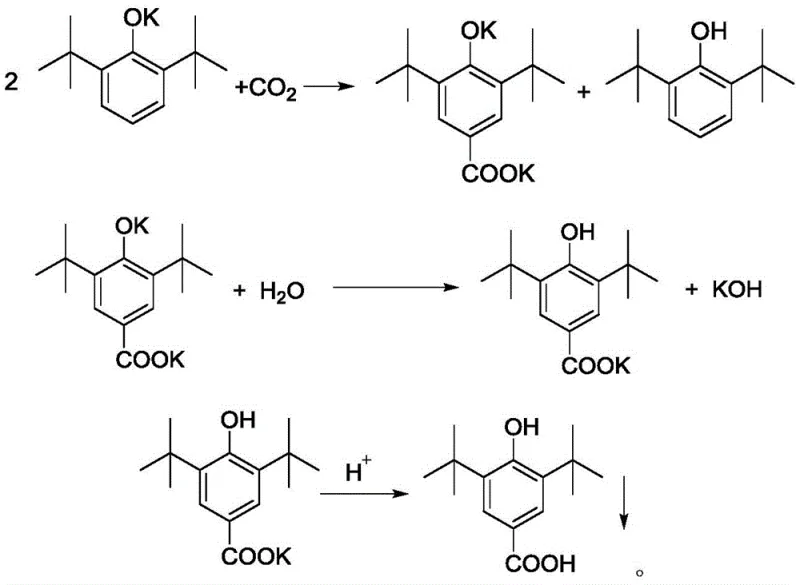
Impurity control is inherently managed through the phase separation capabilities of the toluene-water system. Upon completion of the carboxylation, the addition of distilled water dissolves the potassium salt of the product, separating it from the organic phase containing unreacted phenol and toluene. This physical separation is far superior to chemical quenching methods that might generate emulsions or difficult-to-remove byproducts. The aqueous phase, now containing the pure potassium salt, is subsequently acidified to precipitate the final acid product. This step-wise isolation prevents the co-precipitation of organic impurities, ensuring that the crude product achieves a purity of 99.5% even before final recrystallization. The mechanism effectively decouples the reaction from the purification, allowing each stage to be optimized independently for maximum efficiency and quality.
How to Synthesize 3,5-Di-Tert-Butyl-4-Hydroxybenzoic Acid Efficiently
The synthesis protocol outlined in the patent provides a robust framework for industrial implementation, focusing on reproducibility and safety. The process begins with the preparation of the potassium phenolate suspension, followed by the pressurized carboxylation step, and concludes with a straightforward workup involving phase separation and acidification. This sequence eliminates the need for hazardous dry powder handling and complex vacuum systems, making it accessible for standard chemical manufacturing facilities. The detailed operational parameters, including specific molar ratios and temperature gradients, are designed to maximize throughput while maintaining strict quality control standards. For technical teams looking to implement this route, the following standardized steps provide a comprehensive guide to achieving high-purity results consistently.
- Prepare potassium phenolate by refluxing 2,6-di-tert-butylphenol with aqueous KOH in toluene, utilizing azeotropic distillation to remove water.
- Introduce dried carbon dioxide into the reactor at 0.1-3.0 MPa and maintain temperature between 80-160°C to effect carboxylation.
- Cool the mixture, separate oil and water phases, acidify the aqueous phase to pH 1, and filter to obtain the crude acid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this novel synthesis route offers profound advantages that directly impact the bottom line and supply chain resilience. By transitioning from a hazardous gas-solid process to a manageable liquid-phase reaction, manufacturers can significantly reduce the capital investment required for specialized explosion-proof equipment and complex solid-handling machinery. The elimination of vacuum dehydration steps further lowers energy consumption and equipment maintenance costs. Moreover, the use of toluene, a commodity solvent with established recovery infrastructure, replaces expensive or environmentally problematic alternatives like DMF or high-boiling alcohols. These factors collectively contribute to a substantial reduction in the cost of goods sold (COGS), making the final intermediate more competitive in the global market without compromising on quality or safety standards.
- Cost Reduction in Manufacturing: The process achieves cost efficiency primarily through the optimization of raw material utilization and energy consumption. By avoiding the use of expensive catalysts like 1,3-dimethyl-2-imidazolidinone and eliminating the need for vacuum pumps required by octanol-based methods, the operational expenditure is drastically lowered. Additionally, the high conversion rate means less raw material is wasted, and the ability to recycle toluene and unreacted phenol creates a circular economy within the reactor, further driving down material costs over time.
- Enhanced Supply Chain Reliability: Safety is a critical component of supply chain continuity, and this method mitigates the risk of production halts due to safety incidents. By removing the risk of spontaneous combustion associated with dry alkali metal salts, the facility operates with greater stability and lower insurance premiums. The use of common, readily available reagents like potassium hydroxide and toluene ensures that raw material sourcing is not a bottleneck, allowing for consistent production schedules and reliable delivery timelines to downstream customers.
- Scalability and Environmental Compliance: The liquid-phase nature of the reaction makes it inherently easier to scale from pilot plants to multi-ton commercial production without the heat transfer limitations of gas-solid reactors. Furthermore, the environmental footprint is minimized by avoiding nitrogenous solvents like DMF, which require costly waste treatment. The efficient recycling of toluene and the aqueous workup system align with modern green chemistry principles, facilitating easier regulatory approval and reducing the burden of environmental compliance management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on process parameters and material choices. Understanding these nuances is essential for engineering teams and procurement specialists evaluating the feasibility of adopting this technology for large-scale manufacturing operations.
Q: Why is toluene preferred over DMF or octanol for this synthesis?
A: Toluene is preferred because it forms an azeotrope with water allowing easy dehydration without vacuum equipment, unlike octanol. Furthermore, toluene is less polar than DMF, which significantly increases the solubility of carbon dioxide and reaction conversion rates, while being easier to recover and more environmentally friendly.
Q: What are the safety advantages of this liquid-phase method?
A: This method eliminates the need to handle dry 2,6-di-tert-butylphenol alkali metal salt powders, which are prone to spontaneous combustion. By conducting the reaction in a liquid suspension, heat transfer is uniform, preventing local overheating and coking, thereby drastically reducing operational hazards.
Q: Can sodium hydroxide be used instead of potassium hydroxide?
A: No, comparative experiments indicate that sodium hydroxide hardly reacts with 2,6-di-tert-butylphenol under these conditions. Potassium hydroxide is essential for the efficient formation of the phenolate salt and subsequent carboxylation in this specific toluene-based system.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Di-Tert-Butyl-4-Hydroxybenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented synthesis route in delivering high-value intermediates to the global market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. Our state-of-the-art facilities are equipped to handle the specific pressure and temperature requirements of this liquid-phase Kolbe-Schmitt reaction, adhering to stringent purity specifications and operating within rigorous QC labs to guarantee batch-to-batch consistency. We are committed to leveraging such advanced technologies to provide our partners with superior products that meet the evolving demands of the pharmaceutical and polymer industries.
We invite forward-thinking organizations to collaborate with us to unlock the full commercial potential of this efficient manufacturing process. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of technical excellence and economic viability.
