Advanced Manufacturing of 3,3,3-Trifluoropropanol for Global Pharma and Agrochemical Supply Chains
The global demand for fluorinated building blocks continues to surge, driven by the critical role these moieties play in enhancing the metabolic stability and bioavailability of modern pharmaceuticals and agrochemicals. Within this landscape, 3,3,3-trifluoropropanol stands out as a versatile intermediate, yet its efficient production has historically been hampered by complex purification challenges and low yields. A pivotal breakthrough in this domain is documented in patent CN102766023A, which discloses a robust, two-step synthetic methodology that fundamentally alters the economic and technical feasibility of producing this valuable alcohol. By shifting away from traditional hydroboration pathways, this innovation offers a streamlined route that leverages readily available starting materials like 2-bromo-3,3,3-trifluoropropene and methanol. For R&D directors and procurement strategists alike, understanding this shift is crucial, as it represents a move towards more sustainable and cost-effective manufacturing paradigms in the fluorochemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,3,3-trifluoropropanol relied heavily on hydroboration-oxidation strategies, a pathway fraught with significant operational and economic inefficiencies. As illustrated in the reaction scheme below, the conventional method typically involves the reaction of 3,3,3-trifluoropropene with dibromoborane, followed by oxidative hydrolysis. This approach suffers from inherently low reaction yields, often stagnating around 41%, which drastically impacts the overall material throughput and cost efficiency of the production line. Furthermore, the reliance on aqueous workups in the oxidation step introduces a severe downstream processing bottleneck: the formation of azeotropes between the product alcohol and water. Separating these azeotropic mixtures requires energy-intensive distillation techniques or complex entrainers, complicating the purification process and increasing the environmental footprint of the manufacturing facility.
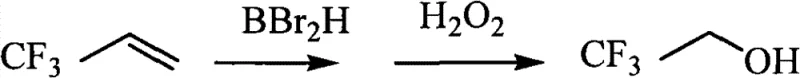
The Novel Approach
In stark contrast to the legacy hydroboration methods, the novel approach detailed in the patent introduces a clever etherification-hydrogenation sequence that circumvents these historical pain points. The process begins with a base-catalyzed nucleophilic substitution where 2-bromo-3,3,3-trifluoropropene reacts with methanol to form 3,3,3-trifluoropropenyl methyl ether. This intermediate is then subjected to catalytic hydrogenation using Raney Nickel under elevated pressure to yield the target alcohol. This strategic detour via an ether intermediate completely avoids the introduction of water during the reaction phase, thereby eliminating the troublesome azeotrope formation that plagues the older methods. Consequently, the final product can be isolated through simple atmospheric distillation with high purity, representing a paradigm shift in process simplicity and operational efficiency for fluorochemical manufacturers.
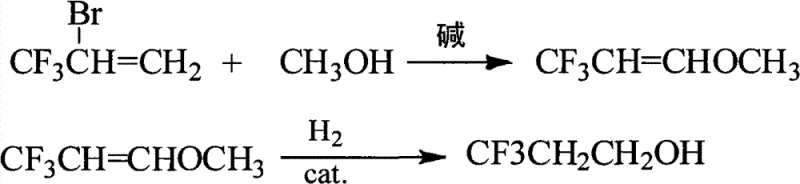
Mechanistic Insights into Base-Catalyzed Etherification and Raney Ni Hydrogenation
The success of this synthetic route lies in the precise control of reaction kinetics and the selection of robust catalytic systems. In the first step, the electron-withdrawing trifluoromethyl group activates the vinyl bromide towards nucleophilic attack by the methoxide ion generated in situ from methanol and alkali (such as sodium hydroxide or potassium hydroxide). The reaction conditions, typically maintained at 70°C for approximately 6 hours, ensure complete conversion while minimizing side reactions such as polymerization or elimination. The stoichiometry is carefully balanced, with a molar ratio of substrate to methanol to alkali optimized between 1:(3-8):(2.4-6.2), ensuring that the basic environment is sufficient to drive the substitution without degrading the sensitive fluorinated backbone. This step effectively installs the oxygen functionality while preserving the carbon skeleton integrity required for the final alcohol.
The subsequent hydrogenation step is equally critical, utilizing activated Raney Nickel as a heterogeneous catalyst to reduce the carbon-carbon double bond of the vinyl ether intermediate. Conducted in an autoclave under a hydrogen pressure of 5MPa and temperatures ranging from 60°C to 120°C, this step achieves high conversion rates with excellent selectivity. The presence of inorganic esters, such as tributyl phosphate or diethyl carbonate, acts as a solvent system that stabilizes the reaction mixture and facilitates heat transfer. Crucially, because the intermediate is an ether and not an alcohol, there is no risk of competing dehydration or rearrangement reactions that often complicate the hydrogenation of allylic alcohols. The result is a clean transformation where the only byproduct is methanol (from the cleavage of the methyl ether during hydrogenolysis or subsequent workup), which is easily separated, leading to a final product purity of 98% as confirmed by gas chromatography analysis.
How to Synthesize 3,3,3-Trifluoropropanol Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters regarding temperature, pressure, and reagent ratios to maximize the total yield, which can reach upwards of 60% over two steps. The process is designed for scalability, moving seamlessly from laboratory glassware to industrial autoclaves without losing efficiency. Operators must ensure the Raney Nickel catalyst is properly activated and handled under inert conditions to prevent pyrophoric hazards, while the initial etherification requires careful pH control during the aqueous wash to remove residual alkali and salts. For a comprehensive breakdown of the exact experimental procedures, reagent quantities, and safety protocols necessary for replication, please refer to the standardized guide below.
- React 2-bromo-3,3,3-trifluoropropene with methanol and alkali (NaOH/KOH) at 70°C for 6 hours to form 3,3,3-trifluoropropenyl methyl ether.
- Purify the intermediate ether through water washing to remove salts and unreacted base.
- Hydrogenate the ether using activated Raney Ni catalyst at 5MPa hydrogen pressure and 60-120°C to obtain the final alcohol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers profound strategic benefits that extend beyond mere technical feasibility. The elimination of expensive and hazardous boron reagents, such as dibromoborane, immediately lowers the raw material cost baseline, removing the volatility associated with sourcing specialized organometallic precursors. Furthermore, the simplification of the downstream processing—specifically the removal of azeotropic distillation requirements—translates directly into reduced utility consumption and shorter batch cycle times. This efficiency gain allows manufacturing facilities to increase throughput capacity without significant capital expenditure on new separation columns or drying equipment, effectively lowering the cost of goods sold (COGS) for this high-value intermediate.
- Cost Reduction in Manufacturing: The replacement of the hydroboration pathway with a methanol-based etherification significantly reduces reagent costs by utilizing commodity chemicals instead of specialized boron species. Additionally, the ability to purify the final product via simple distillation rather than complex azeotropic drying drastically cuts energy consumption and processing time. This streamlining of the workflow removes the need for expensive entrainers and reduces the waste disposal costs associated with boron-containing byproducts, leading to substantial overall cost optimization in fluorochemical manufacturing.
- Enhanced Supply Chain Reliability: By relying on widely available starting materials like 2-bromo-3,3,3-trifluoropropene and methanol, the supply chain becomes far more resilient to market fluctuations compared to routes dependent on niche reagents. The robustness of the Raney Nickel catalyst, which is a standard industrial commodity, further ensures that production schedules are not disrupted by catalyst shortages. This stability allows for more accurate long-term planning and inventory management, ensuring consistent availability of high-purity 3,3,3-trifluoropropanol for downstream pharmaceutical and agrochemical applications.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently in autoclave systems that mimic commercial production environments. The avoidance of aqueous workups in the critical purification stage minimizes the generation of fluorinated wastewater, simplifying effluent treatment and ensuring compliance with increasingly stringent environmental regulations. The solid Raney Nickel catalyst can be filtered and potentially regenerated or disposed of according to standard metal waste protocols, presenting a cleaner and more sustainable manufacturing profile compared to the homogeneous waste streams of traditional hydroboration methods.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 3,3,3-trifluoropropanol based on the patented methodology. These insights are derived directly from the experimental data and process descriptions found in the intellectual property documentation, providing clarity on yield expectations, purity profiles, and operational parameters. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their own synthesis pipelines or for procurement specialists assessing supplier capabilities.
Q: What are the primary advantages of this synthesis method over hydroboration?
A: This method eliminates the use of expensive dibromoborane reagents and avoids the formation of difficult-to-separate azeotropes with water, resulting in higher yields (up to 60%) and simpler purification via standard distillation.
Q: What catalyst is used in the final hydrogenation step?
A: The process utilizes activated Raney Nickel catalyst under high hydrogen pressure (5MPa), which is a cost-effective and widely available heterogeneous catalyst suitable for large-scale industrial application.
Q: Is the purity of the final 3,3,3-trifluoropropanol sufficient for pharmaceutical use?
A: Yes, the patent data indicates that simple atmospheric distillation yields the product with a purity of 98%, meeting the stringent requirements for high-value fine chemical and pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3,3-Trifluoropropanol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful deployment of advanced fluorinated intermediates requires more than just a patent; it demands expert process engineering and rigorous quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel synthesis are fully realized in a GMP-compliant environment. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art gas chromatography and NMR spectroscopy to guarantee that every batch of 3,3,3-trifluoropropanol meets the exacting standards required for API synthesis and high-performance material fabrication.
We invite global partners to collaborate with us to leverage this superior manufacturing technology for their specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this optimized route can improve your margin structures. We encourage potential clients to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of technical excellence and commercial reliability.
