Advanced Manufacturing Of High Purity Gadobutrol For Global MRI Contrast Agent Markets
Advanced Manufacturing Of High Purity Gadobutrol For Global MRI Contrast Agent Markets
The global demand for high-performance Magnetic Resonance Imaging (MRI) contrast agents continues to drive innovation in pharmaceutical intermediate manufacturing. Specifically, Gadobutrol, a non-ionic macrocyclic gadolinium chelate, represents the gold standard for safety and efficacy in clinical diagnostics. However, achieving the requisite ultra-high purity for regulatory approval has historically been a significant bottleneck. The present technical analysis focuses on the breakthrough methodology disclosed in patent CN107001294B, which outlines a novel, mild, and high-yield process for preparing Gadobutrol. This patent addresses critical pain points in the existing art by introducing a streamlined synthetic route that controls intermediate purity through simple crystallization rather than complex chromatographic separation. For R&D directors and procurement specialists, understanding this shift from labor-intensive purification to process-controlled purity is essential for securing a reliable gadobutrol supplier capable of meeting stringent ICH guidelines.
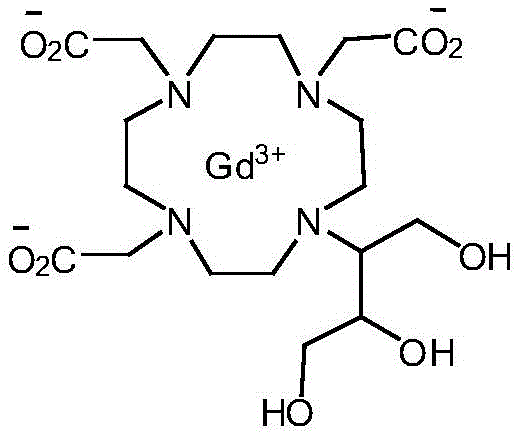
Gadobutrol, chemically defined as 10-(2,3-dihydroxy-1-(hydroxymethyl)propyl)-1,4,7,10-tetraazacyclododecane-1,4,7-triacetic acid gadolinium complex, is unique due to its 1.0 M concentration formulation capability. The structural integrity shown in the figure above relies on the precise stereochemistry and purity of the underlying butrol ligand. Conventional manufacturing struggles to maintain this integrity without significant loss of yield. The disclosed invention in CN107001294B offers a transformative approach, enabling the production of ultra-high-purity gadobutrol (>99.9%) with significantly improved overall yields. This technological leap not only enhances product quality but also fundamentally alters the cost structure of production, making it a pivotal development for cost reduction in MRI contrast agent manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of Gadobutrol was plagued by inefficiencies detailed in literature such as Inorg. Chem. 1997, 36, 6086-6093. Three primary routes were known, yet each possessed fatal flaws for industrial application. Scheme 1, while viable, demanded excessive quantities of ion-exchange resin for purification and necessitated specialized equipment like large-scale columns, driving up capital expenditure and unit costs. Scheme 2 suffered from inherently low yields and poor purity profiles, making it unsuitable for GMP production. Most critically, Scheme 3, though conceptually sound for laboratory synthesis, was explicitly deemed inappropriate for mass production due to unacceptable yield losses. These legacy methods created a supply chain vulnerability where producing high-purity material required sacrificing throughput or incurring prohibitive purification costs, limiting the availability of reliable high-purity gadobutrol intermediates.
The Novel Approach
The patented process introduces a robust three-step sequence that circumvents these historical obstacles by focusing on intermediate crystallization and mild reaction conditions. As illustrated in the comprehensive reaction scheme below, the new route begins with a selective carboxymethylation, proceeds through a controlled acid hydrolysis, and concludes with efficient gadolinium complexation. This approach eliminates the need for the massive resin volumes associated with Scheme 1 and avoids the low-yield pitfalls of Scheme 3. By optimizing solvent systems—such as using water and tetrahydrofuran (THF) mixtures—and implementing precise temperature controls, the process achieves intermediate purities exceeding 99.7% before the final step. This strategic focus on upstream purity control ensures that the final complexation step proceeds with minimal side reactions, resulting in a final API purity of >99.9%.
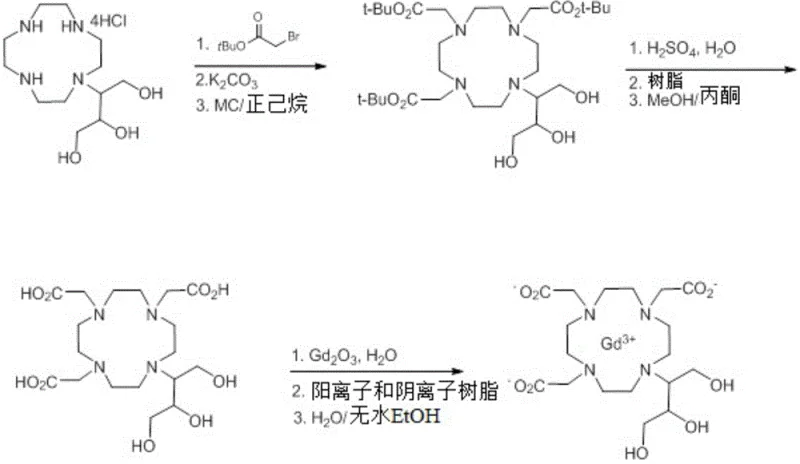
Mechanistic Insights into Carboxymethylation and Complexation
The core of this technological advancement lies in the mechanistic optimization of the carboxymethylation step (S1). In this stage, the starting macrocyclic amine salt reacts with a protected haloacetate, specifically tert-butyl bromoacetate, in the presence of a weak inorganic base like potassium carbonate. The choice of a mixed solvent system comprising water and C4-C11 ethers, preferably THF, is critical. This biphasic-like environment facilitates the dissolution of the inorganic salt while maintaining the organic reactants in solution, promoting efficient nucleophilic substitution without degrading the sensitive macrocyclic ring. The reaction is conducted at a温和 temperature range of 63°C to 68°C, which is sufficiently energetic to drive the alkylation to completion but mild enough to prevent the formation of over-alkylated byproducts or ring-opened impurities that typically plague harsher alkylation conditions.
Following the formation of the tri-tert-butyl ester intermediate, the process employs a sophisticated acid hydrolysis mechanism (S2) to reveal the free carboxylic acids required for metal binding. Unlike brute-force hydrolysis which can lead to racemization or degradation, this method utilizes dilute sulfuric acid at controlled temperatures (57°C to 63°C). Crucially, the patent describes a purification step using a minimal amount of resin (4 to 8 v/w) post-hydrolysis, which is significantly lower than the loads required in conventional Scheme 1. This efficiency is complemented by a crystallization step using methanol and acetone, which physically excludes impurities from the crystal lattice. The final complexation (S3) with gadolinium oxide then proceeds smoothly in aqueous media at 87°C to 93°C, leveraging the high purity of the ligand to form the thermodynamically stable non-ionic complex with exceptional kinetic inertness, a key safety feature for in vivo stability.
How to Synthesize Gadobutrol Efficiently
Implementing this synthesis route requires strict adherence to the specified reaction parameters to replicate the high yields and purity reported in the patent data. The process is designed to be scalable, moving seamlessly from laboratory validation to commercial production lines. Operators must pay particular attention to the crystallization solvents and temperature ramps, as these are the primary drivers of impurity rejection. The following guide outlines the standardized operational framework derived from the patent examples, ensuring consistent batch-to-batch quality.
- Perform carboxymethylation of the macrocyclic amine salt with tert-butyl bromoacetate in a water/THF solvent system using potassium carbonate at 63-68°C.
- Execute acid hydrolysis of the tri-tert-butyl ester intermediate using dilute sulfuric acid at 57-63°C, followed by resin purification and crystallization.
- React the purified ligand with gadolinium oxide in water at 87-93°C, followed by final purification and ethanol recrystallization to obtain the final complex.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel manufacturing process represents a significant opportunity for cost reduction in MRI contrast agent manufacturing. The elimination of specialized column equipment and the drastic reduction in resin consumption directly lower the variable costs per kilogram of produced API. Furthermore, the ability to achieve high purity through crystallization rather than extensive chromatography simplifies the waste stream profile, reducing environmental compliance costs and facilitating easier regulatory approval for new manufacturing sites. This process robustness ensures that supply continuity is maintained even during raw material fluctuations, as the yield improvements provide a buffer against input variability.
- Cost Reduction in Manufacturing: The new process fundamentally alters the cost equation by removing the dependency on large-scale resin columns and specialized tower equipment required by legacy methods. By utilizing standard reactor vessels and common solvents like THF, water, and acetone, the capital intensity of the production line is significantly decreased. Additionally, the higher overall yield means that less raw material is wasted per unit of finished product, leading to substantial cost savings in the bill of materials. The mild reaction conditions also reduce energy consumption associated with heating and cooling cycles, further enhancing the economic viability of large-scale production.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplicity and robustness of the synthetic route. Because the process does not rely on fragile low-yield steps or hard-to-source specialized purification media, it is less susceptible to bottlenecks. The use of commodity chemicals such as potassium carbonate, sulfuric acid, and gadolinium oxide ensures that raw material sourcing remains stable and competitive. This reliability allows suppliers to offer more consistent lead times for high-purity gadobutrol intermediates, mitigating the risk of stockouts that can disrupt the production of finished diagnostic agents.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the process is exceptionally favorable. The reduction in resin usage minimizes solid waste generation, while the reliance on crystallization for purification reduces the volume of organic solvent waste compared to chromatographic methods. The mild conditions (temperatures below 100°C and atmospheric pressure) simplify the engineering requirements for scale-up, allowing for safer operation in multi-ton reactors. This alignment with green chemistry principles not only lowers disposal costs but also future-proofs the manufacturing site against increasingly stringent environmental regulations regarding pharmaceutical effluent.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and background analysis provided in CN107001294B, offering clarity on how this technology resolves historical manufacturing challenges. Understanding these details is crucial for technical teams evaluating potential technology transfers or licensing opportunities.
Q: How does this new process improve impurity control compared to conventional methods?
A: Unlike conventional Scheme 1 which requires massive amounts of resin and special column equipment, this novel method utilizes a mild carboxymethylation step followed by controlled crystallization. This allows for intermediate purity control (>99.7%) before the final complexation, ensuring the final API meets strict ICH guidelines of <0.1% impurities without complex chromatography.
Q: What are the scalability advantages of this synthesis route?
A: The process avoids low-yield pathways (like Scheme 3) and eliminates the need for specialized tower equipment required by older methods. By using standard solvent systems like THF/Water and simple crystallization techniques (DCM/Hexane), the reaction is highly adaptable for commercial scale-up from pilot plants to multi-ton production facilities.
Q: Why is the purity of the intermediate critical in Gadobutrol manufacturing?
A: Gadobutrol is an injectable MRI contrast agent requiring ultra-high purity (>99.9%). The patent demonstrates that controlling the purity of the tri-acetic acid intermediate (Butrol) to >98% via optimized hydrolysis and resin treatment directly correlates to the ability to produce the final gadolinium complex with minimal toxic metal impurities and organic residuals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gadobutrol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the complexity of synthesizing macrocyclic gadolinium chelates requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent laboratory data to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs equipped to verify the <0.1% impurity levels required for injectable contrast agents. Our commitment to quality assurance means that every batch of Gadobutrol intermediate or API is traceable, fully characterized, and compliant with international pharmacopoeia standards.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced manufacturing technology for your supply chain. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to discuss specific COA data, route feasibility assessments, and how our optimized synthesis of Gadobutrol can enhance your product portfolio's competitiveness and reliability in the global market.
