Advanced Silicon-Centered Spiro Ligands for Commercial Asymmetric Catalysis and Scalable Production
The pharmaceutical and fine chemical industries are constantly seeking robust solutions to enhance the efficiency of asymmetric synthesis, a critical process for producing optically pure active pharmaceutical ingredients. Patent CN111217848A introduces a groundbreaking class of spiro-bis-dihydrobenzosilole diphenol compounds that address the limitations of existing chiral ligands. This technology leverages a unique silicon-centered spiro skeleton, offering superior steric properties and electronic characteristics compared to traditional carbon-centered frameworks. For R&D directors and procurement specialists, this represents a significant opportunity to optimize catalytic processes while securing a reliable chiral ligand supplier for complex synthetic pathways. The method described ensures high yields and exceptional enantioselectivity, utilizing cheap and readily available initial raw materials that simplify the supply chain logistics.
Furthermore, the operational simplicity of this synthesis route, characterized by mild reaction conditions and convenient purification protocols, directly translates to enhanced process safety and reduced operational expenditures. The ability to generate these compounds with high optical purity through metal-catalyzed asymmetric hydrosilylation establishes a new benchmark for ligand design. By integrating these advanced materials into your production workflow, organizations can achieve substantial cost savings in pharmaceutical intermediate manufacturing without compromising on the stringent quality standards required for regulatory compliance. This report analyzes the technical merits and commercial viability of adopting this novel silicon-based chiral scaffold.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chiral ligands, such as the widely used BINOL and SPINOL skeletons, rely heavily on carbon-centered axial chirality which, while effective, often presents inherent structural constraints regarding steric bulk and bond flexibility. The carbon-carbon bond lengths in these conventional frameworks are relatively short, which can sometimes limit the spatial arrangement necessary for optimal substrate binding in highly congested catalytic cycles. Additionally, the synthesis of these carbon-based spiro compounds often involves harsh reaction conditions, multiple protection-deprotection steps, and expensive starting materials that drive up the overall cost of goods. For procurement managers, these factors contribute to volatile pricing and extended lead times for high-purity fine chemicals, creating bottlenecks in the supply of critical intermediates needed for drug development.
Moreover, the purification of traditional ligands can be notoriously difficult, often requiring extensive chromatography which is not ideal for large-scale commercial operations. The rigidity of the carbon backbone may also limit the tunability of the ligand environment, restricting its applicability across a diverse range of asymmetric transformations. When scaling up these conventional methods, manufacturers frequently encounter issues with reproducibility and batch-to-batch consistency, which poses significant risks for supply chain heads responsible for maintaining continuous production lines. These structural and process inefficiencies highlight the urgent need for alternative scaffolds that offer greater flexibility and ease of manufacture.
The Novel Approach
The innovative approach detailed in the patent data utilizes a silicon-centered spiro skeleton that fundamentally alters the geometric and electronic landscape of the chiral ligand. Silicon possesses a larger atomic radius and forms longer bonds with carbon compared to carbon-carbon bonds, intrinsically creating a more open and flexible chiral pocket that can accommodate bulky substrates more effectively. This structural advantage allows for excellent chiral induction capability in various reactions, including palladium-catalyzed carbon-hydrogen bond ethynylation, where traditional ligands might struggle. The synthesis pathway is designed for efficiency, employing metal-catalyzed asymmetric hydrosilylation to directly construct the axial chiral spiro skeleton with high stereospecificity.
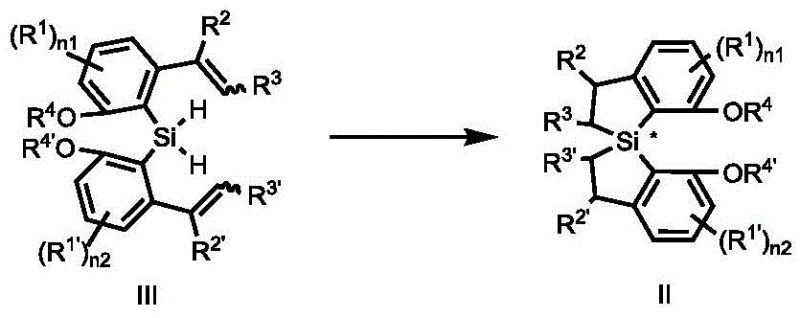
From a manufacturing perspective, this novel approach drastically simplifies the production workflow by using cheap and readily available initial raw materials such as substituted acetophenones or benzaldehydes. The reaction conditions are notably mild, often proceeding at room temperature or with moderate heating, which reduces energy consumption and equipment stress. Purification is streamlined through simple filtration and crystallization steps, eliminating the need for resource-intensive chromatographic separations. This shift towards a more pragmatic synthesis route ensures that the commercial scale-up of complex chiral catalysts becomes economically feasible, providing a stable foundation for long-term supply agreements and cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Rh-Catalyzed Asymmetric Hydrosilylation
The core of this technology lies in the sophisticated mechanism of rhodium-catalyzed asymmetric hydrosilylation, which constructs the chiral silicon center with remarkable precision. The process begins with the activation of a bis(allyl)silane precursor by a monovalent rhodium catalyst complexed with a chiral phosphine ligand. This catalytic system facilitates an intramolecular cyclization where the silicon-hydrogen bonds add across the carbon-carbon double bonds in a highly stereocontrolled manner. The chiral information from the phosphine ligand is effectively transferred to the newly formed silicon stereocenter, establishing the axial chirality of the spiro skeleton. This mechanistic pathway is crucial for R&D teams as it ensures that the resulting ligand possesses the rigorous optical purity necessary for downstream asymmetric catalytic applications.
Following the cyclization, the resulting methoxy-protected spiro-silane intermediate undergoes a demethylation process to reveal the active diphenol functionality. This step typically involves the use of trifluoropentafluoroboric acid and a silane reagent, followed by treatment with a base such as potassium acetate in methanol. The mechanism here ensures the clean removal of protecting groups without racemization of the sensitive chiral center. The final product exhibits excellent stability and solubility profiles, making it suitable for use in various solvent systems ranging from aromatic hydrocarbons to ethers. Understanding these mechanistic details allows technical teams to troubleshoot potential issues and optimize reaction parameters for maximum efficiency and yield.
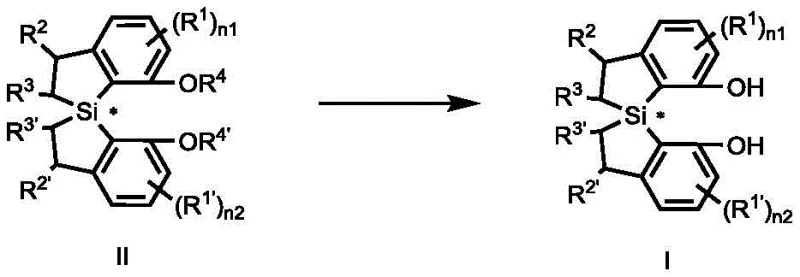
Impurity control is inherently built into this synthetic design, as the high stereoselectivity of the rhodium-catalyzed step minimizes the formation of diastereomers and enantiomeric impurities. The crystallization steps further enhance purity by selectively precipitating the desired isomer while leaving impurities in the mother liquor. For quality assurance personnel, this means that the final ligand meets stringent purity specifications with minimal need for additional refining. The robust nature of the silicon-carbon bonds in the spiro framework also contributes to the thermal and chemical stability of the ligand, ensuring it remains effective even under demanding reaction conditions. This level of mechanistic control is essential for maintaining consistent performance in commercial catalytic processes.
How to Synthesize Spiro-bis-dihydrobenzosilole Diphenol Efficiently
The synthesis of these high-value chiral ligands follows a logical sequence of organic transformations designed for scalability and safety. The process initiates with the preparation of bis(allyl)silane precursors through silicification reactions, followed by the key enantioselective cyclization step that establishes the chiral architecture. Final deprotection yields the active diphenol compound ready for complexation with metal salts. The detailed standardized synthesis steps see the guide below, which outlines the specific reagents, stoichiometry, and conditions required to replicate the high yields and enantiomeric excess values reported in the patent data.
- Prepare bis(allyl)silane precursors via silicification of bromo-alkene intermediates using tetrachlorosilane and reducing agents.
- Execute enantioselective cyclization using a monovalent rhodium catalyst and chiral phosphine ligands to form the spiro-silicon skeleton.
- Perform demethylation using trifluoropentafluoroboric acid and silane reagents followed by base treatment to yield the final diphenol product.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this silicon-centered spiro ligand technology offers profound benefits for procurement and supply chain management, primarily driven by the accessibility of raw materials and the simplicity of the manufacturing process. The reliance on cheap and readily available starting materials such as substituted acetophenones mitigates the risk of supply disruptions associated with exotic or proprietary reagents. This foundational stability allows for more accurate forecasting and inventory planning, ensuring that production schedules are met without unexpected delays. Furthermore, the mild reaction conditions reduce the need for specialized high-pressure or cryogenic equipment, lowering the barrier to entry for contract manufacturing organizations and expanding the pool of potential suppliers.
- Cost Reduction in Manufacturing: The elimination of complex multi-step protection strategies and the use of efficient crystallization for purification significantly lower the operational costs associated with ligand production. By avoiding expensive chromatographic separations and reducing solvent consumption, manufacturers can achieve substantial cost savings that can be passed down the supply chain. The high yields reported in the patent examples indicate minimal waste generation, which further contributes to economic efficiency and aligns with green chemistry principles. This cost structure makes the technology attractive for large-volume applications where margin pressure is a critical consideration.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent batch-to-batch quality, which is vital for maintaining trust between suppliers and pharmaceutical clients. The use of common solvents and reagents means that sourcing is not dependent on single-source vendors, thereby diversifying risk and enhancing supply chain resilience. For supply chain heads, this reliability translates to reduced lead time for high-purity fine chemicals and greater flexibility in responding to market demand fluctuations. The stability of the final product also simplifies storage and transportation logistics, reducing the risk of degradation during transit.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-kilogram commercial production without significant re-engineering. The reduced use of hazardous reagents and the generation of less chemical waste simplify environmental compliance and waste disposal procedures. This alignment with regulatory standards facilitates faster approval processes and reduces the administrative burden on EHS teams. The ability to scale up complex chiral catalysts efficiently ensures that the technology can support the growing demand for enantiomerically pure intermediates in the global pharmaceutical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this spiro-silicon ligand technology. These answers are derived directly from the patent specifications and are intended to clarify the operational capabilities and performance metrics of the compounds. Understanding these details helps stakeholders make informed decisions about integrating this technology into their existing synthetic platforms.
Q: What distinguishes this silicon-centered spiro ligand from traditional carbon-based SPINOL ligands?
A: The silicon-centered skeleton features a longer C-Si bond length compared to C-C bonds and a larger atomic radius for silicon, creating a distinct steric environment that enhances chiral induction in asymmetric catalysis.
Q: Can these ligands be scaled for industrial pharmaceutical intermediate manufacturing?
A: Yes, the synthesis utilizes cheap and readily available initial raw materials with mild reaction conditions and convenient purification steps like crystallization, supporting robust commercial scale-up.
Q: What specific asymmetric reactions are catalyzed by these spiro compounds?
A: They are effective in palladium-catalyzed non-activated C(sp3)-H enantioselective alkynylation, copper-catalyzed intramolecular Ullmann C-N coupling, and asymmetric allylation of ketones.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro-bis-dihydrobenzosilole Diphenol Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and process development, possessing the technical expertise to translate complex patent methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral ligand meets the highest international standards. Our commitment to quality and consistency makes us a trusted partner for multinational corporations seeking to secure their supply of critical catalytic materials.
We invite you to engage with our technical procurement team to discuss how this innovative silicon-centered ligand can optimize your asymmetric synthesis processes. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this novel scaffold. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your specific project requirements. Let us collaborate to drive innovation and efficiency in your chemical manufacturing operations.
