Scalable Manufacturing of Fluzoparib Intermediates via Novel Wurtz-Fittig Coupling Technology
Scalable Manufacturing of Fluzoparib Intermediates via Novel Wurtz-Fittig Coupling Technology
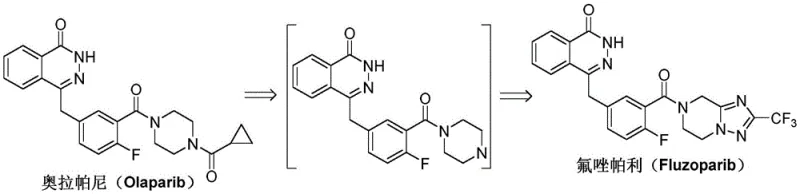
The pharmaceutical landscape for PARP inhibitors has evolved significantly with the emergence of Fluzoparib, a potent agent developed for treating advanced ovarian and fallopian tube cancers. As detailed in the recent patent documentation CN111732594A, a novel preparation method has been disclosed that fundamentally alters the synthetic strategy for this critical active pharmaceutical ingredient. Unlike previous iterations that relied heavily on precious metal catalysis and complex condensation reagents, this new methodology leverages a robust Wurtz-Fittig coupling mechanism to construct the core molecular scaffold. This technological shift represents a paradigm change in how high-purity pharmaceutical intermediates are manufactured, offering a pathway that is not only chemically elegant but also industrially pragmatic. By transitioning away from palladium-dependent hydrogenation steps, the process mitigates the risks associated with heavy metal contamination and reduces the overall environmental footprint of the synthesis. For stakeholders in the fine chemical sector, understanding the nuances of this patent is essential for evaluating supply chain resilience and cost structures in the coming years.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
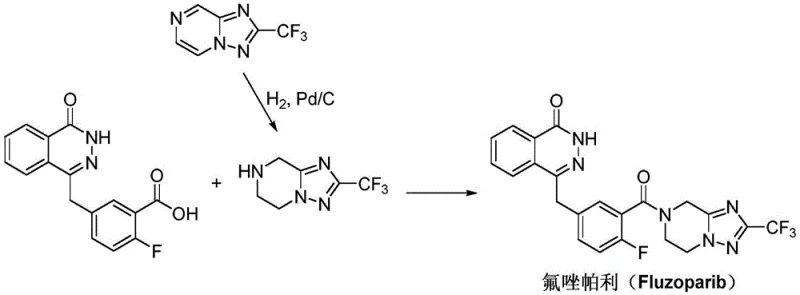
Prior art synthesis routes, such as those described in patents CN102686591B and WO2012019427A1, typically necessitate a multi-step sequence involving Pd/C catalytic hydrogenation to reduce the triazolo-pyrazine core. This reliance on heterogeneous catalysis introduces significant operational complexities, particularly regarding the removal of trace palladium residues which is a stringent requirement for final drug substances. Furthermore, the subsequent amidation condensation between the carboxylic acid and the amine component often demands expensive coupling reagents to drive the reaction to completion. These reagents not only inflate the raw material costs but also generate substantial amounts of byproduct waste that must be meticulously separated during downstream processing. The cumulative effect of these factors is a process that is difficult to scale economically, with purification bottlenecks that can severely impact production throughput and lead times for reliable pharmaceutical intermediate supplier networks.
The Novel Approach
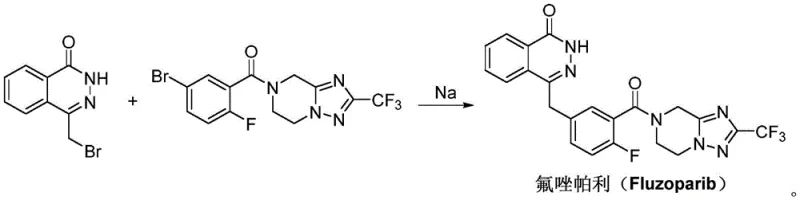
In stark contrast, the methodology outlined in CN111732594A introduces a streamlined three-step sequence that bypasses the need for catalytic hydrogenation entirely. The process initiates with a straightforward acyl chlorination of 5-bromo-2-fluorobenzoic acid, followed by a direct amidation with the tetrahydro-triazolo-pyrazine derivative. The crown jewel of this synthetic strategy is the final coupling step, which employs a Wurtz-Fittig reaction mediated by metallic sodium and tetraphenylethylene. This approach effectively links the phthalazinone moiety with the fluorophenyl-triazole segment in a single operation, eliminating the need for pre-functionalization of the acid component into a complex activated ester. By utilizing commodity chemicals like sodium metal and avoiding proprietary coupling agents, the novel route drastically simplifies the material inventory required for production. This simplification translates directly into enhanced process robustness, allowing for more predictable batch cycles and reduced dependency on specialized reagent supply chains.
Mechanistic Insights into Wurtz-Fittig Coupling and Impurity Control
The core innovation of this synthesis lies in the application of the Wurtz-Fittig coupling reaction to join two distinct aromatic systems under mild thermal conditions. In this specific transformation, metallic sodium acts as a single-electron donor, facilitating the formation of radical intermediates that subsequently recombine to form the new carbon-carbon bond between the bromomethyl group of the phthalazinone and the aryl bromide of the triazole intermediate. The inclusion of tetraphenylethylene serves a critical role as a radical scavenger or mediator, helping to stabilize the reaction environment and suppress side reactions that could lead to homocoupling byproducts. This mechanistic pathway is particularly advantageous because it operates effectively in common organic solvents such as toluene or tetrahydrofuran at moderate temperatures ranging from 50°C to 80°C. The reaction kinetics are favorable enough to achieve high conversion rates within a 2 to 6-hour window, ensuring that the throughput remains high without requiring extreme energy inputs or prolonged reaction times that could degrade sensitive functional groups.
From a quality control perspective, the impurity profile generated by this route is significantly cleaner than that of traditional amide coupling methods. Because the reaction does not involve the formation of urea or phosphonium salt byproducts typical of peptide coupling reagents, the post-reaction mixture is far easier to purify. The patent data indicates that the crude product can often be purified to specification simply through recrystallization from mixed solvent systems like ethyl acetate and petroleum ether, completely obviating the need for silica gel column chromatography. This is a critical advantage for commercial scale-up of complex pharmaceutical intermediates, as column chromatography is notoriously difficult to implement on a multi-ton scale due to solvent volume constraints and safety hazards. By designing a process where impurities are either insoluble or easily washed away during aqueous workups, the method ensures that the final high-purity OLED material or API precursor meets stringent regulatory standards with minimal processing overhead.
How to Synthesize Fluzoparib Efficiently
The execution of this synthesis requires precise control over stoichiometry and temperature to maximize yield while maintaining safety, particularly during the handling of reactive sodium metal. The process begins with the activation of the benzoic acid derivative, followed by the construction of the central amide bond, and culminates in the reductive coupling step. Each stage has been optimized to balance reaction rate with selectivity, ensuring that the intermediate species remain stable throughout the sequence. Operators must adhere to strict anhydrous conditions during the acyl chlorination and coupling phases to prevent hydrolysis of the reactive acid chloride and quenching of the sodium reagent. The following guide outlines the standardized operational parameters derived from the patent examples, providing a clear roadmap for technical teams aiming to replicate this efficient manufacturing protocol.
- Perform acyl chlorination on 5-bromo-2-fluorobenzoic acid using reagents like oxalyl chloride or thionyl chloride at 20-80°C to generate the reactive acyl chloride intermediate.
- Conduct an amidation reaction between the resulting acyl chloride and 2-(trifluoromethyl)-5,6,7,8-tetrahydro-[1,2,4]triazolo[1,5-a]pyrazine in the presence of an acid-binding base.
- Execute a Wurtz-Fittig coupling reaction using sodium metal and tetraphenylethylene to link the bromomethyl-phthalazinone with the triazole-containing ketone intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond simple chemistry. The primary value proposition lies in the decoupling of production from volatile markets for precious metals and specialized coupling reagents. By shifting to a chemistry based on abundant alkali metals and bulk solvents, manufacturers can insulate their cost structures from the price fluctuations that frequently plague palladium and exotic ligand markets. Furthermore, the simplification of the purification workflow means that facility utilization rates can be improved, as reactors are not tied up for extended periods during tedious chromatographic separations. This efficiency gain allows for faster turnover of batches, directly addressing the industry-wide challenge of reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive coupling reagents and palladium catalysts results in a substantial decrease in raw material expenditures per kilogram of output. Additionally, the removal of column chromatography steps significantly lowers solvent consumption and waste disposal costs, which are often hidden but major contributors to the total cost of goods sold. The use of sodium metal, a commodity chemical, further drives down the direct material costs compared to proprietary activating agents. This economic efficiency allows for more competitive pricing strategies in the global market for API intermediates without compromising on margin.
- Enhanced Supply Chain Reliability: Sourcing risks are minimized because the key reagents, such as 5-bromo-2-fluorobenzoic acid and sodium metal, are widely available from multiple global suppliers. This diversification of the supply base ensures continuity of operations even when specific vendors face disruptions. The robustness of the Wurtz-Fittig coupling also means that the process is less sensitive to minor variations in reagent quality, reducing the frequency of batch failures and the need for rework. Consequently, partners can rely on more consistent delivery schedules and greater flexibility in order volumes.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing unit operations like crystallization and filtration that are standard in large-scale chemical plants. The avoidance of heavy metal catalysts simplifies the environmental compliance burden, as there is no need for complex metal recovery systems or rigorous testing for residual palladium in the final product. This aligns perfectly with modern green chemistry principles, reducing the generation of hazardous waste and lowering the overall environmental footprint of the manufacturing site. Such sustainability credentials are increasingly important for maintaining partnerships with top-tier pharmaceutical companies focused on ESG goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical aspects of the method. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the long-term viability of the supply partnership.
Q: Why is the Wurtz-Fittig coupling preferred over traditional amide condensation for Fluzoparib?
A: Traditional methods often rely on expensive coupling reagents and Pd/C catalytic hydrogenation, which increase costs and complicate purification. The Wurtz-Fittig approach utilizes readily available sodium metal and simplifies the workflow by avoiding complex catalyst removal steps.
Q: What are the critical reaction conditions for the acyl chlorination step?
A: The acyl chlorination of 5-bromo-2-fluorobenzoic acid is typically conducted at temperatures between 20°C and 80°C for 1 to 6 hours, using reagents such as oxalyl chloride or thionyl chloride to ensure high conversion to the acid chloride.
Q: How does this process ensure scalability for industrial production?
A: The process eliminates the need for column chromatography purification, relying instead on standard recrystallization techniques. This significantly reduces solvent consumption and processing time, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluzoparib Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory discovery to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics observed in patent examples can be reliably reproduced in our facilities. We maintain stringent purity specifications through our rigorous QC labs, employing advanced analytical techniques to verify that every batch of Fluzoparib intermediate meets the exacting standards required for clinical and commercial applications. Our commitment to quality assurance means that we proactively manage impurity profiles, ensuring that the final product is free from genotoxic impurities and residual metals.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your product quality and your bottom line. Together, we can accelerate the availability of this vital cancer therapy to patients worldwide.
