Advanced Neutral Tridentate Iridium Complexes for Next-Generation Red OLED Manufacturing
Advanced Neutral Tridentate Iridium Complexes for Next-Generation Red OLED Manufacturing
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that offer not only superior electroluminescent efficiency but also exceptional thermal stability and ease of purification. Patent CN102268250A introduces a groundbreaking approach to synthesizing a novel electrically neutral tridentate iridium(III) complex, specifically designed to emit high-purity red light. This technology addresses the longstanding challenge of geometric isomerism found in traditional bidentate systems by utilizing a unique 6-[5-trifluoromethylpyrazole]-2,2′-bipyridine ligand coordinated with 2,6-diphenylpyridine. For R&D directors and procurement specialists seeking a reliable OLED material supplier, this patent represents a significant leap forward in creating robust, high-performance phosphorescent dopants that can be scaled for commercial display and lighting applications without the baggage of complex isomeric mixtures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional phosphorescent iridium complexes typically rely on the coordination of three bidentate ligands to the central metal atom, forming an octahedral geometry. While effective, this architecture suffers from a critical flaw: the potential for multiple geometric isomers due to the asymmetry of the ligands. These isomers often possess nearly identical physical properties, making their separation via standard chromatographic techniques extremely difficult and costly. Furthermore, these metastable isomers can undergo interconversion or decomposition under the thermal and electrical stress of device operation, leading to unpredictable shifts in emission color and reduced device lifetime. The presence of such impurities creates a bottleneck in the supply chain, requiring extensive purification resources that drive up the cost of goods sold and delay time-to-market for new electronic chemical manufacturing projects.
The Novel Approach
The innovation detailed in the patent data circumvents these issues by employing two tridentate chelating ligands instead of three bidentate ones. This structural modification locks the iridium center into a rigid, well-defined geometry that effectively eliminates the possibility of geometric isomer formation. The resulting complex, characterized by its distinct orange-red solid state, exhibits superior homogeneity. 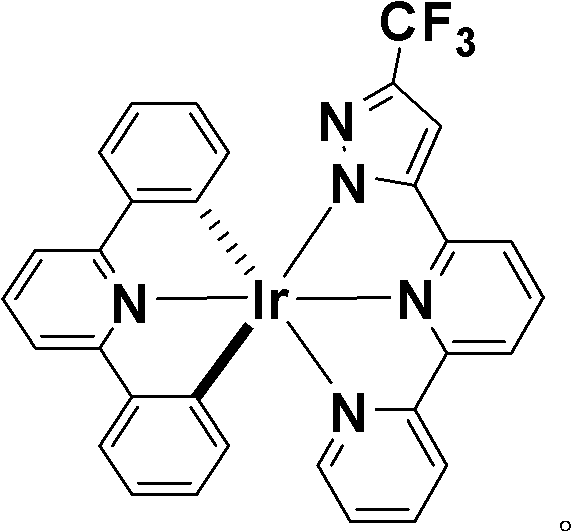 As illustrated in the molecular structure, the incorporation of the trifluoromethyl group on the pyrazole ring not only enhances the electron-withdrawing capability to tune the emission wavelength but also improves the solubility of the complex in common organic solvents. This design ensures that the final product is a single, pure species, drastically simplifying the quality control process and ensuring consistent batch-to-batch performance for high-purity OLED material production.
As illustrated in the molecular structure, the incorporation of the trifluoromethyl group on the pyrazole ring not only enhances the electron-withdrawing capability to tune the emission wavelength but also improves the solubility of the complex in common organic solvents. This design ensures that the final product is a single, pure species, drastically simplifying the quality control process and ensuring consistent batch-to-batch performance for high-purity OLED material production.
Mechanistic Insights into Tridentate Ligand Coordination and Solvent-Free Synthesis
The synthesis mechanism relies on a strategic two-step process that maximizes yield while minimizing environmental impact. The first step involves the formation of a chloro-bridged dimer intermediate by reacting iridium trichloride trihydrate with the trifluoromethyl-substituted bipyridine-pyrazole ligand in ethanol. This step establishes the primary coordination sphere. The critical innovation occurs in the second step, where the chloro-bridged intermediate reacts with 2,6-diphenylpyridine in the presence of silver trifluoromethanesulfonate. Unlike conventional methods that require large volumes of high-boiling solvents like ethylene glycol, this process utilizes a solvent-free technique. 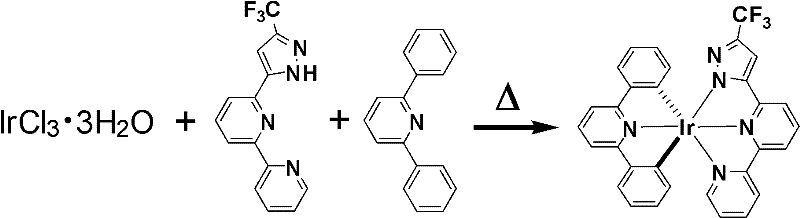 The reaction mixture is ground into a fine powder and heated to 110°C, where the excess 2,6-diphenylpyridine ligand melts and acts as both the reactant and the reaction medium. This molten state facilitates efficient mass transfer and ligand exchange, driving the reaction to completion without the need for external solvents that would later require energy-intensive removal.
The reaction mixture is ground into a fine powder and heated to 110°C, where the excess 2,6-diphenylpyridine ligand melts and acts as both the reactant and the reaction medium. This molten state facilitates efficient mass transfer and ligand exchange, driving the reaction to completion without the need for external solvents that would later require energy-intensive removal.
From a mechanistic purity perspective, the use of the silver salt serves to abstract the chloride ions from the iridium center, generating a cationic intermediate that is highly reactive towards the incoming neutral tridentate ligand. The strong electron-withdrawing nature of the trifluoromethyl group plays a dual role here: it increases the acidity of the pyrazole NH proton, making it easier to deprotonate during coordination, and it sterically hinders adjacent nitrogen atoms to prevent unwanted side reactions. This precise control over the reaction pathway ensures that the final product is obtained with a yield ranging from 30% to 68%, which is highly competitive for such a complex organometallic transformation. The elimination of solvent residues also means the final material has a cleaner impurity profile, which is essential for preventing quenching sites in the emissive layer of an OLED device.
How to Synthesize Ir[III][bpyfpz][dppy] Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and atmospheric control to ensure reproducibility at scale. The process begins with the rigorous exclusion of oxygen and moisture, as the iridium precursors are sensitive to hydrolysis. The initial reflux in ethanol must be maintained for approximately 48 hours to ensure complete consumption of the metal salt, followed by a precipitation step using acetone and ether to isolate the orange-yellow intermediate. The subsequent solvent-free coupling reaction is the heart of the process, where the molar ratio of the intermediate to the ancillary ligand is critical; patents suggest a range of 1:10 to 1:50 to drive the equilibrium forward. Detailed standardized operating procedures for scaling this pathway from gram to kilogram quantities are provided below to assist process engineers in validating the route.
- Reflux IrCl3·3H2O with 6-[5-trifluoromethylpyrazole]-2,2'-bipyridine in ethanol at 78°C for 2 days to form the orange-yellow chloro-bridged intermediate.
- Grind the intermediate with silver trifluoromethanesulfonate and excess 2,6-diphenylpyridine ligand into a fine powder under nitrogen protection.
- Heat the mixture to 110°C for 24 hours in a solvent-free environment, then purify the crude product via silica gel chromatography using dichloromethane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this tridentate synthesis route offers tangible logistical and financial benefits beyond mere technical performance. The most significant advantage lies in the drastic simplification of the purification workflow. By eliminating geometric isomers at the source, the need for repetitive preparative HPLC or complex fractional crystallization is removed, leading to substantial cost savings in downstream processing. Additionally, the solvent-free nature of the second reaction step aligns perfectly with modern green chemistry initiatives, reducing the volume of hazardous waste generated and lowering the associated disposal fees. This streamlined process enhances the overall reliability of the supply chain by reducing the number of unit operations where yield losses typically occur.
- Cost Reduction in Manufacturing: The elimination of high-boiling organic solvents in the second synthetic step removes the need for energy-intensive distillation and solvent recovery systems. This directly translates to lower utility costs and reduced capital expenditure on specialized equipment. Furthermore, the higher selectivity of the tridentate ligand system means that raw materials are converted more efficiently into the desired product rather than wasted on inseparable isomers, optimizing the cost per gram of the final high-purity OLED material.
- Enhanced Supply Chain Reliability: The robustness of the solvent-free method makes the process less sensitive to variations in solvent quality or availability, which can be a bottleneck in global chemical supply chains. The reagents used, such as 2,6-diphenylpyridine and silver trifluoromethanesulfonate, are commercially available commodity chemicals, ensuring a stable supply base. This stability allows for more accurate forecasting and inventory management, reducing the risk of production delays caused by raw material shortages.
- Scalability and Environmental Compliance: Scaling solvent-free reactions is often safer and more straightforward than managing large volumes of flammable organic solvents at elevated temperatures. The reduced solvent load significantly lowers the facility's fire hazard rating and simplifies compliance with increasingly stringent environmental regulations regarding VOC emissions. This makes the technology highly attractive for contract manufacturing organizations looking to expand capacity without incurring heavy regulatory burdens or needing massive infrastructure upgrades.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the stability, purification, and application of this specific iridium complex class. Understanding these nuances is vital for integration into device architectures and for assessing the long-term viability of the material in commercial products. The answers provided are derived directly from the experimental data and theoretical framework established in the patent literature.
Q: Why are tridentate ligands preferred over bidentate ligands for this iridium complex?
A: Tridentate ligands significantly reduce the formation of geometric isomers compared to traditional three-bidentate-ligand systems. This structural rigidity simplifies purification and enhances thermal stability, which is critical for the longevity of OLED devices.
Q: What are the environmental benefits of the solvent-free second step?
A: The second synthetic step utilizes the molten ligand itself as the reaction medium at 110°C, eliminating the need for volatile organic solvents. This reduces waste disposal costs, minimizes environmental impact, and simplifies the downstream workup process.
Q: How does the trifluoromethyl group affect the material properties?
A: The introduction of the electron-withdrawing trifluoromethyl group enhances the proton acidity of the pyrazole NH, facilitating deprotonation. It also improves the photoluminescence quantum efficiency and solubility of the final complex, making it more suitable for solution-processable device fabrication.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ir[III][bpyfpz][dppy] Supplier
As the demand for high-efficiency red emitters continues to surge in the display and lighting sectors, having a partner with deep expertise in organometallic synthesis is crucial. NINGBO INNO PHARMCHEM stands ready to support your development goals with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of complex organometallics meets the exacting standards required for next-generation optoelectronic devices. We understand the critical nature of impurity profiles in OLED materials and employ advanced analytical techniques to guarantee consistency.
We invite you to engage with our technical procurement team to discuss how this novel tridentate iridium technology can be integrated into your supply chain. Whether you require a Customized Cost-Saving Analysis for your current material sourcing or need specific COA data and route feasibility assessments for new project validation, our experts are prepared to provide the detailed support necessary to accelerate your time-to-market. Contact us today to secure a reliable supply of high-performance electronic chemicals tailored to your specific application needs.
