Advanced Synthesis of Racemic Camptothecin Intermediates for Commercial Scale-Up
The pharmaceutical industry's relentless pursuit of potent antitumor agents has long centered on camptothecin (CPT) and its derivatives, renowned for their unique mechanism as specific inhibitors of topoisomerase I. However, the commercial viability of these life-saving drugs often hinges on the efficiency of synthesizing their core structural motifs. Patent CN116120332A introduces a groundbreaking synthesis method for the racemic camptothecin intermediate tricyclic compound, specifically 4-ethyl-7,8-dihydro-IH-pyrano[3,4-f]indolizine-3,6,10(4H)-trione. This innovation directly addresses the critical bottlenecks of cost, safety, and scalability that have plagued previous manufacturing attempts. By leveraging a sequence of conventional organic reactions under mild conditions, this new methodology offers a robust pathway for producing high-purity pharmaceutical intermediates. For R&D directors and supply chain leaders, this represents a significant opportunity to optimize the production of CDE rings, which are essential precursors for the Friedlander condensation used in finalizing camptothecin structures. The strategic shift away from hazardous reagents and expensive catalysts underscores a commitment to sustainable and economically viable chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
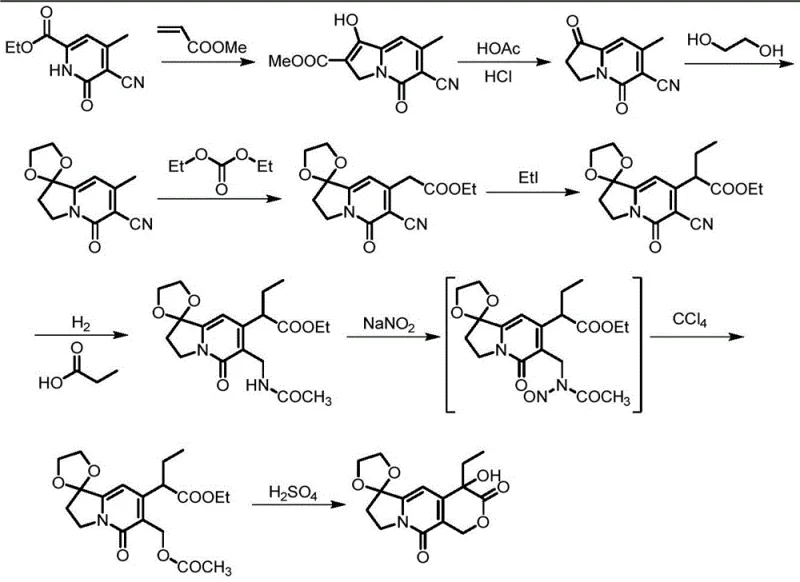
Historically, the synthesis of the racemic CDE ring has been fraught with technical and economic challenges that hinder large-scale adoption. As detailed in the background art, early approaches such as the route described in J. Med. Chem. (1980) suffered from critically low yields in key steps, particularly the initial formation and subsequent ethylation stages. This route necessitated the use of Raney nickel, a pyrophoric catalyst that poses significant safety risks and requires complex handling protocols to prevent ignition and ensure worker safety. Furthermore, the reliance on expensive starting materials like ethyl 5-cyano-4-methyl-6-oxylidene-1,6-dihydropyridine-2-carboxylate drove up the cost of goods significantly. Later iterations, such as the method found in J. Org. Chem. (1993), attempted to streamline the process but introduced new liabilities, including the use of hydrobromic acid for decarboxylation which resulted in poor yields and the generation of highly toxic waste streams that increased environmental compliance costs. More recent strategies involving transition metal catalysts and toxic carbon monoxide gas, while chemically elegant, are ill-suited for mass production due to the extreme safety hazards and the high capital expenditure required for specialized high-pressure equipment.
The Novel Approach
In stark contrast to these legacy methods, the novel approach outlined in patent CN116120332A fundamentally reimagines the synthetic landscape by prioritizing operational simplicity and原料 availability. This new methodology replaces hazardous gases and precious metal catalysts with stable, commodity chemicals that are readily accessible in the global supply chain. The reaction conditions are remarkably mild, predominantly occurring between 0°C and 30°C, which drastically reduces energy consumption and eliminates the need for cryogenic cooling or high-temperature heating that can degrade sensitive intermediates. By shortening the synthetic route and improving the yield at each stage, the overall process efficiency is substantially enhanced. This reduction in step count not only accelerates the time-to-market for the final API but also minimizes the accumulation of impurities, thereby simplifying downstream purification. For procurement managers, this translates to a more reliable supply of critical intermediates with reduced volatility in pricing, as the process is less susceptible to fluctuations in the market for exotic reagents.
Mechanistic Insights into the Multi-Step Cyclization Strategy
The core of this technological breakthrough lies in a meticulously orchestrated eight-step sequence that constructs the complex tricyclic framework with high precision. The process initiates with the activation of dimethyl 1,3-acetonedicarboxylate using a chlorinating agent like 2-chloro-1,3-dimethyl imidazoline chloride, followed by a condensation with 2-pyrrolidone derivatives to form the foundational nitrogen-containing heterocycle. A crucial Knovenagel condensation step then establishes the necessary carbon-carbon double bonds, setting the stage for subsequent functionalization. The introduction of the ethyl group is achieved through a controlled alkylation using potassium tert-butoxide and iodoethane, a reaction that proceeds with excellent conversion rates under the specified mild thermal conditions. The construction of the pyran ring is elegantly accomplished via cyclization with paraformaldehyde in the presence of an acid catalyst, a transformation that closes the third ring of the system efficiently. Finally, the sequence employs ozonolysis for oxidative cleavage and a protective group strategy using ethylene glycol to stabilize the ketone functionalities, ensuring the integrity of the molecule throughout the synthesis.
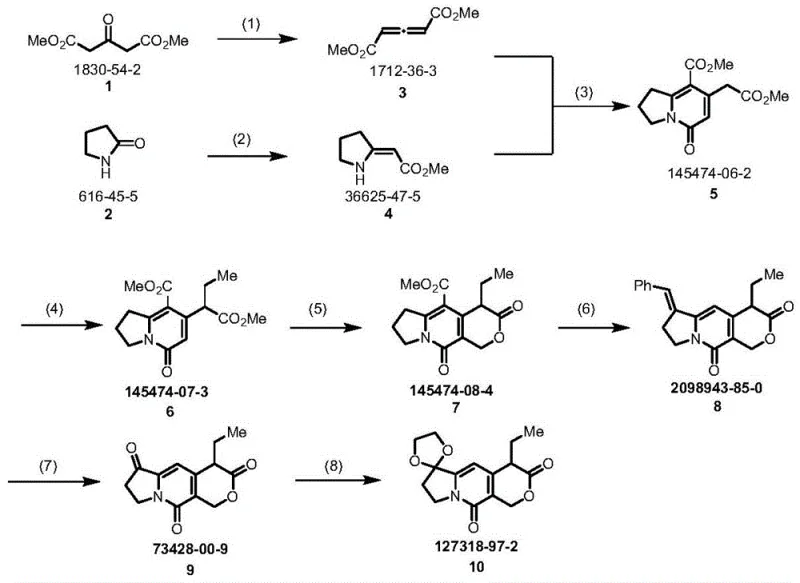
From an impurity control perspective, the choice of reagents and conditions in this new route is strategically designed to minimize side reactions. The avoidance of strong oxidants like potassium permanganate or harsh acidic conditions in the early stages prevents the degradation of the sensitive indolizine core. The use of ozone for oxidation, while requiring careful control, offers a cleaner alternative to heavy metal oxidants, generating volatile byproducts that are easily removed, thus enhancing the purity profile of the intermediate. The final protection step using trimethylchlorosilane ensures that the reactive carbonyl groups are masked effectively, preventing unwanted polymerization or decomposition during storage and transport. This level of mechanistic control is vital for R&D teams aiming to meet stringent regulatory specifications for pharmaceutical intermediates, as it ensures a consistent and reproducible quality profile batch after batch. The ability to purify the final product through standard recrystallization or chromatography further validates the robustness of this chemical design.
How to Synthesize 4-Ethyl-7,8-dihydro-IH-pyrano[3,4-f]indolizine-3,6,10(4H)-trione Efficiently
Implementing this synthesis requires a disciplined approach to reaction monitoring and workup procedures to maximize yield and purity. The process is divided into distinct operational units, beginning with the preparation of the dienyl ester and proceeding through condensation, ethylation, and cyclization. Each step is optimized for scalability, utilizing solvents like dichloromethane, toluene, and ethanol which are standard in industrial settings. The detailed standardized synthesis steps below outline the specific molar ratios, temperatures, and processing times required to replicate the high yields reported in the patent data. Adhering to these parameters is essential for maintaining the structural integrity of the intermediate and ensuring that the final product meets the rigorous demands of downstream API synthesis.
- Preparation of dienyl ester via chlorination of dimethyl 1,3-acetonedicarboxylate followed by condensation with 2-pyrrolidone derivatives.
- Execution of Knovenagel condensation and subsequent ethylation using potassium tert-butoxide and iodoethane to establish the carbon skeleton.
- Cyclization using paraformaldehyde and acid, followed by alkylation, decarboxylation, ozonolysis, and final ethylene glycol protection to yield the target tricyclic compound.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the bottom line and operational continuity, the shift to this novel synthesis route offers profound strategic benefits that extend beyond mere chemical curiosity. The redesign of the manufacturing process directly targets the pain points of cost volatility and supply chain fragility that often disrupt the production of complex oncology intermediates. By eliminating dependencies on scarce or hazardous materials, the new method creates a more resilient supply network capable of withstanding market shocks. The simplification of the process flow also reduces the burden on quality control laboratories, allowing for faster release times and improved inventory turnover. These factors combined create a compelling business case for adopting this technology in commercial-scale operations.
- Cost Reduction in Manufacturing: The economic impact of this new route is driven primarily by the substitution of expensive and specialized reagents with commodity chemicals. By removing the need for transition metal catalysts such as palladium and avoiding the use of toxic carbon monoxide gas, the process eliminates the substantial costs associated with catalyst recovery, heavy metal scavenging, and specialized gas handling infrastructure. Furthermore, the higher yields achieved in each step mean that less raw material is wasted, directly lowering the cost per kilogram of the final intermediate. The reduction in waste disposal costs, owing to the absence of toxic heavy metal sludge and hazardous gaseous byproducts, further contributes to significant overall savings in the manufacturing budget.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of widely available starting materials like dimethyl 1,3-acetonedicarboxylate and 2-pyrrolidone, which are produced in vast quantities by the global chemical industry. Unlike the proprietary or niche reagents required by older methods, these feedstocks are not subject to the same supply constraints or geopolitical risks. The mild reaction conditions also allow for production in a broader range of facilities, reducing the dependency on specialized plants equipped for high-pressure or cryogenic operations. This flexibility ensures a more consistent supply of the intermediate, reducing lead times and mitigating the risk of production stoppages due to raw material shortages.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial production is facilitated by the use of conventional unit operations and standard reactor configurations. The absence of high-pressure carbon monoxide steps removes a major barrier to scale-up, as it negates the need for expensive autoclaves and complex safety interlocks. From an environmental standpoint, the process aligns with green chemistry principles by minimizing the use of toxic substances and reducing the generation of hazardous waste. This compliance with increasingly stringent environmental regulations reduces the risk of fines and shutdowns, ensuring long-term operational sustainability and protecting the company's reputation as a responsible manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the comparative analysis of the patent data and are intended to clarify the operational advantages for potential partners. Understanding these nuances is critical for making informed decisions about process adoption and supply chain integration.
Q: What are the primary safety advantages of this new synthesis route compared to prior art?
A: The new method eliminates the use of toxic carbon monoxide gas required in previous transition-metal catalyzed routes and avoids the handling complexities associated with Raney nickel. Furthermore, the reaction conditions are significantly milder, typically operating between 0°C and 30°C, which reduces thermal hazards and simplifies process control during large-scale manufacturing.
Q: How does this process address the issue of high raw material costs?
A: Unlike earlier methods that relied on expensive starting materials such as ethyl 5-cyano-4-methyl-6-oxylidene-1,6-dihydropyridine-2-carboxylate or alkenyl chlorides, this novel approach utilizes commercially abundant and inexpensive reagents like dimethyl 1,3-acetonedicarboxylate and 2-pyrrolidone. This shift in feedstock significantly lowers the overall bill of materials for the synthesis.
Q: Is the purification process suitable for GMP manufacturing environments?
A: Yes, the process is designed for ease of purification. The synthetic route avoids the generation of complex impurity profiles often associated with harsh oxidation or transition metal catalysis. Standard workup procedures involving extraction, drying, and recrystallization or column chromatography are sufficient to achieve high purity, making it highly compatible with Good Manufacturing Practice (GMP) requirements for pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Ethyl-7,8-dihydro-IH-pyrano[3,4-f]indolizine-3,6,10(4H)-trione Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex oncology drugs depends on the reliability and quality of their key intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demanding volume requirements of global pharmaceutical clients. We are committed to delivering high-purity pharmaceutical intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in process optimization allows us to implement advanced synthetic routes like the one described in CN116120332A, guaranteeing a supply chain that is both cost-effective and robust against disruptions.
We invite you to collaborate with us to leverage these technological advancements for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating exactly how this new synthesis route can improve your margins. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to accelerate the delivery of life-saving therapies to patients worldwide.
