Advanced Chemical Synthesis of Cholic Acid Intermediate A8 for Pharmaceutical Manufacturing
Advanced Chemical Synthesis of Cholic Acid Intermediate A8 for Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust, scalable, and safe methods for producing critical bile acid derivatives, which serve as essential building blocks for various therapeutic agents. Patent CN115536720A introduces a groundbreaking chemical synthesis method for producing cholic acid intermediate A8 and ultimately cholic acid itself. This innovation addresses a significant historical challenge in the sector: the reliance on extraction from animal internal organs, which carries inherent risks of virus transmission and supply chain volatility. By shifting the production paradigm to a total chemical synthesis starting from common plant-source compounds, this technology offers a virus-free alternative that aligns with modern safety standards and regulatory requirements for high-purity active pharmaceutical ingredients.
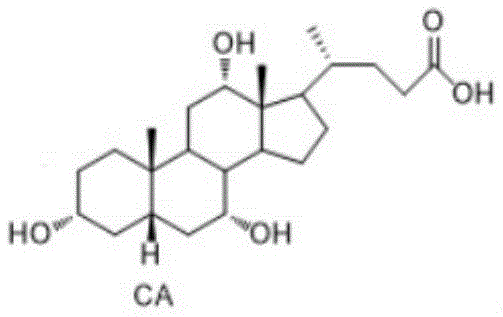
Cholic acid is a pivotal molecule in biochemical research and serves as a medical intermediate for synthesizing other bile acids like ursodeoxycholic acid and lithocholic acid. Traditionally, the market supply has depended on refining internal organs from cattle or sheep, a process fraught with biological contamination risks. The novel approach detailed in the patent utilizes a sophisticated multi-step organic synthesis pathway. This pathway begins with a plant-derived precursor, designated as compound SM (9α-hydroxy BA), and progresses through a series of meticulously optimized transformations including oxidation, elimination, Wittig olefination, ketal protection, and selective reductions. This shift not only mitigates biological hazards but also establishes a foundation for consistent quality control that is difficult to achieve with biological extraction methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the procurement of cholic acid and its intermediates has been heavily dependent on the extraction from animal bile or viscera. This conventional method presents severe limitations for large-scale pharmaceutical manufacturing, primarily concerning biosafety and supply stability. The extraction process inevitably carries the risk of virus infection, which is a critical concern for regulatory bodies overseeing the production of injectables or oral medications intended for chronic use. Furthermore, the quality of extracted materials can vary significantly based on the health and diet of the source animals, leading to inconsistent impurity profiles that complicate downstream purification. From a supply chain perspective, reliance on slaughterhouse by-products makes the availability of these intermediates susceptible to fluctuations in the livestock industry, creating unpredictable lead times for reliable pharmaceutical intermediate suppliers.
The Novel Approach
The synthetic methodology disclosed in the patent represents a paradigm shift towards fully chemical manufacturing. By utilizing plant-source compounds as the starting material, the process completely bypasses the biological risks associated with animal extraction. The route is designed with mild reaction conditions, particularly in the later stages involving intermediate A8, which facilitates easier scale-up and reduces energy consumption. For instance, the hydrolysis of the ester side chain in compound A7 to form A8 is conducted at moderate temperatures of 40-60°C using common solvents like methanol or ethanol. This contrasts sharply with harsh extraction protocols and allows for precise control over reaction kinetics. The result is a high-purity intermediate that serves as a superior substrate for the final reduction to cholic acid, ensuring a cleaner final product with a well-defined impurity spectrum.
Mechanistic Insights into Selective Reduction and Hydrolysis
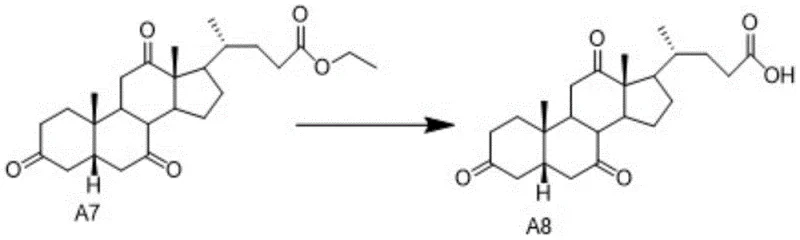
The core of this synthetic innovation lies in the precise control of functional group transformations, particularly the conversion of intermediate A7 to A8 and subsequently to Cholic Acid. The transformation of A7 to A8 involves a base-catalyzed hydrolysis of the ethyl ester side chain. The patent specifies that this reaction proceeds efficiently in solvents such as tetrahydrofuran, acetone, or mixtures of dichloromethane and alcohols. The use of sodium hydroxide or potassium hydroxide in specific mass ratios relative to the substrate ensures complete conversion without degrading the sensitive steroid backbone. Following this, the crude product undergoes a rigorous post-treatment involving pH adjustment to 2-3 using hydrochloric acid, which precipitates the free acid form of intermediate A8. This step is crucial for removing inorganic salts and residual base, setting the stage for the final reduction.
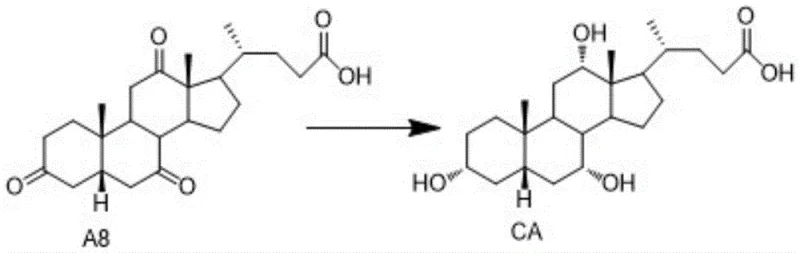
The final step, converting intermediate A8 to Cholic Acid (CA), employs a highly selective reduction strategy. The patent highlights the use of lithium aluminum tri-tert-butoxy hydride as the reducing agent. This bulky hydride reagent is chosen specifically for its ability to reduce ketones to alcohols while tolerating other functional groups, although in this specific context, it reduces the three ketone groups on the steroid nucleus to their corresponding hydroxyl groups with specific stereochemistry. The reaction is initiated at low temperatures (0-5°C) under nitrogen protection to manage exothermicity and prevent side reactions, before warming to 20-25°C for completion over 8 to 13 hours. This controlled addition and temperature profile are vital for achieving the correct stereochemical configuration at the C3, C7, and C12 positions, which defines the biological activity of cholic acid.
How to Synthesize Cholic Acid Intermediate A8 Efficiently
The synthesis of cholic acid intermediate A8 is a critical juncture in the overall production of cholic acid, requiring strict adherence to the patented parameters to ensure yield and purity. The process involves dissolving the precursor compound A7 in a specific solvent system, such as methanol or a dichloromethane-methanol mixture, and treating it with an alkaline solution. The reaction temperature must be carefully maintained between 40-60°C for a duration of 3 to 5 hours to ensure complete hydrolysis of the ester moiety. Upon completion, the reaction mixture is cooled to room temperature, and the pH is adjusted to acidic conditions to isolate the product. Detailed standard operating procedures regarding reagent grades, stirring speeds, and filtration techniques are essential for reproducibility.
- Oxidize starting material SM to aldehyde A1 using TEMPO/NaOCl or Jones reagent.
- Perform elimination on A1 to form enone A2, followed by Wittig reaction to extend the side chain to A3.
- Protect the ketone in A3 to form ketal A4, then oxidize to tri-ketone A5 using chromium-based oxidants.
- Hydrolyze A5 to A6, hydrogenate to A7 using Pd/C, and hydrolyze the ester to obtain intermediate A8.
- Selectively reduce intermediate A8 with lithium aluminum tri-tert-butoxy hydride to yield final Cholic Acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from animal extraction to chemical synthesis offers profound strategic benefits beyond mere cost considerations. The most significant advantage is the stabilization of the supply chain. By decoupling production from the livestock industry, manufacturers can guarantee continuous availability of high-purity pharmaceutical intermediates regardless of seasonal or agricultural fluctuations. This reliability is paramount for maintaining uninterrupted production schedules for downstream API manufacturing. Furthermore, the synthetic route allows for better inventory management and forecasting, as the lead times for chemical raw materials are generally more predictable than those for biological tissues.
- Cost Reduction in Manufacturing: The synthetic route described eliminates the expensive and logistically complex processes associated with collecting and processing animal viscera. By using common plant-source starting materials and standard chemical reagents, the overall cost of goods sold can be significantly optimized. The mild reaction conditions, such as the hydrolysis at 40-60°C, reduce energy consumption compared to high-temperature or high-pressure alternatives. Additionally, the ability to recycle solvents like methanol and ethanol further contributes to substantial cost savings in large-scale operations.
- Enhanced Supply Chain Reliability: Sourcing raw materials from plant-based chemical suppliers rather than slaughterhouses drastically reduces the risk of supply disruptions caused by animal disease outbreaks or regulatory bans on animal products. This shift ensures a more resilient supply chain capable of meeting the rigorous demands of global pharmaceutical markets. The consistency of the synthetic starting material also simplifies quality assurance protocols, reducing the time and resources spent on testing for biological contaminants.
- Scalability and Environmental Compliance: Chemical synthesis is inherently more scalable than biological extraction. The batch processes described, such as the hydrogenation of A6 to A7 and the hydrolysis of A7 to A8, can be easily transferred from pilot plants to multi-ton commercial reactors. Moreover, the synthetic route avoids the generation of biological waste, simplifying wastewater treatment and environmental compliance. The use of established organic chemistry techniques ensures that the process can be scaled up to meet increasing market demand for cholic acid derivatives without the ethical and environmental concerns associated with animal sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of cholic acid intermediate A8 and its subsequent conversion to cholic acid. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on process capabilities and product quality.
Q: What is the primary advantage of synthesizing Cholic Acid intermediate A8 chemically?
A: The primary advantage is the elimination of viral infection risks associated with extracting bile acids from animal viscera. The synthetic route uses common plant-source compounds as raw materials, ensuring a safer and more consistent supply chain for pharmaceutical applications.
Q: What are the critical reaction conditions for converting A7 to A8?
A: The conversion of A7 to A8 involves a side chain hydrolysis reaction under alkaline conditions. Critical parameters include maintaining a reaction temperature between 40-60°C for 3-5 hours using solvents like methanol or ethanol, and bases such as sodium hydroxide or potassium hydroxide.
Q: How is stereochemical purity controlled during the synthesis of intermediate A7?
A: Stereochemical purity, specifically the 5α-H isomer ratio, is controlled during the hydrogenation of A6 to A7. Using a solvent system of absolute ethanol and pyridine, potentially with additives like DMAP or 1-methylimidazole, alongside palladium on carbon catalyst at 30-35°C, significantly enhances the purity of the desired isomer.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cholic Acid Intermediate A8 Supplier
At NINGBO INNO PHARMCHEM, we understand the critical importance of securing a stable and high-quality supply of complex pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the clinical trial phase or full-scale commercialization. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this innovative synthetic technology for your cholic acid projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in fine chemical synthesis can enhance your supply chain efficiency and product quality.
