Advanced Iron-Catalyzed Oxidation for High-Purity Aromatic Benzyl Ketones
Introduction to Green Oxidation Technology
The synthesis of aromatic benzyl ketones represents a critical transformation in the production of fine chemicals and pharmaceutical intermediates, yet traditional methodologies have long been plagued by environmental and safety concerns. Patent CN111606791A introduces a groundbreaking synthetic method that addresses these challenges by employing a novel catalytic system composed of ferric ions, nitrate radicals, and N-hydroxyimide derivatives. This technology enables the efficient oxidation of aromatic benzyl compounds using molecular oxygen or air as the primary oxidant, marking a significant departure from hazardous stoichiometric oxidants. By leveraging the synergistic effects of iron and organic radical initiators, this process achieves high atom utilization rates while completely avoiding the use of toxic heavy metals such as copper and cobalt. For industry leaders seeking a reliable pharmaceutical intermediate supplier, this patent offers a pathway to greener, more sustainable manufacturing protocols that align with modern regulatory standards.
The core innovation lies in the specific molar ratios and interactions within the catalytic triad, where ferric ions act in concert with nitrate anions to promote the generation of active radical species from the N-hydroxyimide derivatives. This mechanism facilitates the selective oxidation of benzylic methylene groups to carbonyl functionalities under remarkably mild conditions, typically ranging from 60°C to 150°C. The versatility of this system is evidenced by its applicability to a wide range of substrates, including simple alkylbenzenes and complex polycyclic aromatic hydrocarbons. As we delve deeper into the technical specifics, it becomes clear that this methodology not only enhances chemical efficiency but also provides substantial strategic advantages for supply chain optimization and cost management in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the auto-oxidation of methylene or methyl compounds has relied heavily on transition metal catalysts such as cobalt, manganese, or cadmium, often in conjunction with bulky biomimetic ligands. While effective in certain contexts, these conventional systems suffer from significant drawbacks that hinder their application in high-value industries like pharmaceuticals. The use of cobalt and manganese frequently results in deep-colored reaction mixtures and, more critically, the formation of stable complexes between residual metal ions and the aromatic rings of the product. This contamination necessitates rigorous and costly purification steps to meet safety standards, as heavy metal residues are strictly regulated in drug substances. Furthermore, prior art such as CN108238946A highlights that some metal-free approaches require excessively high pressures and large excesses of alkali, introducing safety hazards and generating substantial chemical waste. Other methods utilizing TEMPO and halides increase post-treatment complexity and wastewater pollution, making them less attractive for large-scale commercial adoption.
The Novel Approach
In stark contrast, the method disclosed in CN111606791A utilizes a catalytic system based on ferric ions, nitrates, and N-hydroxyimide derivatives (NHIs), which eliminates the need for harmful heavy metals entirely. This approach leverages the redox properties of iron, a biologically benign metal, combined with the radical-generating capability of NHIs to drive the oxidation efficiently. The reaction proceeds smoothly under atmospheric pressure using oxygen or air, significantly reducing equipment costs and operational risks associated with high-pressure reactors. As illustrated in the general reaction scheme below, the transformation of ethylbenzene to acetophenone serves as a prime example of this efficiency, achieving high conversion rates with minimal environmental impact.
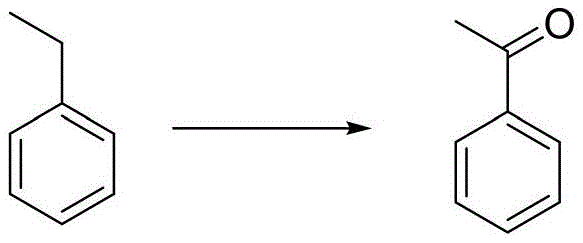
The novel approach also demonstrates superior atom economy, as the oxidant is molecular oxygen, producing water as the primary byproduct rather than toxic salt waste. The flexibility of the solvent system, which accommodates polar organic solvents like acetic acid and benzonitrile, allows for fine-tuning of reaction kinetics to suit different substrate profiles. This adaptability ensures that the process remains robust across a diverse library of aromatic benzyl compounds, providing a universal solution for ketone synthesis that overcomes the selectivity and purity limitations of legacy technologies.
Mechanistic Insights into Fe(III)/Nitrate/NHI Catalytic Synergy
The efficacy of this synthetic method is rooted in the intricate interplay between the three components of the catalytic system: ferric ions, nitrate radicals, and N-hydroxyimide derivatives. The N-hydroxyimide derivatives, such as N-hydroxyphthalimide (NHPI) and its analogs shown in the structural diagram below, function as potent radical initiators. Under the influence of the metal catalyst, these derivatives generate phthalimide-N-oxyl (PINO) radicals, which are responsible for abstracting hydrogen atoms from the benzylic position of the substrate. This hydrogen abstraction is the rate-determining step that initiates the auto-oxidation chain reaction, converting the benzylic methylene group into a carbon-centered radical that subsequently reacts with oxygen.
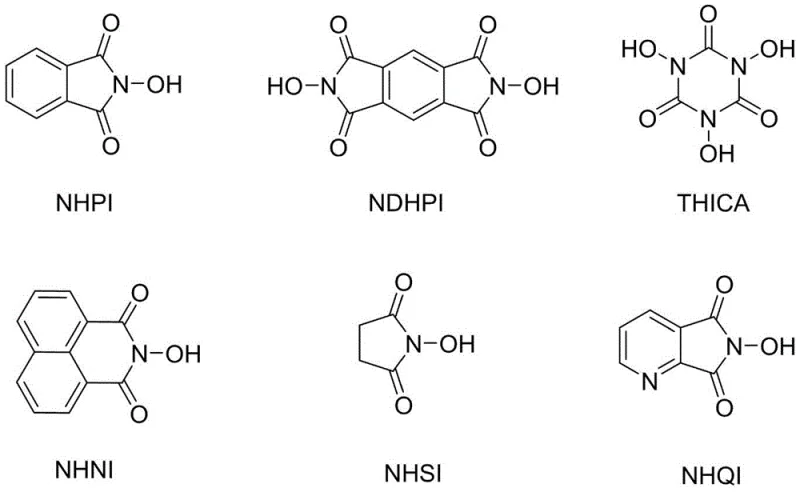
Crucially, the presence of nitrate ions acts as a co-catalyst that significantly enhances the generation of these active radicals, thereby allowing for a reduction in the overall loading of the metal catalyst. The ferric ions facilitate the redox cycling necessary to regenerate the active catalytic species, ensuring the reaction proceeds to completion without the accumulation of inactive byproducts. This synergistic effect means that the system can operate with lower metal concentrations compared to traditional cobalt-based systems, directly translating to reduced metal contamination in the final product. For R&D directors focused on impurity profiles, this mechanism offers a distinct advantage by minimizing the risk of metal-aromatic complexation, which is a common source of difficult-to-remove impurities in conventional oxidation processes.
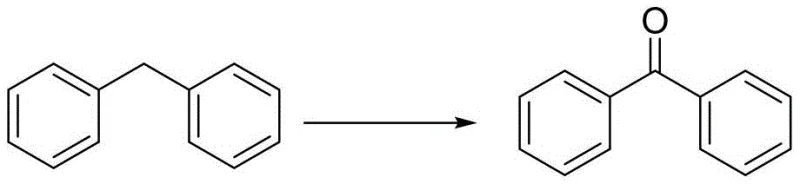
Furthermore, the reaction conditions are optimized to balance reactivity and selectivity. Operating temperatures between 60°C and 150°C, with an optimum around 100°C, provide sufficient thermal energy to sustain the radical chain without promoting excessive over-oxidation or degradation of sensitive functional groups. The use of polar solvents like benzonitrile not only dissolves the reactants effectively but also stabilizes the polar transition states involved in the oxidation pathway. This precise control over the reaction environment ensures high yields and purity, as evidenced by the successful conversion of various substrates including diphenylmethane to benzophenone with nearly quantitative yields.
How to Synthesize Aromatic Benzyl Ketones Efficiently
Implementing this advanced oxidation protocol requires careful attention to the preparation of the catalytic mixture and the control of reaction parameters. The process begins with the selection of appropriate N-hydroxyimide derivatives, with NHSI, NHPI, or NHNI being preferred for their balance of reactivity and stability. The molar ratio of ferric ions to nitrate is maintained between 1:0.5 and 1:5, while the ratio of ferric ions to the N-hydroxyimide derivative is kept within the range of 0.01-0.1 to 0.03-0.2 to ensure optimal radical flux. Detailed standardized synthesis steps for specific substrates are provided in the guide below, outlining the precise quantities and conditions required to replicate the high efficiencies reported in the patent data.
- Prepare the catalytic system by mixing ferric nitrate and N-hydroxyimide derivatives (such as NHPI or NHSI) in a polar organic solvent like acetic acid or benzonitrile.
- Add the aromatic benzyl substrate to the reaction vessel and replace the atmosphere with oxygen or air, maintaining a temperature between 60°C and 150°C.
- Monitor the reaction progress via gas chromatography until conversion is complete, then isolate the product through solvent evaporation and purification methods like recrystallization or column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed oxidation technology presents a compelling value proposition centered on cost reduction and operational reliability. By eliminating the need for expensive and toxic heavy metal catalysts like cobalt and cadmium, manufacturers can significantly reduce raw material costs and avoid the substantial expenses associated with hazardous waste disposal and environmental compliance. The simplified workup procedure, which often involves simple solvent evaporation followed by recrystallization or distillation, reduces processing time and energy consumption. This streamlined workflow enhances overall throughput, allowing facilities to respond more agilely to market demands without the bottlenecks typically caused by complex purification stages required for metal-contaminated products.
- Cost Reduction in Manufacturing: The substitution of precious or toxic metal catalysts with inexpensive ferric nitrate and recyclable organic initiators drives down the direct cost of goods sold. Additionally, the ability to recover and reuse the N-hydroxyimide catalyst, as demonstrated in the recycling experiments, further amplifies these savings over multiple production batches. The avoidance of high-pressure equipment and the use of ambient air or oxygen as the oxidant also lower capital expenditure and utility costs, making the process economically superior to traditional auto-oxidation methods that require specialized infrastructure.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, including iron salts and common N-hydroxyimides, are commercially available in bulk quantities from established chemical suppliers, ensuring a stable and secure supply chain. Unlike proprietary ligand systems that may be subject to supply constraints or intellectual property restrictions, the components of this catalytic system are commoditized, reducing the risk of production delays due to material shortages. This reliability is crucial for maintaining continuous manufacturing operations and meeting strict delivery schedules for downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The mild reaction conditions and atmospheric pressure operation make this technology highly scalable from laboratory benchtop to industrial production volumes. The green chemistry profile of the process, characterized by low toxicity and minimal waste generation, aligns perfectly with increasingly stringent global environmental regulations. This compliance reduces the regulatory burden on manufacturing sites and enhances the corporate sustainability profile, which is becoming a key differentiator in securing contracts with environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidation technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on performance metrics and operational feasibility. Understanding these details is essential for technical teams evaluating the integration of this method into existing production lines.
Q: How does this iron-catalyzed method improve product purity compared to traditional cobalt systems?
A: Traditional methods often utilize cobalt or manganese catalysts which can form stable complexes with aromatic rings, leading to difficult-to-remove heavy metal residues. The patented Fe(III)/NHPI system avoids harmful heavy metals entirely, significantly simplifying downstream purification and ensuring the final aromatic benzyl ketones meet stringent pharmaceutical purity specifications without extensive metal scavenging steps.
Q: Is the N-hydroxyimide catalyst recyclable in this process?
A: Yes, the patent demonstrates that specific N-hydroxyimide derivatives, such as NHSI, can be recovered and reused. In experimental examples involving 4-nitroethylbenzene oxidation, the catalyst system maintained yields above 80% over multiple consecutive cycles, indicating robust catalyst stability and potential for substantial cost reduction in large-scale manufacturing.
Q: What are the safety advantages of using oxygen versus other oxidants in this synthesis?
A: This method utilizes molecular oxygen or air as the terminal oxidant, which is inherently safer and more economical than stoichiometric chemical oxidants that generate hazardous waste. The reaction operates effectively at atmospheric pressure and moderate temperatures (optimally around 100°C), eliminating the need for high-pressure equipment required by some alternative auto-oxidation processes, thereby enhancing operational safety and reducing capital expenditure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Benzyl Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Fe(III)/NHPI catalytic system for the production of high-quality aromatic benzyl ketones. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical manufacturing settings. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of intermediate meets the exacting standards required by the global pharmaceutical industry. We are committed to delivering consistent quality and supply continuity for our partners.
We invite you to collaborate with us to leverage this advanced synthetic route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this green chemistry approach can optimize your budget. Please contact us to request specific COA data and route feasibility assessments, and let us help you accelerate your development timeline with a reliable and efficient supply of critical chemical intermediates.
