Revolutionizing Glycal Production: A Safe, Metal-Free Phosphine Elimination Strategy for Commercial Scale
Revolutionizing Glycal Production: A Safe, Metal-Free Phosphine Elimination Strategy for Commercial Scale
The landscape of carbohydrate chemistry is undergoing a significant transformation with the disclosure of patent CN114456217A, which introduces a groundbreaking synthetic method for glycal compounds. Glycals serve as pivotal building blocks in the construction of complex oligosaccharides, C-glycosides, and various bioactive natural products, yet their production has historically been plagued by hazardous reagents and苛刻 conditions. This new technology leverages a triaryl or trialkyl phosphine-mediated reductive elimination strategy, effectively bypassing the need for toxic heavy metals like zinc or titanium. By shifting the paradigm from metal-based reduction to phosphine-driven elimination, this innovation offers a robust pathway for producing high-purity glycal compounds with exceptional operational simplicity and environmental compatibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
For over a century, the industry has relied heavily on the Ferrier-Zach reduction method, a process that, while effective, imposes severe constraints on manufacturing efficiency and safety. As illustrated in the reaction scheme below, this traditional approach necessitates the use of nearly ten equivalents of zinc powder in conjunction with acidic additives, often requiring cryogenic conditions ranging from -20°C to 0°C to control selectivity.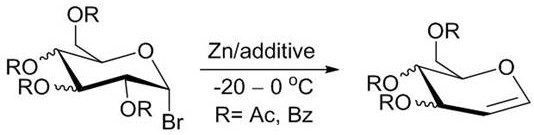
Furthermore, alternative methods utilizing Titanium (III) complexes, such as Cp2TiCl, introduce additional layers of complexity due to the high cost and toxicity of the organometallic reagents involved. These legacy processes generate substantial amounts of heavy metal waste, complicating downstream purification and creating significant environmental liabilities. The requirement for strict inert gas environments and the inherent instability of 1-halo sugar intermediates further exacerbate the operational risks, making these methods ill-suited for modern, sustainable commercial scale-up of complex carbohydrate derivatives.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a mild, two-step sequence initiated by the reaction of protected sugars with trisubstituted phosphines. This approach eliminates the dependency on unstable halogenated precursors and hazardous reducing agents, instead forming a stable phosphonium salt intermediate under ambient or slightly elevated temperatures. The subsequent base-mediated hydrolysis triggers a clean elimination reaction, yielding the target alkene functionality with high stereocontrol. This shift not only drastically simplifies the reactor setup by removing the need for cryogenic cooling but also enhances the overall atom economy of the process. By employing readily available organic solvents and common bases, this method represents a reliable glycal compound supplier strategy that aligns perfectly with green chemistry principles and cost-effective manufacturing goals.
Mechanistic Insights into Phosphine-Mediated Reductive Elimination
The core of this technological breakthrough lies in the unique reactivity of the phosphonium salt intermediate, which serves as a superior leaving group compared to traditional halides. The reaction initiates with the nucleophilic attack of the triaryl or trialkyl phosphine on the anomeric center of the acyl-protected sugar, displacing the leaving group to form the cationic species. This intermediate is remarkably stable, allowing for flexible reaction times between 2 to 36 hours at temperatures ranging from 20°C to 60°C, providing a wide operational window for process optimization. The subsequent addition of a base facilitates the deprotonation at the C2 position, driving the elimination of the phosphine oxide and establishing the characteristic double bond of the glycal structure.
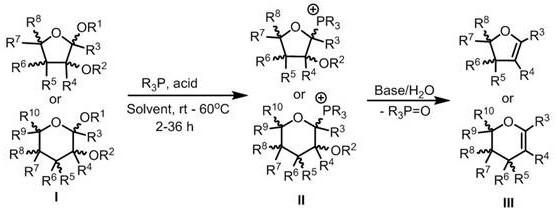
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed routes. The absence of transition metals eliminates the risk of metal-catalyzed side reactions, such as over-reduction or isomerization, which often plague zinc-mediated processes. Furthermore, the mild alkaline conditions used for hydrolysis are compatible with a broad range of protecting groups, including acyl, silyl, and benzyl moieties, ensuring the integrity of the carbohydrate scaffold is maintained throughout the synthesis. This high level of chemoselectivity results in cleaner reaction profiles, reducing the burden on purification teams and ensuring the delivery of high-purity glycal compounds essential for sensitive pharmaceutical applications.
How to Synthesize Glycal Compounds Efficiently
The synthesis protocol outlined in the patent provides a straightforward roadmap for laboratory and pilot-scale production, emphasizing ease of execution and reproducibility. The process begins with the dissolution of the protected sugar substrate and the phosphine reagent in a common organic solvent such as dichloromethane or acetonitrile, followed by heating to facilitate salt formation. Once the intermediate is confirmed via TLC, the reaction mixture is treated with an aqueous base to induce elimination, followed by a standard extractive workup. For detailed procedural specifics regarding stoichiometry, solvent volumes, and purification parameters, please refer to the standardized synthesis guide below.
- Dissolve acyl-protected sugar and triaryl phosphine in an organic solvent like dichloromethane at room temperature.
- Heat the mixture to 20-60°C for 2-36 hours to form the phosphonium salt intermediate.
- Hydrolyze the intermediate under alkaline conditions, extract, and purify via silica gel chromatography to obtain the target glycal.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this phosphine-mediated synthesis route translates into tangible strategic benefits regarding cost stability and operational resilience. The replacement of expensive and volatile metal reagents with commodity chemicals like triphenylphosphine significantly lowers the raw material cost baseline, insulating the supply chain from fluctuations in the precious metals market. Moreover, the simplified reaction conditions reduce the energy consumption associated with cryogenic cooling and inert gas blanketing, contributing to substantial cost savings in utility expenditures. This economic efficiency is compounded by the reduced need for specialized waste treatment facilities, as the process generates non-toxic phosphine oxide byproducts rather than heavy metal sludge.
- Cost Reduction in Manufacturing: The elimination of stoichiometric zinc powder and expensive titanium catalysts removes a major cost driver from the bill of materials. Additionally, the ability to run reactions at near-ambient temperatures reduces energy costs associated with refrigeration, while the simplified workup minimizes solvent usage and labor hours required for purification. These factors collectively drive down the cost of goods sold (COGS), enabling more competitive pricing for downstream pharmaceutical intermediates without compromising on quality or yield.
- Enhanced Supply Chain Reliability: By relying on widely available organic reagents rather than specialized organometallic complexes, the risk of supply disruption is markedly decreased. The robustness of the reaction conditions allows for greater flexibility in scheduling and batch sizing, ensuring consistent output even during periods of high demand. This reliability is crucial for maintaining continuous production lines for API intermediates, preventing costly downtime and ensuring that critical drug development timelines are met without delay.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated on substrates ranging from simple monosaccharides to complex disaccharides and trisaccharides with consistently high yields. The absence of heavy metals simplifies regulatory compliance and environmental reporting, facilitating smoother audits and faster approval for commercial manufacturing sites. This alignment with stringent environmental standards future-proofs the supply chain against tightening global regulations on industrial emissions and waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel glycal synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, aiming to clarify the practical implications for industrial adoption. Understanding these nuances is essential for R&D teams evaluating process transfer and procurement officers assessing vendor capabilities.
Q: What are the primary advantages of this phosphine-mediated method over traditional zinc reduction?
A: Unlike the traditional Ferrier-Zach method which requires hazardous zinc powder, low temperatures (-20°C), and inert atmospheres, this new method operates at mild temperatures (20-60°C) without heavy metals, significantly simplifying safety protocols and waste treatment.
Q: Can this method be used to synthesize deuterated glycal compounds?
A: Yes, the patent explicitly describes that by using deuterated sodium hydroxide and deuterated water during the alkaline hydrolysis step, corresponding 1-D-glycal compounds can be successfully synthesized, which is valuable for metabolic studies.
Q: Is this process suitable for large-scale industrial production?
A: Absolutely. The process utilizes cheap and readily available raw materials, avoids expensive transition metal catalysts, and features a simple workup procedure involving standard extraction and chromatography, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glycal Compound Supplier
As the demand for complex carbohydrate derivatives continues to surge in the pharmaceutical and biotechnology sectors, partnering with an experienced CDMO is critical for successful commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to manufacturing plant is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of glycal compound delivered meets the highest international standards for safety and efficacy.
We invite you to engage with our technical procurement team to discuss how this innovative phosphine-mediated route can be integrated into your specific supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your next project benefits from the latest advancements in sustainable organic synthesis.
