Advanced Chiral Resolution Strategy for Commercial Scale-up of Complex Oxazacycloheptane Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways for complex heterocyclic scaffolds, particularly those containing chiral centers essential for biological activity. Patent CN112479876A introduces a groundbreaking methodology for the preparation of oxazacycloheptane spiro-compounds, a class of nitrogen-containing heterocycles increasingly relevant in modern drug discovery. This invention specifically addresses the challenges associated with stereoselectivity and process efficiency by employing a novel chiral resolution strategy on a key hydroxy-acid intermediate. Unlike traditional approaches that often suffer from poor stereocontrol and excessive step counts, this patented route leverages the formation of diastereomeric salts using chiral amines such as (R)- or (S)-1-(1-naphthyl)ethylamine. This strategic intervention allows for the isolation of high-purity enantiomers with ee values reaching up to 100%, thereby establishing a reliable foundation for the subsequent construction of the spiro-oxazepane core. For R&D directors and procurement specialists, this represents a significant advancement in accessing high-purity pharmaceutical intermediates with improved supply chain stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthetic routes, such as the one disclosed in patent application WO2016033486a1, exemplify the inefficiencies plaguing traditional methods for constructing these complex spiro-systems. The conventional pathway typically initiates with 6-chloro-3,4-dihydro-1(2H)-naphthalenone, requiring a cumbersome sequence involving epoxidation with trimethyl sulfonium iodide followed by Lewis acid-mediated rearrangement to generate an aldehyde. This is subsequently converted to a diol and subjected to mono-substitution with 4-bromobenzoyl chloride. The route further necessitates the use of expensive and hazardous oxidants like Dess-Martin periodinane to convert alcoholic hydroxyl groups into aldehydes, alongside multiple protection and deprotection steps using p-toluenesulfonic acid and trimethyl orthoformate. Such a lengthy synthetic trajectory not only results in low overall yields but also demands rigorous purification via column chromatography at multiple stages, creating substantial bottlenecks for commercial manufacturing.
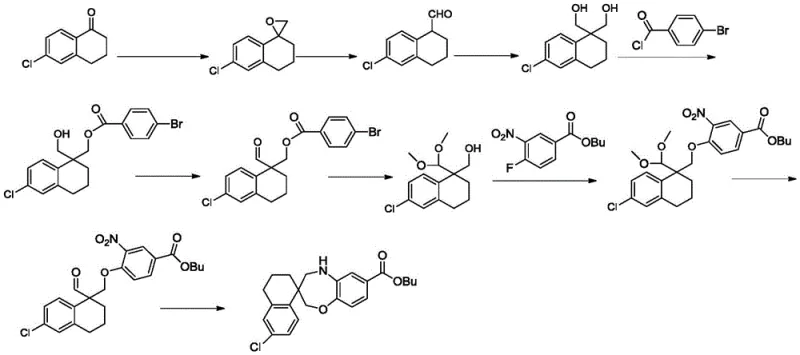
The Novel Approach
In stark contrast, the methodology presented in CN112479876A streamlines the synthesis by introducing a highly efficient chiral resolution step early in the sequence. The process begins with a palladium-catalyzed coupling reaction to establish the carbon skeleton, followed by intramolecular cyclization to form the tetralone core. Crucially, the subsequent reduction yields a hydroxy-acid intermediate which serves as the pivot point for stereochemical control. By reacting this racemic mixture with a chiral resolving agent in an organic solvent such as ethyl acetate or isopropanol, the desired enantiomer precipitates as a salt, allowing for simple filtration and recrystallization. This approach effectively bypasses the need for chiral chromatography or asymmetric catalysis in later stages, drastically simplifying the workflow. The resulting chiral intermediate is then elaborated through etherification and reductive amination to close the oxazepane ring, offering a direct and scalable path to the target spiro-compounds.
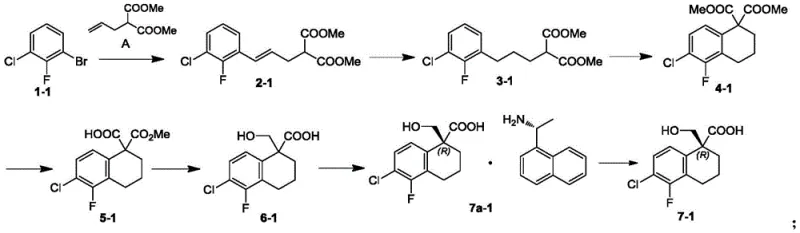
Mechanistic Insights into Palladium-Catalyzed Coupling and Chiral Resolution
The initial construction of the molecular framework relies on a sophisticated palladium-catalyzed coupling reaction between an aryl bromide and an unsaturated diester. In the presence of a ligand such as tris(o-tolyl)phosphine and a base like dicyclohexylmethylamine, the palladium catalyst facilitates the formation of a carbon-carbon bond under inert atmosphere conditions at elevated temperatures around 110°C. This step is critical for establishing the side chain required for subsequent cyclization. Following hydrogenation to saturate the double bond, the molecule undergoes an acid-mediated intramolecular cyclization in the presence of oxidants like manganese acetate or copper acetate. This oxidative cyclization forms the fused ring system essential for the spiro-center. The mechanistic elegance lies in the precise control of reaction conditions, such as maintaining a temperature of 95°C and utilizing acetic acid as a solvent, which ensures high conversion rates while minimizing side reactions like dehalogenation.
Following the formation of the core structure, the stereochemical integrity is secured through a classical yet highly effective resolution process. The hydroxy-acid intermediate reacts with a chiral amine resolving agent, forming diastereomeric salts that exhibit different solubilities in the chosen organic solvent. For instance, using (R)-1-(1-naphthyl)ethylamine allows for the selective precipitation of the (R)-configured salt, which can be isolated via suction filtration. The mother liquor retains the unwanted enantiomer, maximizing atom economy. After isolation, the salt is neutralized with acid to liberate the free chiral acid. This mechanism ensures that the downstream etherification and reductive amination steps proceed with a single enantiomer, guaranteeing the final product meets stringent purity specifications without the need for complex chiral separations at the final stage.
How to Synthesize Oxazacycloheptane Spiro-Compounds Efficiently
The synthesis of these valuable intermediates involves a sequence of well-defined chemical transformations starting from readily available aryl bromides. The process integrates transition metal catalysis for bond formation, followed by selective functional group manipulations to install the necessary oxygen and nitrogen heteroatoms. The key to success lies in the meticulous execution of the chiral resolution step, where parameters such as solvent choice, molar ratios of the resolving agent, and crystallization temperatures must be strictly controlled to achieve optimal optical purity. Detailed standardized synthetic steps see the guide below.
- Perform palladium-catalyzed coupling of aryl bromide with unsaturated diester, followed by hydrogenation and acid-mediated intramolecular cyclization to form the tetralone core.
- Execute selective reduction of the ester group to a hydroxy-acid, followed by chiral resolution using (R)- or (S)-1-(1-naphthyl)ethylamine to isolate the desired enantiomer.
- Conduct etherification with nitrobenzoate, reduce the nitro group, and finalize the structure via intramolecular reductive amination to close the oxazepane ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers transformative benefits regarding cost structure and operational reliability. The primary advantage stems from the drastic simplification of the process flow, which eliminates several high-cost unit operations present in legacy methods. By replacing expensive stoichiometric oxidants and avoiding repetitive column chromatography purifications, the manufacturing process becomes significantly more lean. This reduction in processing complexity translates directly into lower production costs and reduced waste generation, aligning with modern green chemistry principles. Furthermore, the reliance on commodity chemicals and standard catalysts ensures that raw material sourcing remains stable and unaffected by niche supply constraints.
- Cost Reduction in Manufacturing: The elimination of costly reagents like Dess-Martin periodinane and the reduction in solvent consumption due to fewer purification steps lead to substantial cost savings. Additionally, the ability to recover and recycle the chiral resolving agent through neutralization and extraction further enhances the economic viability of the process. These factors collectively contribute to a more competitive pricing structure for the final intermediate, allowing pharmaceutical companies to optimize their COGS without compromising on quality.
- Enhanced Supply Chain Reliability: The use of robust, scalable reactions such as palladium-catalyzed coupling and catalytic hydrogenation ensures that the process can be easily transferred from laboratory to pilot and commercial scales. The avoidance of sensitive intermediates that require cryogenic conditions or specialized handling reduces the risk of batch failures. This reliability is crucial for maintaining continuous supply lines for API production, minimizing the risk of stockouts and ensuring timely delivery to downstream customers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents like toluene, ethyl acetate, and isopropanol which are widely accepted in large-scale manufacturing. The simplified workup procedures, primarily involving filtration and extraction rather than chromatography, reduce the volume of hazardous waste generated. This not only lowers disposal costs but also facilitates compliance with increasingly stringent environmental regulations, making the process sustainable for long-term commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of these oxazacycloheptane derivatives. The answers are derived directly from the experimental data and process descriptions provided in the patent literature, ensuring accuracy and relevance for technical decision-makers.
Q: How does the new chiral resolution method improve upon previous synthetic routes?
A: The new method described in CN112479876A utilizes an early-stage chiral resolution on a hydroxy-acid intermediate using naphthyl-ethylamine, achieving ee values up to 100%. This avoids the lengthy, low-yield sequences involving epoxy formation and multiple protection/deprotection steps found in prior art like WO2016033486a1.
Q: What are the key cost drivers eliminated in this manufacturing process?
A: The process eliminates the need for expensive oxidants like Dess-Martin periodinane and reduces reliance on extensive column chromatography purification. By utilizing recrystallization for chiral separation and standard catalytic hydrogenation, the overall operational expenditure is significantly lowered.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the route employs robust reactions such as Pd-catalyzed coupling and metal-mediated reductions which are well-suited for scale-up. The use of common solvents like toluene, ethyl acetate, and isopropanol further enhances the feasibility of commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxazacycloheptane Spiro-Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation therapeutics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative routes described in CN112479876A can be seamlessly implemented at an industrial level. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex chiral resolutions and multi-step syntheses positions us as a strategic partner for your drug development programs.
We invite you to collaborate with us to leverage these advanced synthetic technologies for your specific projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your timeline to market while optimizing your manufacturing budget.
