Advanced Palladium-Catalyzed Synthesis of 1,1-Diaryl-2-Alkyl Ethylene Derivatives for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 1,1-Diaryl-2-Alkyl Ethylene Derivatives for Commercial Scale-Up
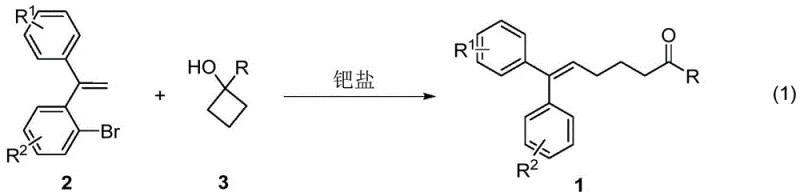
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex polysubstituted olefin scaffolds, which serve as critical structural motifs in numerous bioactive molecules. A groundbreaking approach detailed in patent CN112209812B introduces a highly efficient synthetic route for preparing 1,1-diaryl-2-alkyl ethylene derivatives. This technology leverages a palladium-catalyzed ring-opening coupling reaction between readily available o-bromostilbenes and cyclobutanols. By addressing the longstanding challenges of stereoselectivity and substrate scope associated with traditional olefin synthesis, this innovation offers a transformative pathway for the production of high-value intermediates used in drug discovery and development. The method stands out for its operational simplicity, mild reaction conditions, and exceptional atom economy, positioning it as a superior alternative for manufacturers aiming to optimize their supply chains for complex organic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the stereoselective synthesis of polysubstituted olefins has been fraught with significant technical hurdles that impede efficient large-scale manufacturing. Traditional strategies often rely on controlling stereoselectivity through steric effects, a method that frequently fails when two similar groups are introduced at one end of the olefin, leading to mixtures of isomers that are difficult and costly to separate. Another common approach involves the use of directing groups to guide the reaction; however, this necessitates the presence of specific functional handles on the substrate, thereby limiting the diversity of molecules that can be synthesized and adding extra synthetic steps for installation and removal of these groups. Furthermore, existing reports on 1,4-palladium migration strategies have largely been restricted to C-H boronation or alkenylation, leaving a critical gap in methodologies for direct C-H alkylation of olefins. These limitations result in lower overall yields, increased waste generation, and higher production costs, creating a pressing need for more versatile and direct synthetic solutions in the field of pharmaceutical intermediates.
The Novel Approach
The methodology disclosed in patent CN112209812B overcomes these barriers by employing a direct ring-opening coupling reaction that constructs the C-C bond with remarkable precision. As illustrated in the general reaction scheme below, the process utilizes o-bromostilbene and cyclobutanol as starting materials, which undergo a palladium-catalyzed transformation to yield the target 1,1-diaryl-2-alkyl ethylene derivative in a stereospecific manner. This novel approach eliminates the need for pre-functionalized directing groups or reliance on subtle steric differences, thereby vastly expanding the scope of accessible chemical space. The reaction proceeds under mild thermal conditions using standard laboratory equipment, and the catalyst system is robust enough to tolerate a wide array of functional groups including halogens, ethers, and esters. This represents a paradigm shift in olefin alkylation, providing a reliable pharmaceutical intermediates supplier with a tool to access complex structures that were previously difficult to obtain efficiently.
Mechanistic Insights into Pd-Catalyzed Ring-Opening Coupling
The success of this synthetic route lies in the intricate interplay between the palladium catalyst and the unique reactivity of the cyclobutanol ring. The mechanism likely initiates with the oxidative addition of the palladium(0) species into the carbon-bromine bond of the o-bromostilbene substrate, forming an aryl-palladium(II) intermediate. This is followed by the coordination and subsequent insertion of the cyclobutanol moiety. A key step in this catalytic cycle is the beta-carbon elimination, which drives the ring-opening of the strained four-membered cyclobutane ring. This ring-opening event is thermodynamically favorable due to the release of ring strain and results in the formation of a new carbon-carbon bond while establishing the trisubstituted olefin geometry. The presence of specific additives, such as cesium carbonate and o-fluorophenol, plays a crucial role in facilitating proton transfer and stabilizing the catalytic species, ensuring high turnover numbers and minimizing side reactions. This mechanistic understanding allows for fine-tuning of reaction parameters to maximize yield and purity, which is essential for meeting the stringent quality standards required in API manufacturing.
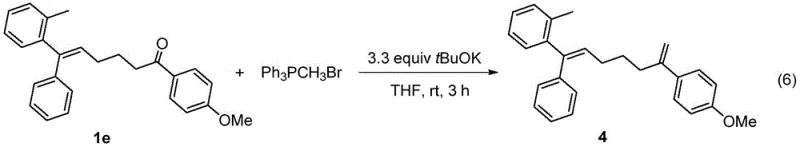
Beyond the primary coupling reaction, the utility of the resulting 1,1-diaryl-2-alkyl ethylene derivatives is further enhanced by their potential for downstream functionalization. The patent data demonstrates that the ketone functionality present in the initial products can be subjected to further transformations, such as Wittig olefination, to generate even more complex architectures containing terminal alkenes. For instance, the conversion of compound 1e into compound 4 via reaction with a phosphonium salt highlights the versatility of this scaffold. This ability to serve as a divergent intermediate means that a single synthetic platform can feed into multiple drug discovery pipelines, significantly increasing the value proposition for research and development teams. The preservation of stereochemistry throughout these transformations ensures that the biological activity of the final molecules remains consistent, a critical factor in the development of chiral drugs.
How to Synthesize 1,1-Diaryl-2-Alkyl Ethylene Efficiently
The practical implementation of this chemistry is straightforward and amenable to standard organic synthesis protocols. The process typically involves combining the o-bromostilbene substrate with the cyclobutanol coupling partner in the presence of a palladium catalyst system, specifically PdCl2 and tricyclohexylphosphine, along with cesium carbonate and o-fluorophenol as additives. The reaction is conducted in toluene at elevated temperatures, typically around 110 °C, for a duration of approximately 24 hours. Upon completion, the workup procedure is remarkably simple, often requiring only direct silica gel column chromatography to isolate the pure product without the need for extensive aqueous extractions or recrystallizations. This streamlined workflow reduces solvent consumption and labor time, contributing to overall process efficiency. For detailed standardized operating procedures and safety guidelines regarding this specific synthesis, please refer to the technical guide below.
- In a glove box, weigh o-bromostilbene (0.3 mmol), PdCl2 (0.015 mmol), tricyclohexylphosphine (0.018 mmol), cyclobutanol (0.45 mmol), cesium carbonate (0.6 mmol), and o-fluorophenol (0.6 mmol) into a 25 mL branched tube.
- Add toluene (3 mL) under a nitrogen atmosphere and heat the reaction mixture to 110 °C for 24 hours to facilitate the ring-opening coupling reaction.
- Upon completion, cool to room temperature and purify the crude mixture directly via silica gel column chromatography using petroleum ether/ethyl acetate (20: 1 v/v) to isolate the target product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this palladium-catalyzed ring-opening coupling technology offers substantial benefits for procurement managers and supply chain directors focused on cost reduction in API manufacturing. The primary advantage stems from the use of easily obtainable and inexpensive raw materials; o-bromostilbenes and cyclobutanols are commercially available or can be synthesized via established routes, ensuring a stable supply base. Furthermore, the reaction does not require the addition of external oxidants or reductants, which simplifies the reagent inventory and reduces the hazard profile of the manufacturing process. The high atom economy of the ring-opening strategy means that a larger proportion of the starting material mass is incorporated into the final product, minimizing waste disposal costs and aligning with green chemistry principles. These factors collectively contribute to a more sustainable and economically viable production model for high-purity polysubstituted olefins.
- Cost Reduction in Manufacturing: The elimination of complex protecting group strategies and the use of a robust catalyst system significantly lower the operational expenditure associated with synthesis. By avoiding the need for harsh reaction conditions or expensive specialized reagents, manufacturers can achieve substantial cost savings per kilogram of product. The high yields reported in the patent examples, often exceeding 90%, further enhance the economic feasibility by reducing the amount of starting material required to meet production targets. Additionally, the simplified purification process reduces the consumption of chromatography media and solvents, which are often major cost drivers in fine chemical production.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent product quality across different batches, which is vital for maintaining supply continuity. The tolerance of the catalyst system to various functional groups allows for flexibility in sourcing raw materials, as slight variations in substrate purity or substitution patterns do not necessarily compromise the reaction outcome. This resilience mitigates the risk of production delays caused by raw material shortages or quality issues. Moreover, the scalability of the process from milligram to multi-kilogram scales has been demonstrated, providing confidence that supply demands can be met as clinical programs advance from early-stage discovery to commercial launch.
- Scalability and Environmental Compliance: The use of toluene as a solvent and the absence of toxic heavy metal oxidants simplify the environmental compliance landscape. Waste streams are easier to treat, and the overall E-factor of the process is improved compared to traditional multi-step sequences. The ability to run the reaction at moderate temperatures (110 °C) reduces energy consumption relative to processes requiring cryogenic conditions or extreme heating. These environmental advantages not only reduce regulatory burdens but also enhance the corporate social responsibility profile of the manufacturing operation, making it an attractive partner for global pharmaceutical companies committed to sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthetic methodology. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this chemistry into their existing workflows. The answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for process chemists and project managers.
Q: What is the stereoselectivity of this palladium-catalyzed olefin synthesis?
A: The method described in patent CN112209812B achieves stereospecific synthesis of polysubstituted olefins. Unlike traditional methods relying on steric effects or directing groups which have limitations, this ring-opening coupling strategy effectively controls the geometry of the double bond, producing specific 1,1-diaryl-2-alkyl ethylene derivatives with high fidelity.
Q: Can this process accommodate diverse functional groups on the substrate?
A: Yes, the process demonstrates excellent substrate tolerance. The o-bromostilbene starting material can bear various substituents such as methyl, methoxy, fluorine, chlorine, trifluoromethyl, and ethoxycarbonyl groups at different positions (R1 and R2). Similarly, the cyclobutanol component allows for variation in the alkyl chain (methyl, ethyl, n-hexyl, aryl), making it highly versatile for generating diverse chemical libraries.
Q: Is the catalyst system suitable for large-scale production?
A: The catalytic system utilizes commercially available palladium salts like PdCl2 and standard ligands like PCy3. The reaction operates under relatively mild conditions (110 °C) in common solvents like toluene without the need for exotic oxidants or reductants. These factors, combined with high yields (up to 95% in examples), indicate strong potential for commercial scale-up and cost-effective manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,1-Diaryl-2-Alkyl Ethylene Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to accelerate drug development timelines. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to industrial manufacturing is seamless. We are equipped with rigorous QC labs and adhere to stringent purity specifications to deliver high-purity polysubstituted olefins that meet the exacting standards of the global pharmaceutical industry. By leveraging the innovative palladium-catalyzed ring-opening coupling method, we can provide you with a competitive edge in synthesizing complex intermediates efficiently and reliably.
We invite you to collaborate with us to explore how this technology can optimize your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs and timeline constraints. Please contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis projects. Let us be your partner in turning complex chemical challenges into commercial successes.
