Advanced Cis-Selective Hydrogenation for High-Purity Sertraline Intermediates and Commercial Scalability
Advanced Cis-Selective Hydrogenation for High-Purity Sertraline Intermediates and Commercial Scalability
The pharmaceutical industry constantly seeks robust synthetic routes that maximize yield while minimizing stereochemical impurities, particularly for high-volume antidepressants like Sertraline. Patent CN1213991C introduces a groundbreaking process for the cis-selective catalytic hydrogenation of cyclohexylidenamines, addressing a critical bottleneck in the manufacturing of Sertraline-type cyclic amines. This technology leverages copper-containing catalysts in protic solvents to achieve unprecedented diastereoselectivity, fundamentally altering the economic and technical landscape for producing these vital active pharmaceutical ingredient (API) intermediates. By shifting away from traditional noble metal catalysts, this method offers a pathway to significantly reduce production costs and simplify purification workflows.
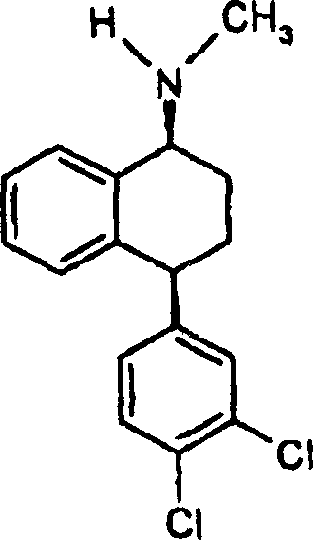
Sertraline, chemically known as (1S,4S)-4-(3,4-dichlorophenyl)-1,2,3,4-tetrahydro-N-methyl-1-naphthylamine, relies heavily on the specific spatial arrangement of its substituents for biological activity. The synthesis of the core tetrahydronaphthalene amine structure involves creating two chiral centers at the 1 and 4 positions. Historically, controlling the relative stereochemistry (cis vs. trans) has been a major challenge. The innovation described in CN1213991C provides a reliable sertraline intermediate supplier with the tools to produce the desired cis-isomer with exceptional purity, directly impacting the efficiency of the final drug substance manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
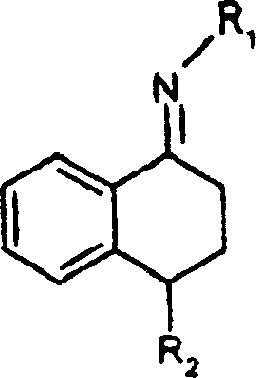
Traditional synthetic routes for generating the sertraline core have struggled with poor stereocontrol during the critical hydrogenation step. As documented in prior art such as US-A 4,536,518, the use of palladium on carbon (Pd/C) catalysts for hydrogenating the corresponding imine precursors typically results in a mixture containing only about 70% of the desired cis-racemate and 30% of the unwanted trans-racemate. This lack of selectivity creates a massive burden on downstream processing, requiring extensive and costly separation techniques to isolate the therapeutically relevant isomer. Furthermore, attempts to improve this ratio using Raney nickel, as proposed in WO 93/01161, only achieved a cis/trans ratio of approximately 8:1, which is still insufficient for streamlined commercial production without significant material loss.
The presence of substantial amounts of trans-isomers not only lowers the overall yield of the final API but also complicates the regulatory profile of the intermediate due to the need to strictly control impurity levels. In a commercial setting, dealing with a 30% impurity load at an early stage means larger reactors, more solvent consumption, and additional unit operations for purification, all of which drive up the cost of goods sold (COGS). For procurement managers and supply chain heads, these inefficiencies translate into longer lead times and higher vulnerability to supply disruptions, making the search for a more selective catalytic system a top priority for cost reduction in API manufacturing.
The Novel Approach
The process disclosed in CN1213991C represents a paradigm shift by utilizing copper-containing catalysts instead of noble metals. This novel approach involves the hydrogenation of cyclohexylidenamines (imines) or their precursors (nitrones) in the presence of a copper catalyst and a protic solvent. Experimental data from the patent demonstrates that this method consistently achieves cis/trans ratios exceeding 95:5, with optimized embodiments reaching an astonishing 99.6:0.4. This level of diastereoselectivity effectively eliminates the trans-isomer as a significant impurity, streamlining the entire production workflow. The use of copper catalysts, such as copper chromite or modified copper-zinc-aluminum oxides, provides a cost-effective alternative to expensive palladium or platinum systems while delivering superior performance.
Moreover, the reaction conditions are operationally simple and robust, typically conducted at temperatures between 90°C and 150°C and hydrogen pressures ranging from 10 to 20 bar. The compatibility with common protic solvents like ethanol, methanol, or isopropanol further enhances the green chemistry profile of the process. For a reliable agrochemical intermediate supplier or pharma partner, this translates to a process that is not only chemically superior but also easier to implement in existing infrastructure. The ability to generate the cis-isomer with such high fidelity means that subsequent resolution steps to obtain the single enantiomer (1S,4S) become much more efficient, as the starting material for resolution is already enriched in the correct diastereomer.
Mechanistic Insights into Copper-Catalyzed Diastereoselective Hydrogenation
The success of this copper-catalyzed system lies in its unique interaction with the substrate during the hydrogenation event. Unlike palladium, which often facilitates thermodynamic equilibration leading to mixtures, the copper surface appears to favor the kinetic formation of the cis-configuration. The mechanism likely involves the adsorption of the imine or nitrone onto the copper active sites in a specific orientation that directs the addition of hydrogen to the same face of the ring system. The presence of a protic solvent plays a crucial role, potentially stabilizing transition states or modifying the catalyst surface to enhance this selectivity. Understanding this mechanistic nuance is vital for R&D directors aiming to replicate or optimize the process for specific analogues within the sertraline family.
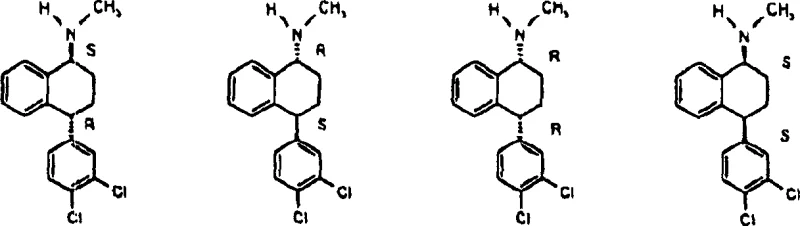
Furthermore, the process handles the complexity of chirality with elegance. As shown in the structural analysis, the molecule possesses two chiral centers, theoretically allowing for four stereoisomers: (1R,4R)-cis, (1S,4S)-cis, (1R,4S)-trans, and (1S,4R)-trans. The copper-catalyzed hydrogenation selectively produces the pair of cis-enantiomers ((1R,4R) and (1S,4S)) while suppressing the formation of the trans-pair. This diastereoselectivity is the key value driver. Even if the starting material is a racemic mixture, the reaction outputs a cis-racemate with minimal trans-contamination. This simplifies the subsequent chiral resolution, where agents like D-(-)-mandelic acid can be used to crystallize the desired (1S,4S) enantiomer from the cis-mixture with high efficiency, avoiding the difficult task of separating cis from trans isomers which often have similar physical properties.
Impurity control is another critical aspect where this mechanism shines. By preventing the formation of trans-isomers at the source, the process reduces the burden on analytical laboratories to detect and quantify these impurities at trace levels. For quality assurance teams, a cleaner reaction profile means more consistent batch-to-batch quality and a lower risk of failing specification limits. The robustness of the copper catalyst also suggests a tolerance to various substituents on the aromatic ring, making this methodology applicable to a broader range of substituted cyclohexylamines beyond just sertraline, adding versatility to the manufacturing platform.
How to Synthesize 4-(3,4-dichlorophenyl)-1,2,3,4-tetrahydro-N-methyl-1-naphthylamine Efficiently
To implement this high-efficiency synthesis, manufacturers must adhere to specific protocol parameters regarding catalyst activation and reaction conditions. The process generally begins with the preparation of the imine or nitrone precursor, followed by the critical hydrogenation step. Detailed operational guidelines emphasize the importance of catalyst pre-activation (pre-hydrogenation) to ensure maximum activity and selectivity. The choice of solvent, specifically lower alcohols like ethanol, is non-negotiable for achieving the reported high cis-selectivity. Below is the structured approach for executing this synthesis in a GMP-compliant environment.
- Preparation of the imine or nitrone precursor through the reaction of the corresponding ketone with methylamine or N-methylhydroxylamine.
- Hydrogenation of the precursor in a protic solvent (e.g., ethanol) using a copper-containing catalyst (e.g., copper chromite or CuCrBa oxide) at elevated temperatures (90-150°C) and pressure (10-20 bar).
- Isolation of the cis-enriched product via filtration of the catalyst and solvent removal, followed by optional chiral resolution if optical purity is required.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed technology offers tangible strategic advantages that go beyond mere chemical yield. The primary benefit is the drastic simplification of the supply chain for raw materials. Copper catalysts are abundant, inexpensive, and widely available globally, unlike palladium or rhodium which are subject to volatile market prices and geopolitical supply risks. By switching to a base metal catalyst, companies can secure long-term pricing stability and reduce the working capital tied up in expensive catalytic materials. This shift supports a strategy of cost reduction in API manufacturing by lowering the direct material costs associated with the hydrogenation step.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts directly lowers the bill of materials. Furthermore, the high selectivity (>95% cis) significantly reduces the volume of solvents and reagents needed for purification. In traditional processes, removing 30% trans-impurity requires extensive chromatography or multiple recrystallizations, which are solvent-intensive and time-consuming. By minimizing these downstream operations, the process achieves substantial cost savings in utility consumption and waste disposal, enhancing the overall economic viability of the production line.
- Enhanced Supply Chain Reliability: Relying on copper-based chemistry mitigates the risk of supply disruptions associated with precious metals. Copper catalysts like copper chromite or Raney copper are commodity chemicals with multiple qualified suppliers worldwide. This diversification ensures that production schedules are not held hostage by the availability of a single source of palladium. Additionally, the robustness of the reaction conditions (moderate temperature and pressure) allows for flexible manufacturing across different facilities, ensuring continuity of supply even if one production site faces maintenance or regulatory issues.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in autoclaves suitable for pilot and commercial scales. The use of simple alcohols as solvents aligns well with environmental, health, and safety (EHS) guidelines, as these solvents are easier to recover and recycle compared to chlorinated or aromatic solvents often used in older methods. Reduced waste generation from higher selectivity means lower environmental compliance costs and a smaller carbon footprint, which is increasingly important for meeting corporate sustainability goals and satisfying the requirements of environmentally conscious clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cis-selective hydrogenation technology. These answers are derived directly from the experimental data and claims within patent CN1213991C, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What is the primary advantage of using copper catalysts over palladium for sertraline synthesis?
A: Copper-containing catalysts provide significantly higher diastereoselectivity, achieving cis/trans ratios greater than 95:5 (up to 99.6:0.4), compared to traditional palladium on carbon which typically yields a 70:30 ratio, thereby drastically reducing purification costs.
Q: Can this process be scaled for industrial production of API intermediates?
A: Yes, the process is highly scalable as it utilizes robust heterogeneous copper catalysts and standard hydrogenation equipment capable of operating at moderate pressures (10-20 bar) and temperatures, making it suitable for commercial scale-up from kilograms to metric tons.
Q: Does this method eliminate the need for complex chiral separation steps?
A: While the process produces a racemic cis-mixture with extremely high selectivity against trans-isomers, it simplifies the downstream processing significantly. If optical purity is needed, the high cis-content allows for more efficient resolution via crystallization (e.g., using mandelic acid) compared to separating a complex mixture of four isomers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sertraline Intermediate Supplier
The technological breakthroughs detailed in CN1213991C underscore the potential for highly efficient, cost-effective production of critical antidepressant intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate these patent innovations into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high selectivity observed in the lab is maintained at the plant scale. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of sertraline intermediate meets the highest global standards for potency and impurity profiles.
We invite pharmaceutical partners to collaborate with us to leverage this advanced copper-catalyzed route. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain for sertraline and related cyclic amines, ensuring a reliable flow of high-quality intermediates for your final drug product manufacturing.
