Advanced Enzymatic Synthesis of Hexahydrofurofuranol Derivatives for Commercial API Production
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective pathways for synthesizing complex chiral intermediates, particularly for antiretroviral therapies. Patent CN110372532B, published on November 29, 2022, introduces a groundbreaking process for the preparation of hexahydrofurofuranol derivatives, specifically targeting the key intermediate (3R, 3aS, 6aR)-hexahydrofuro[2,3-b]furan-3-ol used in the synthesis of the HIV protease inhibitor Darunavir. This technology represents a significant paradigm shift from traditional chemical catalysis to a highly selective biocatalytic approach. By leveraging a specifically engineered aldehyde ketone reductase derived from Saccharomyces kudriavzevii, the disclosed method achieves exceptional optical purity under mild reaction conditions, addressing critical pain points in impurity control and environmental sustainability. For global procurement teams and R&D directors, this patent offers a viable alternative to existing routes that rely on expensive chiral ligands or harsh reducing agents.
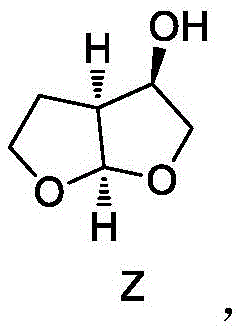
The structural complexity of the target molecule, as depicted above, necessitates precise stereochemical control, which this enzymatic route delivers with remarkable efficiency. The invention not only covers the final cyclization steps but also provides novel intermediate compounds and their preparation methods, creating a comprehensive intellectual property landscape for manufacturers aiming to secure their supply chains for next-generation antiretroviral drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Darunavir intermediates has relied heavily on chemical methodologies that present substantial challenges for large-scale manufacturing. Prior art, such as processes utilizing chiral ligand catalysts (e.g., those employed by Sumitomo Chemical), often involves the use of precious transition metals and complex ligand systems that are not only costly but also difficult to remove to the stringent ppm levels required for pharmaceutical ingredients. Furthermore, these chemical reductions frequently require low temperatures and anhydrous conditions, leading to high energy consumption and operational complexity. Another existing approach involves the use of carbonyl reductase polypeptides, as seen in European patent EP2634180A1; however, the enzymes utilized in these earlier methods often suffer from suboptimal activity or insufficient stereoselectivity, resulting in lower yields and the formation of difficult-to-separate diastereomeric impurities. These limitations collectively drive up the cost of goods sold (COGS) and introduce significant supply chain risks due to the reliance on specialized reagents and rigorous process controls.
The Novel Approach
In stark contrast, the methodology disclosed in CN110372532B utilizes a tailored biocatalytic strategy that fundamentally simplifies the chiral construction step. The core innovation lies in the application of a specific aldehyde ketone reductase (SEQ ID NO:1) which demonstrates superior catalytic efficiency and stereoselectivity compared to wild-type enzymes or those described in prior patents. This novel approach allows for the reduction of dicarbonyl precursors (Formula B) to chiral hydroxy-ketones (Formula C) with extremely high diastereomeric excess (de) and enantiomeric excess (ee) values, often exceeding 99%. The reaction proceeds in aqueous-organic biphasic systems at ambient temperatures (25-37°C), drastically reducing energy requirements and eliminating the need for cryogenic cooling. Moreover, the use of whole-cell biocatalysts facilitates easy separation of the biocatalyst from the product stream, streamlining the downstream purification process and enhancing the overall economic viability of the synthesis for commercial scale-up.
Mechanistic Insights into Enzymatic Stereoselective Reduction
The heart of this technological advancement is the highly specific interaction between the engineered aldehyde ketone reductase and the prochiral dicarbonyl substrate. The enzyme, derived from Saccharomyces kudriavzevii and expressed in E. coli BL21(DE3), possesses a unique active site architecture that strictly governs the hydride transfer from the cofactor NADPH to the specific carbonyl carbon. This precise orientation ensures that the reduction occurs exclusively to form the desired (S, R) configuration at the newly formed chiral centers. The mechanism involves a dynamic kinetic resolution or a highly selective reduction where the enzyme discriminates between the two carbonyl groups and the two faces of the prochiral ketone with exceptional fidelity. To sustain the catalytic cycle, the process incorporates a cofactor regeneration system utilizing glucose dehydrogenase (GDH), which oxidizes glucose to gluconolactone while recycling NADP+ back to NADPH. This coupled enzyme system ensures that only catalytic amounts of the expensive cofactor are needed, making the process economically feasible for ton-scale production.
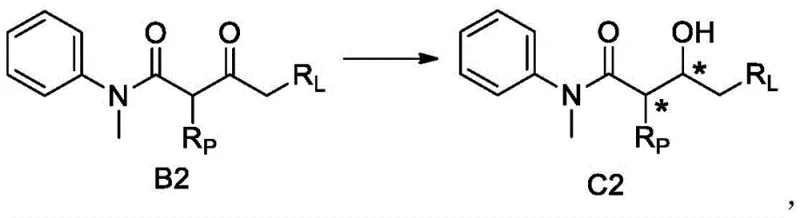
As illustrated in the reaction scheme above, the transformation of the achiral or racemic precursor (Formula B2) into the chiral intermediate (Formula C2) is the critical chirality-building step. The patent data indicates that the engineered enzyme achieves conversion rates as high as 97.8% with minimal formation of the undesired syn-isomer or opposite enantiomer. The high selectivity is attributed to specific amino acid residues within the enzyme sequence (SEQ ID NO:1) that create a steric environment favoring the attack on the Re-face or Si-face of the ketone, depending on the substrate orientation. This level of control effectively suppresses the generation of the four possible stereoisomers, isolating the single desired diastereomer required for the subsequent cyclization to the bis-tetrahydrofuran core of Darunavir.
Furthermore, the impurity profile is significantly improved compared to chemical reduction. In chemical methods using borohydrides or aluminum hydrides, non-selective reduction often leads to a mixture of diols that require extensive chromatographic purification. In this enzymatic route, the primary impurity is the unreacted starting material, which can be easily recycled or removed during the extraction phase. The high de value (up to 99.1%) and ee value (up to 99.7%) reported in the examples confirm that the enzymatic machinery effectively filters out incorrect stereochemical pathways, ensuring that the final API meets the rigorous regulatory standards for chiral purity without the need for complex chiral resolution steps later in the synthesis.
How to Synthesize Hexahydrofurofuranol Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory scale to pilot and commercial production. The process begins with the preparation of the dicarbonyl substrate, followed by the critical biocatalytic reduction, and concludes with chemical cyclization. The operational simplicity of the enzymatic step, which can be performed in standard stirred-tank reactors using whole-cell suspensions, makes it highly attractive for contract development and manufacturing organizations (CDMOs). The detailed standardized synthesis steps, including specific buffer conditions, co-solvent ratios, and temperature profiles, are essential for replicating the high yields and purity reported in the patent examples.
- Preparation of the dicarbonyl precursor (Formula B2) via substitution reactions involving N-methylaniline and halogenated esters.
- Biocatalytic reduction using whole cells of engineered aldehyde ketone reductase (SEQ ID NO: 1) with glucose dehydrogenase for cofactor regeneration at 37°C.
- Downstream processing involving solvent extraction, drying, and final cyclization to yield the target hexahydrofurofuranol derivative with >99% ee.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic technology offers tangible strategic benefits beyond mere technical feasibility. The shift from chemical to biological catalysis fundamentally alters the cost structure and risk profile of the supply chain. By eliminating the dependency on scarce or volatile raw materials associated with traditional chiral synthesis, manufacturers can achieve greater stability in their sourcing strategies. The following points detail how this innovation translates into commercial value for your organization.
- Cost Reduction in Manufacturing: The implementation of this enzymatic route leads to significant cost optimization by removing the need for expensive chiral ligands and precious metal catalysts, which are major cost drivers in conventional asymmetric synthesis. Additionally, the mild reaction conditions (ambient temperature and pressure) result in substantially lower energy consumption compared to cryogenic chemical reductions. The high conversion rates and selectivity minimize waste generation and reduce the volume of solvents required for purification, further driving down the operational expenditures associated with waste disposal and solvent recovery. The ability to use whole-cell biocatalysts also simplifies the enzyme production process, allowing for in-house fermentation rather than purchasing costly immobilized enzymes.
- Enhanced Supply Chain Reliability: Relying on a biocatalytic process diversifies the supply chain away from petrochemical-derived chiral auxiliaries, which can be subject to market fluctuations and geopolitical instability. The key reagents, such as glucose and the genetically engineered bacterial strains, are readily available and can be produced at scale with high consistency. This robustness ensures a continuous supply of high-quality intermediates, reducing the risk of production delays caused by raw material shortages. Furthermore, the simplified workflow reduces the number of unit operations, thereby decreasing the potential for equipment bottlenecks and enhancing the overall throughput of the manufacturing facility.
- Scalability and Environmental Compliance: The process is inherently green, aligning with increasingly strict environmental regulations and corporate sustainability goals. The use of water as the primary reaction medium and the avoidance of toxic heavy metals significantly reduce the environmental footprint of the manufacturing process. This 'green' profile facilitates easier regulatory approval and permits for new manufacturing sites. From a scalability perspective, fermentation-based enzyme production and aqueous-phase biotransformations are well-established technologies that scale linearly from liters to cubic meters, ensuring that the process remains efficient and controllable even at multi-ton annual production volumes required for global API supply.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the process suitability for your specific manufacturing needs.
Q: What is the primary advantage of the enzymatic method described in CN110372532B over traditional chemical synthesis?
A: The enzymatic method utilizes a specific aldehyde ketone reductase derived from Saccharomyces kudriavzevii to construct chirality under mild conditions (25-37°C), achieving significantly higher optical purity (ee >99%) compared to traditional chiral ligand catalysis which often requires harsh conditions and expensive metals.
Q: Can this process be scaled for industrial manufacturing of Darunavir intermediates?
A: Yes, the patent explicitly states the method is suitable for industrial production. It employs robust whole-cell biocatalysts that can be produced via fermentation, eliminating the need for costly transition metal catalysts and simplifying the purification workflow for large-scale operations.
Q: What are the key impurities controlled in this synthetic route?
A: The high stereoselectivity of the engineered enzyme minimizes the formation of unwanted diastereomers and enantiomers. The process achieves a de value of up to 99.1% and an ee (anti) value of 99.7%, effectively suppressing the generation of stereochemical impurities common in non-enzymatic reductions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hexahydrofurofuranol Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of key antiviral intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate complex patented routes like CN110372532B into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from process development to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and mass spectrometry to guarantee that every batch of hexahydrofurofuranol derivative meets the highest international pharmacopeial standards.
We invite you to collaborate with us to leverage this innovative enzymatic technology for your Darunavir supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your current production volumes and quality requirements. Please contact us today to request specific COA data, route feasibility assessments, and a comprehensive proposal on how we can support your long-term strategic goals with reliable, cost-effective, and sustainable manufacturing solutions.
