Optimizing Analgesic Intermediate Synthesis via Novel Tertiary Chlorination Technology
Optimizing Analgesic Intermediate Synthesis via Novel Tertiary Chlorination Technology
The pharmaceutical industry continuously seeks robust synthetic routes for complex analgesic intermediates, particularly those involving sterically hindered functional groups. Patent CN1662486A introduces a transformative methodology for converting tertiary hydroxyl groups into tertiary chlorides, a critical step in the synthesis of potent pain-relieving compounds. This innovation addresses long-standing inefficiencies in halogenation chemistry by replacing excessive reagent usage with a precise, solvent-mediated approach. By utilizing toluene or xylene derivatives as the reaction medium rather than relying on neat thionyl chloride, the process achieves a dramatic improvement in yield and operational safety. For R&D teams focused on high-purity pharmaceutical intermediate production, this technology offers a pathway to reduce impurity profiles while maintaining strict stereochemical integrity. The implications for commercial manufacturing are profound, offering a reliable pharmaceutical intermediate supplier the ability to scale production with significantly reduced environmental footprint and cost.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chlorination of tertiary alcohols, such as those found in cyclohexyl-based analgesic precursors, relied heavily on methods described in earlier literature like EP0753506A1. These conventional protocols typically employed thionyl chloride not merely as a chlorinating agent but as the bulk solvent itself. This approach necessitated the use of massive molar excesses of the reagent, creating severe logistical and safety challenges during the workup phase. The removal of excess thionyl chloride required energy-intensive distillation or purging with nitrogen streams, which placed considerable stress on equipment and personnel due to the release of corrosive sulfur dioxide and hydrogen chloride gases. Furthermore, the chemical efficiency of these legacy methods was suboptimal, often capping yields at approximately 55% of theory. Such low conversion rates result in substantial material loss and complicate downstream purification, making the process economically unviable for large-scale commercial operations seeking cost reduction in API manufacturing.
The Novel Approach
The methodology disclosed in CN1662486A represents a paradigm shift by introducing an inert aromatic solvent system, specifically selecting from toluene, o-xylene, m-xylene, or p-xylene. In this optimized regime, thionyl chloride is used in a controlled stoichiometric ratio, preferably between 1.5 to 1.7 equivalents, rather than as a solvent. This adjustment fundamentally alters the reaction thermodynamics, allowing the starting tertiary alcohol hydrochloride salt to dissolve completely upon reaction initiation without requiring vast quantities of hazardous reagents. The result is a homogeneous solution that facilitates superior heat transfer and reaction control. Yields consistently exceed 70% of theory, with specific embodiments demonstrating up to 90% conversion. This leap in efficiency directly translates to enhanced supply chain reliability, as less raw material is required to produce the same output of high-purity analgesic intermediates, thereby stabilizing production schedules and reducing waste disposal costs.
Mechanistic Insights into Thionyl Chloride-Mediated Chlorination
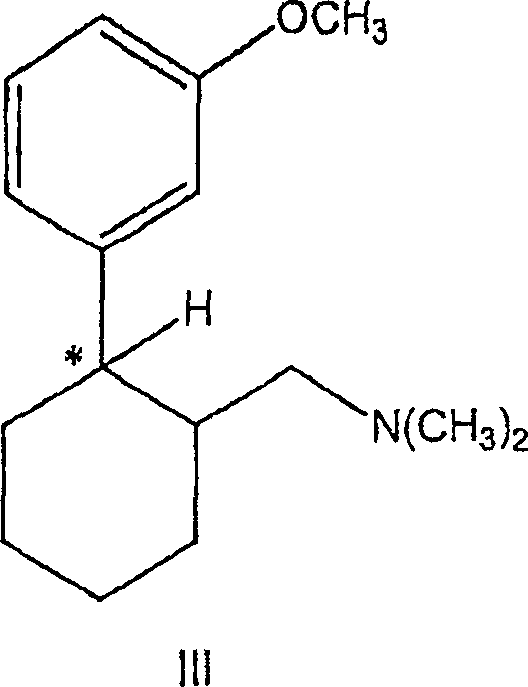
A detailed examination of the reaction mechanism reveals why this solvent-switch strategy is so effective for sterically crowded tertiary centers. The reaction proceeds through the formation of a chlorosulfite intermediate, which subsequently undergoes nucleophilic substitution by the chloride ion. Crucially, the use of toluene allows for the stabilization of reactive species without promoting the elimination side reactions that often plague tertiary alcohol transformations. The patent data indicates that the stereochemistry at the chiral center marked with an asterisk is rigorously preserved throughout the transformation. This retention of configuration is vital for the biological efficacy of the final drug substance, ensuring that the produced (1S,2S)-configured chloride maintains the necessary spatial arrangement for receptor binding. The ability to maintain optical purity without racemization distinguishes this process as a superior choice for the commercial scale-up of complex pharmaceutical additives.
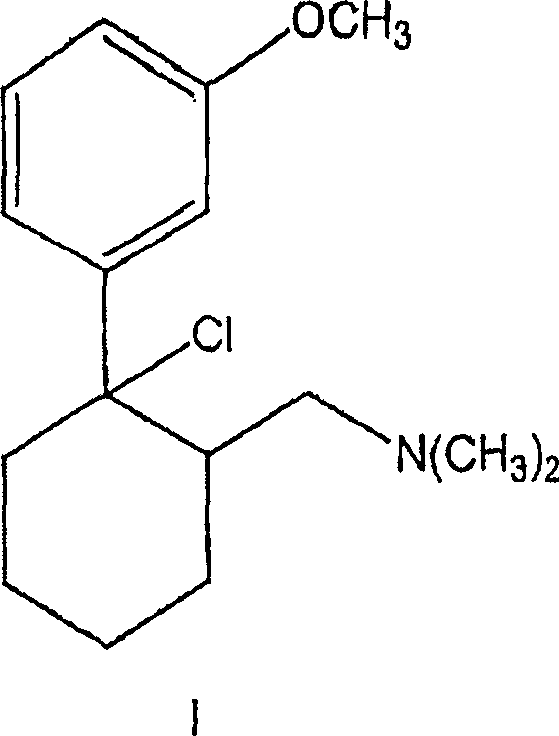
Furthermore, an unexpected mechanistic phenomenon observed in this process is the suppression of gas evolution during the heating phase. Typically, the reaction of alcohols with thionyl chloride releases sulfur dioxide and hydrogen chloride gases vigorously. However, in this toluene-based system, it is hypothesized that the amine functionality within the substrate forms a stable complex with the generated acidic byproducts, keeping them in solution. This complexation prevents the premature release of toxic gases, simplifying the reactor venting requirements and enhancing operator safety. Following the chlorination, the intermediate can be subjected to catalytic hydrogenation to remove the chlorine atom, yielding the desired amine structure. This subsequent step, illustrated in the reaction scheme below, utilizes palladium on carbon in alcoholic solvents to achieve quantitative conversion while preserving the critical stereocenter.
Impurity control is another cornerstone of this mechanistic advantage. By avoiding the harsh conditions of neat thionyl chloride, the formation of elimination byproducts (alkenes) is minimized. The patent specifies that non-polar solvents like hexane or THF are unsuitable as they lead to numerous side products, highlighting the unique solvation properties of toluene that favor the substitution pathway. The resulting crude product can be isolated either by distilling off the solvent and byproducts under vacuum or by cooling the mixture to precipitate the solid hydrochloride salt. Both isolation methods yield material with high chemical and optical purity, ready for the next synthetic transformation without extensive chromatographic purification. This streamlined workflow is essential for maintaining a lean manufacturing profile in high-volume facilities.
How to Synthesize Tertiary Chloride Intermediates Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize the benefits of the patented technology. The process begins by suspending the tertiary alcohol starting material in toluene, followed by the controlled addition of thionyl chloride at moderate temperatures. The detailed standardized synthesis steps see the guide below for specific operational parameters regarding temperature ramps and stirring rates. Adhering to these protocols ensures that the reaction mixture transitions smoothly from a suspension to a clear solution, indicating complete conversion. The flexibility to isolate the product via crystallization or distillation provides manufacturers with options to optimize based on their specific equipment capabilities and energy constraints.
- Suspend the tertiary alcohol hydrochloride salt in toluene and heat to 30°C to form a stable suspension.
- Add 1.5 to 1.7 equivalents of thionyl chloride gradually while maintaining temperature control to initiate the chlorination reaction.
- Heat the mixture to 35-45°C for 2-3 hours until a clear solution forms, then isolate the product via cooling precipitation or vacuum distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this chlorination technology offers tangible strategic benefits beyond mere chemical yield. The primary advantage lies in the drastic reduction of reagent consumption. By shifting from using thionyl chloride as a solvent to using it as a stoichiometric reagent, the volume of hazardous chemical purchasing is significantly reduced. This reduction lowers the direct material costs and diminishes the logistical burden associated with storing and handling large quantities of corrosive liquids. Additionally, the simplified workup procedure eliminates the need for complex gas scrubbing systems required to handle the massive off-gassing of the old method. This simplification reduces capital expenditure on safety infrastructure and lowers the ongoing operational costs related to waste neutralization and disposal, contributing to substantial cost savings in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the efficient utilization of raw materials. Since the yield increases from a theoretical maximum of 55% to over 70% or even 90%, the amount of starting alcohol required per kilogram of final product drops precipitously. This efficiency gain means that for every ton of product manufactured, significantly less precursor is consumed, directly improving the gross margin. Furthermore, the ability to recover and recycle the toluene solvent adds another layer of cost optimization, as the solvent can be distilled and reused in subsequent batches without loss of performance. The elimination of expensive purification steps to remove elimination byproducts further reduces the cost of goods sold, making the final analgesic intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by processes that are sensitive to minor variations or require hard-to-source reagents in massive quantities. This method enhances reliability by using common, commodity-grade solvents like toluene, which are readily available from multiple global suppliers. The robustness of the reaction, which tolerates slight variations in temperature and stoichiometry without forming intractable tars, ensures consistent batch-to-batch quality. This consistency reduces the risk of production delays caused by failed batches or out-of-specification results. Moreover, the reduced dependency on excessive thionyl chloride mitigates the risk of supply disruptions related to this specific reagent, allowing the supply chain to remain agile and responsive to fluctuating market demands for pain management medications.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production often exposes hidden safety and environmental flaws. This technology is inherently scalable because it avoids the thermal hazards associated with heating large volumes of neat thionyl chloride. The containment of sulfur dioxide and hydrogen chloride within the reaction mixture during the heating phase significantly lowers the emission load on factory scrubbers, facilitating compliance with increasingly stringent environmental regulations. The ability to operate at moderate temperatures (35-45°C) reduces energy consumption for heating and cooling, aligning with green chemistry principles. This environmental stewardship not only protects the corporate reputation but also future-proofs the manufacturing site against potential regulatory bans on high-emission processes, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this chlorination technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this route into existing production lines. The focus is on practical outcomes such as yield improvement, safety enhancements, and stereochemical fidelity.
Q: How does the toluene-based method improve yield compared to conventional thionyl chloride processes?
A: Conventional methods often use thionyl chloride as both reagent and solvent, leading to excessive waste and difficult workups with yields around 55%. The novel toluene-based method utilizes stoichiometric amounts of thionyl chloride (1-3 equivalents), achieving yields higher than 70% and up to 90% by facilitating better solubility and controlled reaction kinetics.
Q: Does this chlorination process affect the stereochemistry of the chiral center?
A: No, the process is designed to retain the stereochemistry at the reaction center. Starting from specific enantiomers like (1S,2S)-configured alcohols, the reaction produces the corresponding chloride with retained configuration, which is critical for the biological activity of the downstream analgesic compounds.
Q: What are the safety advantages regarding gas evolution in this new method?
A: Unlike traditional methods that release significant amounts of sulfur dioxide and hydrogen chloride gas during heating, this method surprisingly retains these byproducts in solution, likely through complexation with the amine substrate. This significantly reduces the load on scrubbing systems and improves operational safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Analgesic Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields demonstrated in patent CN1662486A are fully realized in your supply chain. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced HPLC and NMR capabilities to verify the stereochemical integrity of every batch. Our commitment to quality ensures that the analgesic intermediates we deliver meet the exacting standards required for downstream pharmaceutical formulation, providing you with a foundation of trust and reliability.
We invite you to collaborate with us to leverage this advanced chlorination technology for your specific product needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this solvent-mediated process. Please contact us to request specific COA data and route feasibility assessments tailored to your project timelines. By partnering with us, you gain access to a supply chain that prioritizes efficiency, safety, and scientific excellence, positioning your organization for success in the competitive landscape of pain management therapeutics.
