Scalable Manufacturing of High-Purity Tropane Intermediates via Novel Phase Transfer Catalysis
The pharmaceutical industry continuously seeks robust synthetic routes for central nervous system (CNS) active ingredients, particularly for treating neurodegenerative conditions such as Parkinson's and Alzheimer's disease. Patent CN1910180A introduces a groundbreaking methodology for the preparation of 2-(ethoxymethyl)-tropane derivatives, addressing critical limitations found in prior art regarding safety and reproducibility. This innovation shifts the paradigm from hazardous hydride-based chemistry to a milder, phase-transfer catalyzed etherification process that ensures consistent quality and operational safety. By utilizing ethyl bromide in the presence of a base and a specific phase transfer catalyst, manufacturers can now access these valuable intermediates with significantly improved efficiency. The structural integrity of the target molecule, as depicted below, is maintained with high fidelity, ensuring the biological activity required for downstream drug formulation.
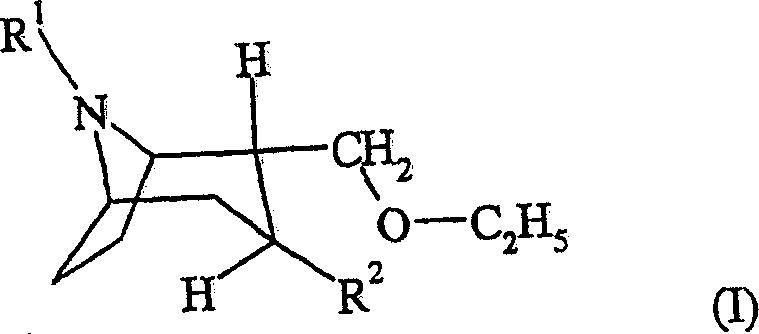
For R&D directors and process chemists, the ability to source a reliable pharmaceutical intermediates supplier who understands these nuanced synthetic improvements is paramount. The transition to this novel pathway not only enhances the purity profile of the final active substance but also streamlines the regulatory documentation required for drug master files. As we delve deeper into the technical specifics, it becomes evident that this patent represents a substantial leap forward in the cost reduction in pharmaceutical intermediates manufacturing, offering a sustainable alternative to legacy processes that have long plagued production teams with safety concerns and variable yields.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of 2-(ethoxymethyl)-tropane derivatives was predominantly governed by the teachings of International Patent Application WO97/30997, which relied heavily on the use of sodium hydride as a base and diethyl sulfate or tosylates as alkylating agents. From an industrial safety perspective, the use of sodium hydride is fraught with peril; it is a pyrophoric solid that reacts violently with moisture, necessitating expensive inert atmosphere equipment and rigorous safety protocols that drive up operational expenditures. Furthermore, diethyl sulfate is a known carcinogen and potent alkylating agent, posing severe health risks to personnel and creating complex waste disposal challenges that burden environmental compliance teams. The legacy process also suffered from poor reproducibility, often requiring prolonged reaction times that led to the formation of difficult-to-separate solid byproducts, ultimately resulting in unsatisfactory yields that hovered around 70% and compromised the economic viability of large-scale campaigns.
The Novel Approach
In stark contrast, the methodology disclosed in CN1910180A circumvents these hazards by employing a phase transfer catalysis (PTC) system using pulverized potassium hydroxide and ethyl bromide. This approach eliminates the need for pyrophoric reagents and toxic sulfates, replacing them with commercially available, easier-to-handle chemicals that significantly lower the barrier to entry for safe manufacturing. The reaction proceeds under much milder conditions, typically between 20°C and 65°C, which reduces energy consumption and minimizes thermal degradation of the sensitive tropane backbone. By optimizing the stoichiometry with approximately 4 equivalents of ethyl bromide and utilizing specific quaternary ammonium salts like tetra-n-butylammonium bisulfate, the process achieves yields exceeding 83% with purity levels greater than 99.4%. This dramatic improvement in both safety and efficiency makes the novel approach the preferred choice for any organization seeking the commercial scale-up of complex pharmaceutical intermediates without compromising on worker safety or environmental standards.
Mechanistic Insights into Phase Transfer Catalyzed Etherification
The core of this technological advancement lies in the sophisticated application of phase transfer catalysis to facilitate the nucleophilic substitution of the hydroxymethyl group. In this mechanism, the pulverized potassium hydroxide serves as the base to deprotonate the 2-(hydroxymethyl)-tropane precursor, generating the corresponding alkoxide ion at the solid-liquid interface. The phase transfer catalyst, typically a tetraalkylammonium salt, plays a critical role by solubilizing the hydroxide or alkoxide species into the organic phase, where the concentration of the electrophile (ethyl bromide) is highest. This intimate contact between the nucleophile and the electrophile within the organic solvent, such as 1,2-dimethoxyethane (DME) or toluene, accelerates the SN2 reaction rate significantly compared to heterogeneous conditions without a catalyst. The choice of tetra-n-butylammonium bisulfate is particularly strategic, as its lipophilicity ensures efficient transport across the phase boundary while maintaining stability under the reaction conditions, thereby driving the equilibrium towards the desired ether product with minimal formation of elimination byproducts.
Impurity control is another critical aspect where this mechanistic understanding translates to commercial value. Traditional methods using strong, non-nucleophilic bases like sodium hydride in aprotic solvents can sometimes promote E2 elimination pathways, leading to olefinic impurities that are structurally similar to the product and difficult to remove. The PTC system described here operates under kinetic control that favors substitution over elimination, largely due to the specific solvation environment created by the quaternary ammonium cation. Furthermore, the subsequent acid treatment step, preferably using citric acid to form the citrate salt, acts as a powerful purification tool. This salt formation selectively precipitates the desired product while leaving neutral organic impurities in the mother liquor, ensuring that the final isolated material meets stringent purity specifications without the need for column chromatography. This level of impurity management is essential for reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to move swiftly from pilot plant to full commercial production.
How to Synthesize 2-(Ethoxymethyl)-tropane Derivatives Efficiently
The practical execution of this synthesis requires careful attention to reagent addition rates and temperature control to maximize the benefits of the phase transfer system. The process begins by suspending the 2-(hydroxymethyl)-tropane precursor in a suitable diluent like DME, followed by the addition of pulverized potassium hydroxide and the catalytic amount of the phase transfer agent. Ethyl bromide is then metered in slowly to manage the exotherm and prevent local overheating, which could degrade the catalyst or the product. Once the addition is complete, the reaction mixture is heated to facilitate the completion of the etherification, followed by a straightforward aqueous workup and salt formation. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the operational parameters.
- Prepare a reaction mixture containing 1 equivalent of 2-(hydroxymethyl)-tropane derivative, approximately 8 equivalents of pulverized potassium hydroxide, and 0.1 equivalents of tetra-n-butylammonium bisulfate in 1,2-dimethoxyethane (DME).
- Meter in 4 equivalents of ethyl bromide over 15 minutes while maintaining the temperature between 20°C and 35°C, then heat the mixture to 58-62°C for 1.5 hours to complete the etherification.
- Quench the reaction with water, separate the organic phase, concentrate under reduced pressure, and treat the residue with citric acid in methanol to isolate the pure citrate salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process offers tangible benefits that extend far beyond the laboratory bench, fundamentally altering the cost structure and risk profile of the supply chain. By eliminating the requirement for specialized handling of pyrophoric materials and carcinogenic alkylating agents, facilities can reduce their insurance premiums and safety infrastructure costs, leading to substantial cost savings in the overall manufacturing budget. The use of common, commodity chemicals like ethyl bromide and potassium hydroxide ensures a stable and resilient supply of raw materials, shielding production schedules from the volatility often associated with specialty reagents. Moreover, the simplified workup procedure, which avoids complex distillation or chromatography, reduces the cycle time per batch, thereby increasing the throughput capacity of existing manufacturing assets without the need for capital-intensive expansion.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous reagents like sodium hydride and diethyl sulfate with cost-effective alternatives such as ethyl bromide and potassium hydroxide directly lowers the bill of materials. Additionally, the high yield and purity achieved in a single step eliminate the need for resource-intensive purification processes, further driving down the cost of goods sold. The reduction in waste disposal costs, owing to the absence of toxic sulfate byproducts, contributes to a leaner and more economically efficient production model that enhances overall profitability.
- Enhanced Supply Chain Reliability: The robustness of this chemical process ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply to downstream pharmaceutical customers. The mild reaction conditions reduce the likelihood of equipment corrosion or failure, minimizing unplanned downtime and maintenance requirements. Furthermore, the availability of raw materials on the global market means that procurement teams can easily source multiple vendors for key inputs, mitigating the risk of supply disruptions and ensuring continuity of operations even during market fluctuations.
- Scalability and Environmental Compliance: This process is inherently scalable, having been designed with industrial application in mind, allowing for seamless transition from kilogram to metric-ton scales. The avoidance of persistent organic pollutants and toxic heavy metals simplifies the environmental permitting process and reduces the burden on wastewater treatment facilities. By aligning with green chemistry principles through the use of safer solvents and catalysts, manufacturers can meet increasingly stringent regulatory requirements while enhancing their corporate sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, providing clarity for stakeholders evaluating its adoption. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these nuances is crucial for assessing the feasibility of integrating this technology into existing production portfolios.
Q: Why is the new PTC method safer than the conventional Sodium Hydride route?
A: The conventional method described in WO97/30997 utilizes sodium hydride, which is pyrophoric and poses significant fire hazards on an industrial scale, alongside diethyl sulfate which is highly toxic. The novel process replaces these with solid potassium hydroxide and ethyl bromide under phase transfer conditions, drastically reducing operational risk and simplifying safety protocols for large-scale manufacturing.
Q: What purity levels can be achieved with this improved synthesis route?
A: Experimental data from the patent indicates that the resulting 2-(ethoxymethyl)-tropane citrate salts achieve purity levels exceeding 99.4% to 99.5% directly after crystallization. This high purity is attributed to the selective nature of the phase transfer catalysis which minimizes side reactions and eliminates the need for complex chromatographic purification steps.
Q: Is this process suitable for commercial scale-up of complex pharmaceutical intermediates?
A: Yes, the process is specifically designed for industrial scalability. It operates at moderate temperatures (20-65°C) using common solvents like DME or toluene and avoids hazardous reagents that require specialized containment. The high space-time yield and simplified workup procedure make it highly viable for metric-ton production campaigns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(Ethoxymethyl)-tropane Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert process chemists has thoroughly analyzed patent CN1910180A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative route to life. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs that ensure every batch conforms to the highest international standards. Our facility is equipped to handle the specific requirements of phase transfer catalysis, ensuring that the safety and efficiency benefits of this process are fully realized for our clients.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and innovation in your drug development pipeline.
