Advanced Palladium-Catalyzed Synthesis of Chiral Oxygen-Containing Eight-Membered Ring Compounds for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex macrocyclic scaffolds, which serve as critical cores for bioactive molecules. A significant breakthrough in this domain is detailed in patent CN113583015A, which discloses a highly efficient method for synthesizing chiral oxygen-containing eight-membered ring compounds via a palladium-catalyzed asymmetric allylic cycloaddition reaction. This innovative approach addresses the longstanding synthetic challenges associated with medium-to-large ring formation, offering a streamlined pathway to valuable intermediates found in antimalarial agents like Decursine and antiulcer lignans. By leveraging a specialized palladium catalytic system coupled with a novel chiral phosphoramidite ligand, this technology enables the direct assembly of these complex structures from readily available gamma-methylene-delta-valerolactones and o-quinone methides. For R&D directors and procurement specialists, this represents a pivotal shift towards more reliable and scalable manufacturing processes for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of chiral oxygen-containing eight-membered ring compounds has been fraught with significant technical and economic hurdles. Traditional reliance on natural product separation or biosynthesis is inherently inefficient, characterized by time-consuming isolation procedures, extensive labor requirements, and notoriously low yields that cannot meet commercial demand. Furthermore, conventional chemical synthetic strategies often struggle with the thermodynamic penalties associated with forming medium-sized rings, specifically the unfavorable enthalpy and entropy effects alongside transannular interactions that destabilize the transition states. These factors frequently result in poor regioselectivity and stereoselectivity, necessitating complex purification steps that drive up costs and extend lead times. Consequently, the existing methods lack the robustness required for the commercial scale-up of complex pharmaceutical intermediates, creating a bottleneck in the supply chain for drug development programs targeting diseases where these macrocycles are essential.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN113583015A introduces a transformative [4+4] cycloaddition strategy that bypasses traditional thermodynamic barriers. This novel approach utilizes a palladium-catalyzed asymmetric allylic cycloaddition between a gamma-methylene-delta-valerolactone and an o-quinone methide to construct the eight-membered ring in a single, convergent step. The reaction operates under remarkably mild conditions, typically at temperatures as low as -20°C in solvents like ethylbenzene, which minimizes thermal degradation and side reactions. By employing a specific chiral ligand system, the process achieves exceptional stereocontrol, delivering products with high diastereoselectivity (dr > 20:1) and enantiomeric excess (ee > 90%). This methodological leap not only simplifies the synthetic route but also drastically improves atom economy, making it an ideal solution for cost reduction in chiral macrocycle manufacturing.
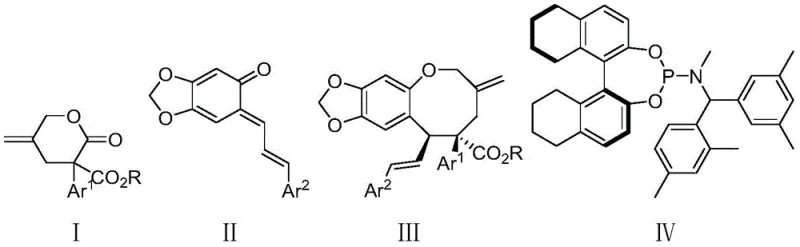
Mechanistic Insights into Palladium-Catalyzed Asymmetric Allylic Cycloaddition
The core of this synthetic breakthrough lies in the sophisticated interplay between the palladium catalyst and the chiral phosphoramidite ligand (Formula IV). The mechanism initiates with the oxidative addition of the zero-valent palladium species to the gamma-methylene-delta-valerolactone substrate, generating a reactive pi-allyl palladium intermediate. This step is crucial as it activates the relatively inert lactone ring towards nucleophilic attack. The chiral environment created by the bulky binaphthyl-based ligand dictates the facial selectivity of the subsequent bond formation, ensuring that the o-quinone methide attacks from the preferred trajectory. This precise spatial arrangement is what allows the reaction to overcome the entropic penalty of ring closure, effectively templating the formation of the eight-membered ring with high fidelity. Understanding this catalytic cycle is vital for process chemists aiming to optimize reaction parameters for specific substrate variants.
Furthermore, the impurity profile of the reaction is tightly controlled through the specific design of the catalytic system. The use of 4A molecular sieves plays a dual role: it acts as a water scavenger to prevent hydrolysis of the sensitive o-quinone methide intermediate and enhances the reaction rate by shifting the equilibrium. The ligand structure, featuring a phosphoramidite backbone with specific aryl substitutions, provides the necessary steric bulk to suppress competing [4+2] cycloaddition pathways or polymerization side reactions. This results in a clean reaction mixture where the desired chiral oxygen-containing eight-membered ring is the predominant species, significantly reducing the burden on downstream purification. Such mechanistic clarity ensures that the process is robust enough for transfer from laboratory discovery to pilot plant operations without unexpected scalability issues.
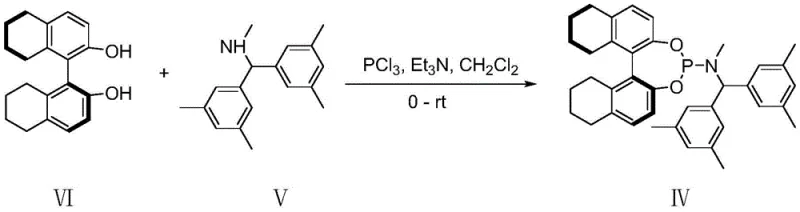
How to Synthesize Chiral Oxygen-Containing Eight-Membered Ring Compounds Efficiently
Implementing this synthesis requires careful attention to the preparation of the catalytic system and the handling of moisture-sensitive reagents. The process begins with the in situ generation of the active palladium catalyst by mixing the palladium source, such as Pd2(dba)3·CHCl3, with the chiral ligand in an anhydrous solvent. This pre-activation step is critical to ensure the formation of the correct catalytic species before the substrates are introduced. The reaction is then conducted under an inert atmosphere, typically nitrogen or argon, to prevent oxidation of the catalyst. Detailed standard operating procedures regarding stoichiometry, temperature ramps, and workup protocols are essential for reproducibility. For a comprehensive guide on the specific experimental conditions and purification techniques, please refer to the standardized synthesis steps outlined below.
- Prepare the palladium catalytic system by mixing Pd2(dba)3·CHCl3 and the specific chiral phosphoramidite ligand (Formula IV) in anhydrous ethylbenzene under inert gas.
- Combine the gamma-methylene-delta-valerolactone compound (I) and o-quinone methide (II) with 4A molecular sieves in a reaction flask.
- Transfer the catalyst mixture to the substrate flask and stir at -20°C for 10-24 hours, followed by silica gel column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial benefits that directly address the pain points of procurement managers and supply chain heads. The shift from extraction-based or multi-step linear syntheses to a convergent catalytic process fundamentally alters the cost structure of producing these high-value intermediates. By utilizing a catalyst system composed of inexpensive palladium sources and easily synthesizable ligands, the overall material costs are significantly reduced. Moreover, the high yields and selectivity mean that less raw material is wasted, and the need for expensive chiral resolution steps post-synthesis is eliminated. This efficiency translates into a more predictable and stable supply of critical building blocks for drug manufacturing, mitigating the risks associated with raw material volatility.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of costly purification stages and the use of cheap, commodity-grade starting materials. Since the reaction proceeds with high atom economy and minimal byproduct formation, the downstream processing costs are drastically simplified. The catalyst loading is low, and the ligand can be prepared from accessible precursors, avoiding the need for exotic or proprietary reagents that often inflate the price of specialty chemicals. This lean manufacturing approach ensures that the final cost of goods sold (COGS) for these chiral intermediates remains competitive, even at smaller production scales.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the broad substrate scope of this reaction, which tolerates various functional groups on both the lactone and the quinone methide components. This flexibility allows manufacturers to source alternative starting materials if specific aryl aldehydes or diols face shortages, without needing to revalidate an entirely new synthetic route. Additionally, the reaction conditions are mild and do not require extreme pressures or temperatures, reducing the dependency on specialized high-pressure equipment and allowing production in standard multipurpose reactors. This adaptability ensures that production schedules can be maintained consistently, reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram quantities is straightforward due to the homogeneous nature of the catalysis and the simplicity of the workup procedure. The use of common organic solvents like ethylbenzene facilitates solvent recovery and recycling, aligning with green chemistry principles and reducing environmental waste disposal costs. The high selectivity of the reaction minimizes the generation of hazardous waste streams associated with failed isomers or polymeric byproducts. Consequently, this method supports sustainable manufacturing practices, making it easier for facilities to comply with increasingly stringent environmental regulations while expanding capacity to meet global demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed cycloaddition technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the process capabilities. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing production workflows. For further technical specifications or custom synthesis requests, our team is available to provide detailed route feasibility assessments.
Q: What are the key advantages of this palladium-catalyzed method over traditional extraction?
A: This method offers significantly higher yields and enantioselectivity compared to natural product separation, which is often labor-intensive and low-yielding. It avoids the unfavorable enthalpy and entropy effects typical of traditional macrocyclization.
Q: Is the chiral ligand used in this process commercially viable for large-scale production?
A: Yes, the chiral ligand (Formula IV) is synthesized from readily available precursors like binaphthol derivatives and amines, ensuring cost-effectiveness and supply chain stability for industrial applications.
Q: What represents the primary impurity challenge in this [4+4] cycloaddition?
A: The primary challenge is controlling diastereoselectivity and enantioselectivity. The use of the specific phosphoramidite ligand and low temperature (-20°C) effectively suppresses side reactions, achieving dr > 20:1 and ee > 90%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Oxygen-Containing Eight-Membered Ring Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of accessing advanced synthetic technologies like the one described in CN113583015A to maintain a competitive edge in the pharmaceutical market. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped to handle sensitive palladium-catalyzed reactions under strict inert conditions, guaranteeing the delivery of products with stringent purity specifications. We leverage our rigorous QC labs to verify enantiomeric excess and diastereomeric ratios, ensuring that every batch of chiral oxygen-containing eight-membered ring compounds meets the highest quality standards required for clinical and commercial applications.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through the adoption of this efficient catalytic process. Our technical team is ready to conduct a Customized Cost-Saving Analysis tailored to your specific target molecules, identifying opportunities to streamline your synthesis and improve margins. We encourage you to contact our technical procurement team to request specific COA data for similar structures and discuss route feasibility assessments for your upcoming projects. By partnering with us, you gain access to a reliable network of chemical expertise dedicated to accelerating your drug development timeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
