Advanced Triarylmethane Synthesis: Scalable Iron-Catalyzed Routes for Commercial Manufacturing
Introduction to Next-Generation Triarylmethane Manufacturing
The landscape of fine chemical synthesis is undergoing a significant transformation, driven by the urgent need for sustainable, cost-effective, and scalable manufacturing processes. A pivotal development in this sector is detailed in patent CN101037375A, which introduces a robust methodology for the synthesis of triarylmethane and its derivatives. This technology represents a paradigm shift away from traditional reliance on precious metal catalysts and harsh reaction conditions, offering a pathway that aligns perfectly with modern green chemistry principles. By utilizing earth-abundant iron-based catalysts, this method addresses critical pain points in the supply chain, including raw material volatility and environmental compliance. For R&D directors and procurement strategists, understanding the nuances of this iron-catalyzed electrophilic aromatic substitution is essential for optimizing the production of key pharmaceutical intermediates and functional materials. The following analysis dissects the technical merits and commercial viability of this approach, highlighting its potential to redefine industry standards for reliability and efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of triarylmethane scaffolds has been plagued by significant technical and economic hurdles that hinder large-scale adoption. Traditional protocols often necessitate the use of corrosive strong acids or require high-pressure environments that demand specialized, capital-intensive reactor infrastructure. Furthermore, prior art frequently relies on noble metal catalysts, such as gold salts or iridium complexes, which not only inflate the bill of materials but also introduce complex downstream processing requirements for metal removal to meet stringent pharmaceutical purity specifications. These legacy methods often suffer from limited substrate scope, failing to accommodate electron-deficient aromatics without drastic drops in yield. Additionally, the use of toxic organic solvents and the generation of substantial hazardous waste streams pose severe environmental liabilities, complicating regulatory approval and increasing the total cost of ownership for manufacturers seeking to maintain a sustainable operation.
The Novel Approach
In stark contrast, the methodology disclosed in CN101037375A leverages a mild, Lewis acid-catalyzed system that operates efficiently under atmospheric pressure and moderate temperatures ranging from 20°C to 120°C. By employing ferric chloride or indium trichloride in the presence of acetic anhydride, this novel route achieves exceptional reaction specificity and high yields without the need for exotic reagents. The process demonstrates remarkable versatility, accommodating a wide array of substituted benzaldehydes and benzenes, including those with halogen, alkyl, and nitro functionalities. This flexibility allows for the precise construction of complex molecular architectures required in advanced drug discovery. The elimination of precious metals and the use of benign reaction conditions drastically simplify the workup procedure, reducing solvent consumption and waste generation. This streamlined approach not only enhances safety profiles but also significantly lowers the barrier to entry for commercial scale-up, making it an ideal candidate for reliable triarylmethane supplier networks aiming to optimize their production portfolios.
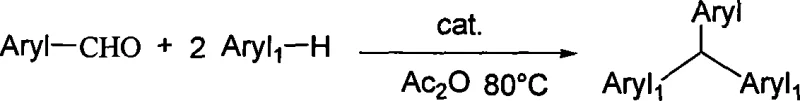
Mechanistic Insights into FeCl3-Catalyzed Electrophilic Substitution
The core of this technological breakthrough lies in the efficient activation of the carbonyl group via Lewis acid catalysis. In this mechanism, the iron trichloride catalyst coordinates with the oxygen atom of the substituted benzaldehyde, thereby increasing the electrophilicity of the carbonyl carbon. This activation facilitates a nucleophilic attack by the electron-rich aromatic ring of the substituted benzene, leading to the formation of a diarylmethanol intermediate or a direct carbocation species depending on the specific reaction conditions and the presence of acetic anhydride. The acetic anhydride likely serves a dual role: acting as a dehydrating agent to drive the equilibrium forward by removing water formed during the condensation, and potentially acetylating intermediate hydroxyl groups to form better leaving groups. This synergistic effect ensures that the reaction proceeds to completion even with less reactive substrates, such as nitro-substituted benzaldehydes, which typically resist Friedel-Crafts type alkylations. The result is a highly controlled transformation that minimizes side reactions like polymerization or over-alkylation, ensuring a clean impurity profile that is critical for downstream pharmaceutical applications.
Furthermore, the protocol offers a sophisticated strategy for controlling regioselectivity and synthesizing unsymmetrical derivatives, a common challenge in triarylmethane chemistry. By modulating the addition sequence of the aromatic components, chemists can direct the substitution pattern with high precision. For instance, reacting the aldehyde with one equivalent of a first arene followed by the introduction of a second, distinct arene allows for the construction of diverse molecular libraries. This stepwise control is vital for generating specific isomers required for structure-activity relationship (SAR) studies in medicinal chemistry. The robustness of the iron catalyst ensures that the reaction tolerates various functional groups without requiring extensive protecting group strategies, thereby shortening the overall synthetic timeline. This mechanistic elegance translates directly into operational simplicity, allowing manufacturing teams to reproduce high-purity results consistently across different batches and scales.
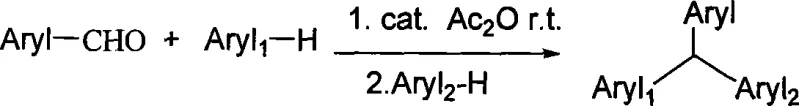
How to Synthesize Triarylmethane Derivatives Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and thermal management to maximize yield and purity. The process begins with the dissolution of the substituted benzaldehyde and the iron-based catalyst in a suitable halohydrocarbon solvent, followed by the controlled addition of acetic anhydride. Once the mixture is homogenized, the substituted benzene component is introduced, and the system is heated to the optimal temperature window, typically between 60°C and 90°C for most substrates. Reaction monitoring is essential, with completion times varying from 1 to 50 hours depending on the electronic nature of the substituents. Upon completion, the reaction mixture is concentrated, and the crude product is purified using standard chromatographic techniques to isolate the target triarylmethane derivative with purity exceeding 99%. For detailed operational parameters and specific embodiment data, please refer to the structured guide below.
- Combine substituted benzaldehyde, acetic anhydride, and iron trichloride catalyst in an organic solvent such as a halohydrocarbon.
- Add the substituted benzene compound to the mixture and heat the reaction system to a temperature between 20°C and 120°C.
- Maintain stirring for 1 to 50 hours, then concentrate the reaction mixture and purify the crude product via chromatography to obtain high-purity triarylmethane.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this iron-catalyzed methodology offers profound advantages that directly impact the bottom line and supply chain resilience. The substitution of expensive noble metal catalysts with commodity-grade iron salts results in a drastic reduction in raw material costs, freeing up capital for other critical R&D investments. Moreover, the availability of iron chloride and acetic anhydride in the global chemical market ensures a stable supply chain, mitigating the risks associated with the geopolitical volatility often seen in the sourcing of rare earth or precious metal catalysts. The mild reaction conditions also translate to lower energy consumption, as the process does not require extreme heating or high-pressure equipment, further contributing to operational expenditure savings. These factors combined create a compelling value proposition for procurement managers seeking to optimize the cost structure of fine chemical manufacturing without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The elimination of gold and iridium catalysts removes a significant cost driver from the bill of materials, while the use of inexpensive solvents and reagents further drives down the unit cost of production. The simplified workup procedure reduces the consumption of purification media and solvents, leading to substantial cost savings in waste disposal and material handling. Additionally, the high atom economy of the reaction minimizes the loss of valuable starting materials, ensuring that every kilogram of input contributes effectively to the final output. This economic efficiency makes the process highly competitive for large-scale commercial production of pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: By relying on widely available, non-strategic raw materials, manufacturers can secure long-term supply contracts with multiple vendors, reducing the risk of production stoppages due to material shortages. The robustness of the reaction conditions means that the process is less sensitive to minor variations in utility supplies, such as steam pressure or cooling water temperature, enhancing overall plant reliability. This stability is crucial for meeting the rigorous delivery schedules demanded by multinational pharmaceutical clients, ensuring a continuous flow of high-quality intermediates. The ability to source catalysts and solvents locally in many regions further shortens the logistics chain and reduces lead times for high-purity triarylmethane derivatives.
- Scalability and Environmental Compliance: The absence of corrosive acids and high-pressure requirements simplifies the engineering controls needed for scale-up, allowing for seamless transition from pilot plant to multi-ton production facilities. The reduced generation of hazardous waste aligns with increasingly stringent environmental regulations, lowering the compliance burden and associated fees. The use of iron, a non-toxic metal, simplifies the residual metal testing and clearance processes required for API manufacturing, accelerating the regulatory approval timeline. This environmental stewardship not only protects the ecosystem but also enhances the corporate reputation of the manufacturer as a responsible partner in the global supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific product pipelines. The answers cover catalyst selection, substrate compatibility, and purification strategies to ensure successful technology transfer.
Q: What are the advantages of using FeCl3 over noble metal catalysts for triarylmethane synthesis?
A: According to patent CN101037375A, using iron trichloride eliminates the need for expensive gold or iridium salts, significantly reducing raw material costs while maintaining high reaction specificity and yield under mild conditions.
Q: Can this method synthesize unsymmetrical triarylmethane derivatives?
A: Yes, the process allows for the sequential addition of different substituted benzene compounds, enabling the synthesis of diverse unsymmetrical triarylmethane structures with high selectivity.
Q: What is the typical purity achievable with this synthetic route?
A: Experimental data within the patent indicates that the final products can achieve purity levels greater than 99% after standard purification processes like silica gel chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triarylmethane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic routes to maintain competitiveness in the global fine chemicals market. Our technical team has extensively evaluated the iron-catalyzed triarylmethane synthesis described in CN101037375A and confirmed its potential for delivering high-value intermediates with exceptional purity and consistency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a steady supply of materials that meet the most rigorous quality standards. Our state-of-the-art facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch of triarylmethane derivative we produce is ready for immediate use in sensitive pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your desired compounds. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain partner dedicated to driving innovation and efficiency in the production of complex organic intermediates.
