Advanced Manufacturing of Chiral Sulfonamide-Aminoalcohol Ligands for Asymmetric Catalysis
The landscape of asymmetric synthesis is constantly evolving, driven by the demand for more efficient and sustainable chiral catalysts. Patent CN100584827C introduces a significant breakthrough in the field of chiral sulfonamide-aminoalcohol ligands, offering a robust solution for the asymmetric addition of diethylzinc to aldehydes. This technology leverages naturally occurring chiral pool materials, specifically L-prolinol, (-)-ephedrine, or (+)-pseudoephedrine, to construct complex ligand frameworks containing one to three chiral centers. The core innovation lies in the direct, selective ring-opening reaction of cyclic ethyleneimines (aziridines) with these amino alcohols. This approach not only streamlines the synthetic pathway but also ensures high atom economy and exceptional optical purity. For R&D directors and process chemists seeking reliable pharmaceutical intermediate suppliers, this methodology represents a paradigm shift from multi-step, low-yield processes to a concise, high-efficiency manufacturing route.
Historically, the synthesis of effective sulfonamide ligands for asymmetric catalysis has been plagued by inefficiencies and operational complexities. Prior art, such as the methods described in Tetrahedron: Asymmetry (1999) and J. Org. Chem. (1999), typically involves laborious multi-step sequences ranging from three to five distinct reaction stages. These conventional routes often suffer from low overall yields and necessitate purification via column chromatography, a technique that is notoriously difficult to scale and generates significant solvent waste. Furthermore, the application of these legacy ligands frequently demands the presence of titanium tetraisopropoxide (Ti(OiPr)4) as a critical additive to achieve acceptable catalytic activity. This requirement introduces severe handling challenges due to the reagent's sensitivity to moisture and air, often mandating cryogenic reaction conditions as low as -40°C to maintain stereocontrol.
In stark contrast, the novel approach detailed in the patent data offers a drastically simplified alternative that addresses these pain points directly. By utilizing a one-step ring-opening strategy, the new method bypasses the need for intermediate isolation and complex protection/deprotection sequences. The reaction proceeds smoothly under reflux in common organic solvents like acetonitrile, eliminating the need for extreme cooling. Perhaps most importantly for cost reduction in chiral ligand manufacturing, the final product is obtained through simple recrystallization rather than chromatography. This modification transforms the purification process from a bottleneck into a scalable unit operation. The resulting ligands are stable, easy to handle, and remarkably, they exhibit high catalytic efficiency in asymmetric diethylzinc additions without requiring any titanium additives, operating effectively even at room temperature.
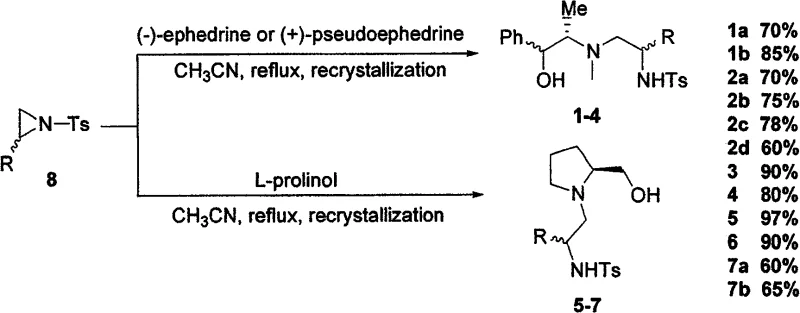
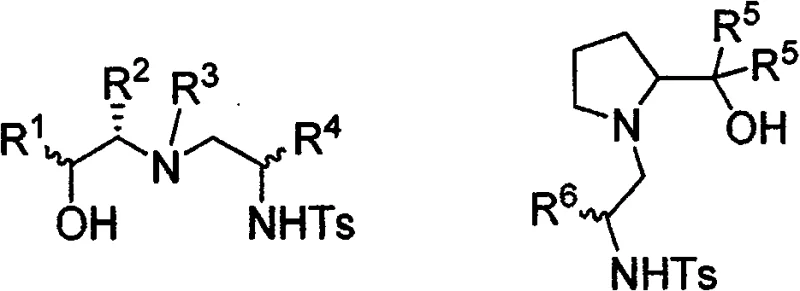
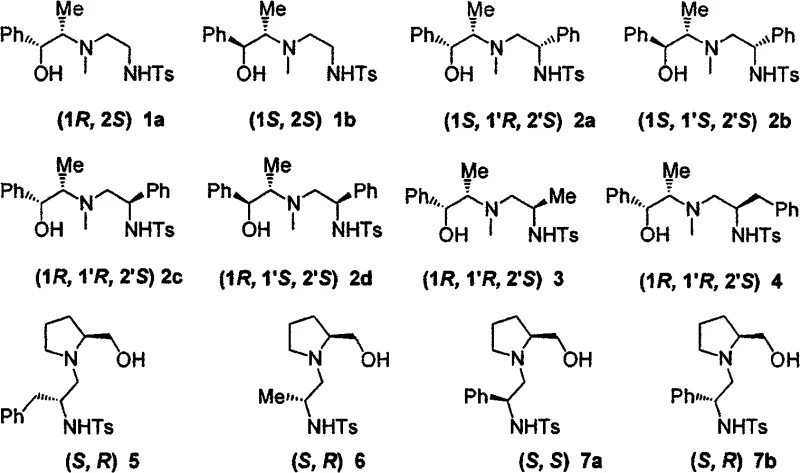
The mechanistic elegance of this system lies in the precise construction of the chiral environment around the nitrogen and oxygen donor atoms. The ligands feature a sulfonamide backbone coupled with a hydroxyl group, creating a bidentate coordination sphere that effectively complexes with zinc species. The structural diversity is achieved by varying the substituents on the aziridine ring and the starting amino alcohol, allowing for the fine-tuning of steric and electronic properties. As illustrated in the specific examples, variations in R groups (such as phenyl, benzyl, or methyl) allow chemists to optimize the catalyst for specific substrates. This modularity is crucial for achieving high enantioselectivity across a broad range of aldehydes, including sterically hindered aromatic aldehydes and reactive aliphatic variants. The ability to selectively produce products with opposite absolute configurations simply by switching the chirality of the starting material (e.g., ephedrine vs. pseudoephedrine) provides a powerful tool for accessing both enantiomers of a target drug intermediate.
To implement this synthesis in a laboratory or pilot plant setting, the procedure is remarkably straightforward and amenable to standard equipment. The process begins with the dissolution of the chiral amino alcohol and the tosyl-protected aziridine in a suitable solvent. Following the reaction period, the workup involves basic distillation and a crystallization step that yields high-purity solids suitable for immediate use in catalysis. For detailed operational parameters and safety considerations regarding the scale-up of this specific chemistry, please refer to the standardized protocol below.
For procurement managers and supply chain leaders, the commercial implications of this technology are profound. The elimination of column chromatography is not merely a technical improvement; it is a major economic driver. Chromatography is a batch process that limits throughput and consumes vast quantities of silica and solvents, driving up the cost of goods sold (COGS). By replacing this with recrystallization, the process becomes continuous-friendly and significantly reduces waste disposal costs. Furthermore, the removal of titanium tetraisopropoxide from the application phase simplifies the downstream processing of the final pharmaceutical active ingredient, as there is no need for rigorous metal scavenging steps to meet regulatory limits on heavy metals. This leads to substantial cost savings and a cleaner impurity profile.
Supply chain reliability is further enhanced by the use of commodity starting materials. L-prolinol and ephedrine are commercially available in bulk quantities from established natural product supply chains, reducing the risk of raw material shortages. The robustness of the reaction conditions—reflux in acetonitrile followed by ambient temperature crystallization—means that the manufacturing process is less susceptible to equipment failure or utility fluctuations compared to cryogenic processes. This stability ensures consistent delivery schedules and supports the commercial scale-up of complex pharmaceutical intermediates without the need for specialized low-temperature reactors. Additionally, the high atom economy of the ring-opening reaction aligns with modern green chemistry principles, facilitating easier environmental compliance and reducing the carbon footprint of the manufacturing facility.
Frequently asked questions regarding this technology often center on its versatility and ease of adoption. The following section addresses common inquiries from technical teams evaluating this ligand system for their specific synthetic pathways. These answers are derived directly from the experimental data and comparative studies presented in the patent documentation, providing a clear picture of the ligand's capabilities and limitations in real-world applications.
- Dissolve natural ephedrine, pseudoephedrine, or L-prolinol and the corresponding cyclic ethyleneimine (aziridine) in an organic solvent such as acetonitrile at a molar ratio of 1-1.5: 1.
- Heat the mixture under reflux conditions for 10 to 40 hours to facilitate the selective ring-opening reaction.
- Remove the solvent under reduced pressure and recrystallize the residue using a mixed solvent of dichloromethane and petroleum ether to obtain the chemically pure target ligand.
Frequently Asked Questions (FAQ)
Q: Does this ligand synthesis require titanium tetraisopropoxide additives?
A: No, unlike conventional sulfonamide ligands described in prior literature which require Ti(OiPr)4 for catalytic activity, the ligands disclosed in patent CN100584827C function effectively without this moisture-sensitive additive, simplifying the reaction setup.
Q: What is the substrate scope for these chiral ligands in asymmetric additions?
A: These ligands demonstrate a broad substrate scope, successfully catalyzing the asymmetric addition of diethylzinc to aromatic aldehydes, aliphatic aldehydes, and α,β-unsaturated aldehydes with high enantioselectivity.
Q: How is the purity of the final ligand achieved without column chromatography?
A: The process utilizes a highly efficient recrystallization step using a specific mixture of dichloromethane and petroleum ether, which eliminates the need for cumbersome and costly column chromatography purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Sulfonamide-Aminoalcohol Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance chiral ligands play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of ligand meets the exacting standards required for GMP synthesis. Our infrastructure is designed to support the rapid deployment of innovative chemistries like the one described in CN100584827C, providing our partners with a secure and scalable source of critical chiral building blocks.
We invite you to collaborate with us to leverage these advanced synthetic methodologies for your upcoming projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and process constraints. Please contact us to request specific COA data, route feasibility assessments, and samples for evaluation. Let us help you optimize your asymmetric synthesis workflows and reduce your time to market with our reliable supply of high-purity chiral intermediates.
