Revolutionizing (S)-Nicotine Synthesis: Advanced Biocatalytic Routes for Commercial Scale Production
Revolutionizing (S)-Nicotine Synthesis: Advanced Biocatalytic Routes for Commercial Scale Production
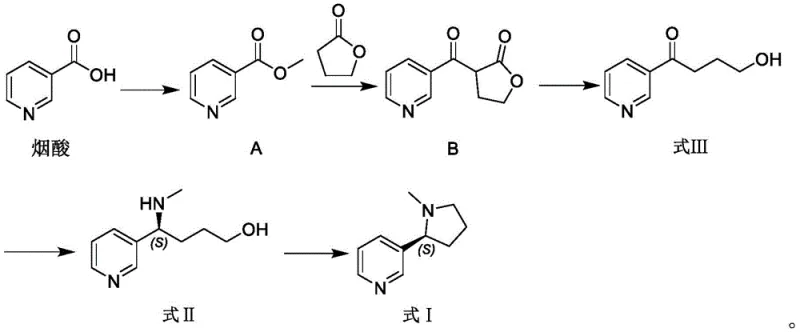
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways for producing high-value alkaloids, particularly (S)-nicotine, which serves as a critical active pharmaceutical ingredient (API) for smoking cessation therapies. A significant technological breakthrough in this domain is detailed in patent CN115404249A, which discloses a novel preparation method for an (S)-nicotine intermediate and its subsequent application in synthesizing (S)-nicotine. Unlike conventional strategies that rely heavily on tobacco extraction or complex chemical resolutions with limited scalability, this invention introduces a brand-new biocatalytic strategy. The core innovation lies in the direct enzymatic conversion of a ketone precursor (Formula III) into a chiral amine intermediate (Formula II) using methylamine, effectively bypassing the need for a separate, hazardous methylation process. This approach not only streamlines the synthetic route but also offers substantial improvements in operational safety, yield, and optical purity, positioning it as a superior alternative for reliable pharmaceutical intermediate suppliers aiming to optimize their manufacturing portfolios.
The strategic value of this patent extends beyond mere academic interest; it addresses critical bottlenecks in the global supply chain for nicotine-based therapeutics. By leveraging specific imine reductases and a coenzyme circulation system, the process achieves high conversion efficiencies under mild aqueous conditions. For procurement managers and supply chain heads, this translates to a potential reduction in dependency on volatile agricultural markets for tobacco leaf extraction and a move towards consistent, fermentation-based production. The ability to produce high-purity intermediates with 100% optical purity ensures that downstream processing is minimized, reducing waste and energy consumption. As we delve deeper into the technical specifics, it becomes evident that this biocatalytic route represents a paradigm shift in how complex chiral amines are manufactured, offering a robust framework for cost reduction in API manufacturing while adhering to stringent environmental and quality standards required by international regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of (S)-nicotine has been fraught with significant technical and economic challenges that hinder efficient commercial scale-up of complex pharmaceutical intermediates. The most prevalent method remains the extraction from tobacco leaves, which inherently ties production capacity to agricultural yields, seasonal variations, and the fluctuating quality of raw plant materials. This dependency creates substantial supply chain risks and limits the ability to respond rapidly to market demand surges. Furthermore, chemical synthesis methods reported in prior art, such as chemical resolution, asymmetric hydrogenation, or the use of chiral auxiliary reagents, often suffer from low atom economy and the generation of substantial hazardous waste. Specifically, existing biocatalytic approaches, such as those disclosed in patent US10913962B, typically involve a multi-step sequence where (S)-nornicotine is first produced and subsequently methylated using formaldehyde and formic acid. This additional methylation step introduces toxic reagents into the process, necessitates complex purification protocols to remove residual formaldehyde, and increases the overall cost of goods sold. The reliance on such indirect pathways underscores the urgent need for a more direct, safe, and reliable agrochemical intermediate or pharmaceutical synthesis strategy.
The Novel Approach
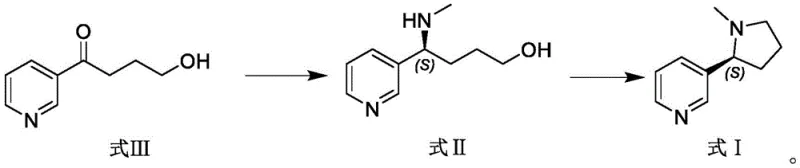
In stark contrast to these legacy methods, the technology described in CN115404249A pioneers a direct enzymatic amination strategy that fundamentally simplifies the molecular construction of the nicotine scaffold. The novel approach utilizes a specific class of imine reductases to catalyze the reductive amination of 4-hydroxy-1-(3-pyridyl)-1-butanone (Formula III) directly with methylamine. This single-step biocatalytic transformation installs both the amine functionality and the crucial methyl group simultaneously, effectively skipping the nornicotine intermediate stage entirely. The process operates in a buffered aqueous system at mild temperatures ranging from 15°C to 45°C and a neutral pH of 6.0 to 8.0, which drastically reduces energy consumption compared to high-temperature chemical refluxes. Moreover, the integration of a coenzyme recycling system involving glucose dehydrogenase ensures that expensive cofactors like NADP are regenerated in situ, making the process economically viable for large-scale operations. This streamlined workflow not only enhances the overall yield but also significantly improves the safety profile of the manufacturing process by eliminating the need for handling volatile and toxic methylating agents like methyl iodide or formaldehyde in the critical chirality-setting step.
Mechanistic Insights into Imine Reductase-Catalyzed Asymmetric Amination
The heart of this innovative synthesis lies in the precise mechanistic action of the engineered imine reductases, which exhibit remarkable stereoselectivity towards the prochiral ketone substrate. The patent highlights that imine reductases comprising amino acid sequences SEQ ID No. 2 through SEQ ID No. 9 are particularly effective, with SEQ ID No. 2, 4, and 5 demonstrating superior catalytic efficiency. Mechanistically, these enzymes facilitate the formation of an imine intermediate between the ketone carbonyl of Formula III and methylamine, which is then stereoselectively reduced by the hydride transfer from the reduced cofactor NADPH. The enzyme's active site architecture rigidly constrains the substrate orientation, ensuring that hydride delivery occurs exclusively from one face of the planar imine bond, thereby yielding the (S)-configured amine with exceptional enantiomeric excess. Experimental data within the patent confirms that specific variants, such as the enzyme corresponding to SEQ ID No. 5, can achieve conversion rates exceeding 83%, far surpassing the negligible activity observed in non-optimized enzymes like SEQ ID No. 8. This high level of catalytic proficiency is further bolstered by the coenzyme circulation system, where glucose dehydrogenase oxidizes glucose to gluconolactone, regenerating NADPH from NADP+, thus driving the equilibrium towards product formation without the need for stoichiometric amounts of expensive cofactors.
From an impurity control perspective, this biocatalytic mechanism offers distinct advantages over chemical catalysis. Traditional metal-catalyzed asymmetric hydrogenation often leaves trace amounts of heavy metals in the final product, necessitating costly and time-consuming scavenging steps to meet pharmaceutical purity specifications. In contrast, the enzymatic process operates under metal-free conditions, inherently producing a cleaner crude reaction mixture. The specificity of the enzyme minimizes the formation of regioisomers or over-reduced byproducts that are common in non-selective chemical reductions. Furthermore, the subsequent cyclization step, which converts the chiral intermediate (Formula II) into (S)-nicotine (Formula I) using chlorinating agents like thionyl chloride or methanesulfonyl chloride, proceeds with high fidelity due to the high purity of the enzymatic precursor. The patent reports optical purity levels of up to 100% for the intermediate and 96-97% for the final nicotine product, demonstrating that the biocatalytic step effectively sets the stereochemistry without erosion in downstream processing. This robust impurity profile simplifies the purification workflow, allowing for high-purity pharmaceutical intermediates to be obtained with fewer unit operations.
How to Synthesize (S)-Nicotine Intermediate Efficiently
Implementing this biocatalytic route requires a systematic approach to reaction engineering and enzyme management to ensure consistent quality and yield. The process begins with the preparation of the ketone precursor (Formula III) from readily available nicotinic acid, followed by the critical enzymatic amination step where reaction parameters such as pH, temperature, and substrate loading must be tightly controlled to maximize enzyme turnover. The final cyclization step requires careful handling of chlorinating agents to ensure complete ring closure while maintaining the integrity of the chiral center. For R&D teams looking to adopt this technology, understanding the interplay between enzyme stability and substrate solubility is paramount. The patent provides detailed embodiments showing that the reaction can be scaled from milligram screening assays to multi-gram preparative runs with consistent results, indicating strong potential for industrial translation. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these high-efficiency results.
- Synthesize the ketone precursor (Formula III) from nicotinic acid via esterification and acylation with gamma-butyrolactone.
- Perform asymmetric reductive amination of Formula III using methylamine and specific imine reductases (e.g., SEQ ID No. 2, 4, 5) with coenzyme recycling.
- Execute intramolecular cyclization of the chiral intermediate (Formula II) using a chlorinating agent followed by pH adjustment to yield (S)-nicotine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this biocatalytic technology presents a compelling value proposition centered around risk mitigation and long-term cost efficiency. The shift from extraction-dependent or chemically intensive processes to a fermentation-based enzymatic route decouples production from agricultural volatility, ensuring a more predictable and stable supply of critical intermediates. This reliability is essential for maintaining continuous manufacturing lines for smoking cessation products, where interruptions can have significant market consequences. Furthermore, the simplified synthetic route reduces the number of processing units required, lowering capital expenditure (CAPEX) for new facilities and reducing the operational footprint of existing plants. The elimination of hazardous methylation reagents also translates to lower costs associated with environmental compliance, waste disposal, and worker safety protocols, contributing to a more sustainable and economically resilient supply chain.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the consolidation of synthetic steps and the use of inexpensive feedstocks. By directly utilizing methylamine instead of a multi-step methylation sequence involving formaldehyde and reducing agents, the process reduces raw material costs and minimizes solvent usage. Additionally, the high conversion rates and optical purity achieved by the specific imine reductases reduce the need for expensive chiral chromatography or recrystallization steps to upgrade enantiomeric excess. The coenzyme recycling system further lowers operational costs by minimizing the consumption of NADP, a traditionally expensive reagent. These factors collectively contribute to a significantly lower cost of goods sold (COGS), allowing manufacturers to offer competitive pricing while maintaining healthy margins in the highly regulated pharmaceutical market.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved through the use of recombinant enzymes and commodity chemical precursors. Unlike tobacco extraction, which is subject to crop failures and geopolitical trade restrictions, the enzymes used in this process are produced via fermentation in controlled bioreactors, ensuring a consistent and scalable supply. The precursor, nicotinic acid, is a bulk chemical available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the enzymatic reaction under mild conditions also means that manufacturing can be distributed across different geographic locations without requiring specialized high-pressure or high-temperature infrastructure. This flexibility allows for a more agile supply chain capable of adapting to regional demand fluctuations and mitigating logistics disruptions.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with patent examples demonstrating successful reactions in multi-liter reactors using standard stirred-tank equipment. The aqueous nature of the biocatalytic step reduces the reliance on volatile organic compounds (VOCs), aligning with increasingly stringent environmental regulations regarding solvent emissions. The absence of heavy metal catalysts eliminates the need for complex metal removal steps and reduces the toxicity of process waste streams. This green chemistry profile not only facilitates easier regulatory approval but also enhances the corporate sustainability credentials of the manufacturer. The ability to scale from pilot to commercial production without significant process re-engineering ensures a faster time-to-market for new generic formulations or proprietary derivatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic synthesis route. These insights are derived directly from the experimental data and claims presented in patent CN115404249A, providing a factual basis for decision-making. Understanding these nuances is crucial for technical teams evaluating the feasibility of integrating this technology into their existing production workflows. The answers highlight the specific advantages of the enzymatic approach over traditional methods, focusing on yield, purity, and operational safety.
Q: What is the primary advantage of this biocatalytic method over traditional chemical synthesis?
A: The primary advantage is the elimination of a separate methylation step. Traditional methods often require synthesizing nornicotine first and then methylating it with formaldehyde, which involves hazardous reagents. This patent utilizes a specific imine reductase to directly introduce the methyl group from methylamine during the reduction step, significantly simplifying the workflow and improving safety.
Q: Which enzymes are most effective for this transformation according to the patent data?
A: The patent identifies specific imine reductases with amino acid sequences SEQ ID No. 2, SEQ ID No. 4, and SEQ ID No. 5 as highly efficient. These variants demonstrated conversion rates exceeding 55%, with some reaching over 80%, significantly outperforming other tested enzymes like SEQ ID No. 8 and 9 which showed negligible activity.
Q: How does this process address the issue of optical purity in (S)-nicotine production?
A: The process achieves exceptional stereocontrol through the use of engineered imine reductases. Experimental data in the patent indicates that the resulting intermediate (Formula II) can achieve 100% optical purity (ee value), which is critical for pharmaceutical applications where the (S)-enantiomer is the biologically active form required for smoking cessation therapies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-Nicotine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the biocatalytic routes described in CN115404249A and are fully equipped to bring this advanced chemistry to commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We understand that high-purity pharmaceutical intermediates require rigorous quality control; therefore, our state-of-the-art QC labs and stringent purity specifications guarantee that every batch of (S)-nicotine intermediate meets the exacting standards required by global regulatory agencies. Whether you require custom enzyme engineering support or large-scale GMP manufacturing, our infrastructure is designed to support your most complex synthesis challenges with precision and reliability.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this biocatalytic route can optimize your budget. Please contact us today to request specific COA data for our pilot batches and comprehensive route feasibility assessments. Let us partner with you to secure a sustainable, cost-effective, and high-quality supply of (S)-nicotine intermediates for the global market.
