Advanced Synthetic Route for NL-101 Benzimidazole Derivatives Enabling Safe Commercial Scale-Up
Advanced Synthetic Route for NL-101 Benzimidazole Derivatives Enabling Safe Commercial Scale-Up
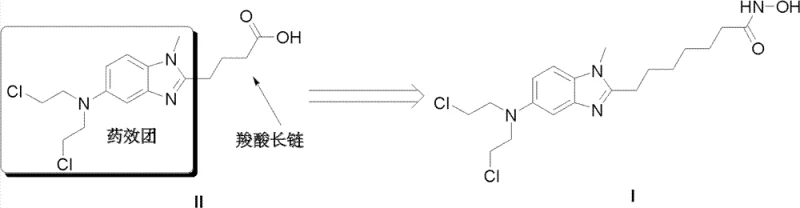
The pharmaceutical industry is constantly seeking robust manufacturing pathways for complex antitumor agents, particularly those targeting histone deacetylase (HDAC). Patent CN102993102A discloses a groundbreaking synthetic method for [1-methyl-2-(8'-octyl hydroxamic acid group)-5-N,N-bi(2'-chloroethyl)]-1H-benzimidazole, commonly referred to as NL-101. This compound represents a strategic structural modification of bendamustine, designed to combine DNA alkylating properties with HDAC inhibition activity similar to vorinostat. The core innovation lies in replacing the hazardous ethylene oxide reagent found in prior art with chloroacetic acid, thereby fundamentally altering the safety profile and operational feasibility of the synthesis. This technical advancement addresses critical pain points regarding process safety, environmental compliance, and scalability, offering a viable pathway for the reliable pharmaceutical intermediate supplier market to produce high-value oncology candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those described in WO 2010085377, rely heavily on the use of ethylene oxide to introduce the nitrogen mustard functionality. Ethylene oxide is a colorless, flammable gas with a boiling point of merely 10.73°C, presenting severe logistical and safety challenges. In an industrial setting, transferring this reagent requires cooling it to a liquid state, and any accidental temperature rise above its boiling point during transfer can lead to rapid volume expansion and potential explosion hazards. Furthermore, ethylene oxide is highly toxic, posing significant risks to worker health and requiring stringent containment measures that drive up capital expenditure. The conventional seven-step synthesis is not only lengthy but also suffers from poor atom economy and difficult temperature control, making it unsuitable for large-scale commercial production where safety and consistency are paramount.
The Novel Approach
The patented methodology introduces a streamlined, safer alternative by utilizing chloroacetic acid as the starting material for the nitrogen mustard side chain. This substitution eliminates the need for handling volatile, explosive gases, allowing reactions to proceed under standard atmospheric pressure and manageable temperatures. The new route condenses the synthesis into fewer operational steps, often employing one-pot multi-reaction strategies that minimize intermediate isolation and purification losses. By integrating amidation, catalytic hydrogenation, and reductive alkylation into a cohesive workflow, the process significantly reduces solvent consumption and waste generation. This approach not only enhances the green chemistry profile of the manufacturing process but also ensures that the reaction environment remains stable and controllable, facilitating a smoother transition from laboratory bench to pilot plant and full-scale production.
Mechanistic Insights into Catalytic Cyclization and Reductive Alkylation
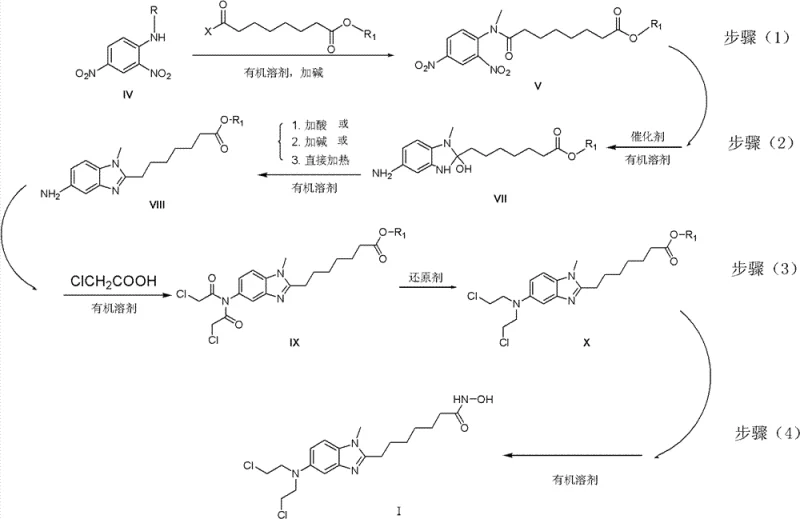
The heart of this synthetic strategy involves a sophisticated sequence of catalytic transformations. The initial step entails the amidation of 2,4-dinitroaniline derivatives with suberic acid monoesters or their acylates under alkaline conditions, forming a stable nitro-amide intermediate. This is followed by a critical catalytic hydrogenation step using palladium on carbon (Pd/C) catalysts. Unlike simple nitro reductions, this step is engineered to facilitate simultaneous cyclization. Under controlled hydrogen pressure (1 to 7 atm) and temperatures ranging from 0°C to 60°C, the nitro group is reduced to an amine, which immediately undergoes intramolecular condensation with the adjacent amide carbonyl to form the benzimidazole ring system. The choice of solvent, typically methanol or ethanol, plays a crucial role in stabilizing the transition states and ensuring high conversion rates without over-reduction of sensitive functional groups.
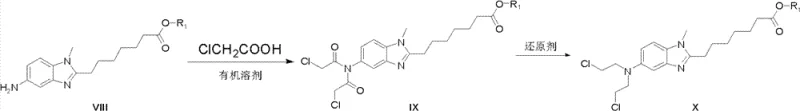
Subsequent functionalization involves the installation of the bis(2-chloroethyl)amino group, a key pharmacophore for DNA alkylation. This is achieved by reacting the amino-benzimidazole intermediate with chloroacetic acid to form a bis-chloroacetamide, followed by a selective reduction using borane-tetrahydrofuran (borane-THF) complexes. This reduction step is mechanistically distinct as it converts the amide carbonyls directly to methylene groups, effectively reconstructing the nitrogen mustard motif without exposing the molecule to harsh conditions that could degrade the hydroxamic acid precursor chain. The use of borane reagents allows for precise control over the reduction potential, minimizing side reactions such as dehalogenation. Finally, the terminal ester is converted to the hydroxamic acid via nucleophilic attack by hydroxylamine, completing the dual-acting pharmacophore structure essential for HDAC inhibition.
How to Synthesize NL-101 Benzimidazole Derivatives Efficiently
The synthesis of NL-101 requires precise control over reaction parameters to maximize yield and purity while maintaining safety standards. The process begins with the preparation of the nitro-amide backbone, followed by the pivotal cyclization and side-chain installation. Operators must pay close attention to the stoichiometry of the reducing agents and the pH levels during workup to prevent hydrolysis of the sensitive ester or hydroxamic acid moieties. The following guide outlines the standardized operational framework derived from the patent examples, ensuring reproducibility across different manufacturing scales.
- Perform amidation of 2,4-dinitroaniline with suberic acid monoester derivatives under alkaline conditions to form the nitro-amide intermediate.
- Execute catalytic hydrogenation using Pd/C followed by acid or base mediated dehydration to achieve cyclization into the benzimidazole core.
- React the amino-benzimidazole intermediate with chloroacetic acid and reduce the resulting amide using borane-THF to install the nitrogen mustard moiety.
- Convert the terminal ester to the hydroxamic acid functionality using hydroxylamine or O-(tetrahydropyran-2-yl)hydroxylamine to yield the final NL-101 product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from ethylene oxide-based chemistry to chloroacetic acid-based synthesis represents a substantial opportunity for cost reduction in API manufacturing. The elimination of cryogenic infrastructure and high-pressure gas handling systems drastically lowers the barrier to entry for contract manufacturing organizations (CMOs). This process optimization translates directly into lower overhead costs, as facilities do not require specialized explosion-proof zones or complex gas scrubbing systems associated with toxic volatile organics. Furthermore, the use of commodity chemicals like chloroacetic acid and common solvents such as THF and methanol ensures a stable and resilient supply chain, mitigating the risk of raw material shortages that often plague specialty gas markets.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous ethylene oxide with inexpensive chloroacetic acid significantly lowers the raw material cost base. Additionally, the shortened synthetic route reduces the number of unit operations, leading to decreased labor costs, lower energy consumption for heating and cooling, and reduced solvent waste disposal fees. The ability to perform multiple transformations in a single pot further enhances efficiency by minimizing intermediate isolation steps, which are typically the most resource-intensive parts of a chemical process.
- Enhanced Supply Chain Reliability: By relying on stable, non-volatile solid and liquid reagents, the manufacturing process becomes far less susceptible to logistical disruptions. Chloroacetic acid is a widely available bulk chemical with a robust global supply network, unlike ethylene oxide which requires specialized transport and storage. This stability ensures consistent production schedules and reliable delivery timelines for downstream pharmaceutical clients, reducing the risk of project delays due to raw material availability issues.
- Scalability and Environmental Compliance: The improved safety profile of the new route facilitates easier regulatory approval for commercial scale-up of complex pharmaceutical intermediates. The absence of highly toxic gases simplifies environmental impact assessments and reduces the burden of emissions monitoring. Moreover, the process generates less hazardous waste, aligning with modern green chemistry principles and helping manufacturers meet increasingly stringent environmental regulations without costly retrofitting of existing production lines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of NL-101 and related benzimidazole derivatives. These insights are derived directly from the experimental data and process descriptions within the patent documentation, providing clarity on the feasibility and advantages of this novel synthetic approach for industry stakeholders.
Q: What is the primary safety advantage of the new NL-101 synthesis method?
A: The novel method replaces highly toxic and explosive ethylene oxide (boiling point 10.73°C) with stable, solid chloroacetic acid, eliminating the need for cryogenic storage and high-pressure gas handling.
Q: What purity levels can be achieved with this synthetic route?
A: Analytical characterization indicates that the crude NL-101 product achieves a purity of approximately 95%, which can be further refined to greater than 99% through standard recrystallization processes.
Q: How does this process improve supply chain reliability for HDAC inhibitors?
A: By utilizing common organic solvents like THF and methanol and avoiding specialized high-pressure equipment for gaseous reagents, the process allows for easier scale-up in standard multipurpose chemical reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable NL-101 Supplier
At NINGBO INNO PHARMCHEM, we understand the critical importance of safety and efficiency in the production of potent antitumor intermediates like NL-101. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to market-ready supply is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch meets the highest standards required for clinical and commercial applications. Our commitment to process innovation allows us to offer competitive pricing without compromising on quality or safety.
We invite pharmaceutical partners to collaborate with us on the development and supply of this advanced HDAC inhibitor intermediate. By leveraging our technical expertise, we can provide a Customized Cost-Saving Analysis tailored to your specific project needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us support your pipeline with reliable, high-quality chemical solutions.
