Advanced Biocatalytic Reductive Amination for Scalable Secondary Amine Manufacturing
Advanced Biocatalytic Reductive Amination for Scalable Secondary Amine Manufacturing
The landscape of fine chemical synthesis is undergoing a paradigm shift driven by the urgent need for sustainable and highly selective manufacturing processes. Patent CN113583992A introduces a groundbreaking advancement in this sector by disclosing a novel reductive amination enzyme, designated as RsRedAm, derived from the strain Rhizobium sophorae (Accession No. ACCC 19914). This biocatalyst addresses critical limitations in the synthesis of secondary amines, which are indispensable building blocks for active pharmaceutical ingredients (APIs) and agrochemicals. Unlike traditional chemical methods that often rely on harsh conditions and precious metal catalysts, this enzymatic approach leverages the inherent specificity of biological systems to achieve high conversion rates under mild physiological conditions. For R&D directors and process chemists, this technology represents a significant opportunity to streamline synthetic routes, reduce impurity profiles, and enhance the overall sustainability of amine production workflows.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chemical reductive amination typically involves the reaction of a ketone with an amine in the presence of a reducing agent such as sodium cyanoborohydride or hydrogen gas over a heterogeneous metal catalyst like palladium or platinum. These conventional pathways are fraught with significant operational challenges and safety hazards that complicate large-scale manufacturing. The use of high-pressure hydrogenation requires specialized equipment and rigorous safety protocols to mitigate explosion risks, while chemical reducing agents often generate substantial quantities of toxic waste streams that require costly disposal procedures. Furthermore, chemical catalysts frequently lack the stereo- and chemoselectivity required for complex molecule synthesis, leading to the formation of unwanted by-products such as tertiary amines or over-reduced species, which necessitates extensive and yield-loss-inducing purification steps to meet the stringent purity specifications demanded by the pharmaceutical industry.
The Novel Approach
In stark contrast, the biocatalytic strategy outlined in the patent utilizes the RsRedAm enzyme to facilitate the direct reductive amination of cyclohexanone with a diverse array of amine donors in an aqueous buffer system. This biological route operates at ambient temperatures ranging from 20°C to 30°C and atmospheric pressure, effectively eliminating the need for energy-intensive heating or high-pressure reactors. The enzyme exhibits remarkable stability and activity, capable of driving reactions to near-complete conversion within 24 hours without the rapid inactivation observed in previously reported reductive aminases at elevated temperatures. By employing a whole-cell or crude enzyme system coupled with a glucose dehydrogenase cofactor regeneration cycle, the process ensures economic viability by minimizing the consumption of expensive NADPH cofactors, thereby offering a greener, safer, and more cost-effective alternative for the production of high-value secondary amine intermediates.
Mechanistic Insights into RsRedAm-Catalyzed Reductive Amination
The catalytic mechanism of the RsRedAm enzyme relies on its dependency on the nicotinamide adenine dinucleotide phosphate (NADPH) cofactor to drive the reduction of the imine intermediate formed between the ketone and the amine donor. The enzyme facilitates the nucleophilic attack of the amine on the carbonyl carbon of cyclohexanone, followed by the stereoselective transfer of a hydride ion from NADPH to the resulting iminium ion, yielding the chiral secondary amine product. To maintain catalytic turnover without the prohibitive cost of stoichiometric cofactor addition, the system integrates a glucose dehydrogenase (GDH) auxiliary enzyme. This coupled system oxidizes glucose to gluconolactone, simultaneously regenerating NADPH from NADP+, thus creating a self-sustaining catalytic cycle that allows for high substrate loading and efficient product formation. The kinetic data indicates that the enzyme maintains high specific activity across a broad spectrum of amine substrates, demonstrating its versatility as a biocatalyst for diverse chemical libraries.
Control of impurities is a critical advantage of this enzymatic pathway, particularly regarding the suppression of side reactions common in chemical synthesis. The high regioselectivity of the RsRedAm active site prevents the over-alkylation of the primary amine product to form tertiary amines, a frequent issue in non-enzymatic reductive aminations. Additionally, the mild pH conditions (approximately pH 8.5) and low reaction temperature minimize the risk of substrate degradation or polymerization, which can occur under the acidic or basic conditions often required for chemical imine formation and reduction. The downstream processing is further simplified by the solubility characteristics of the products; the patent describes an efficient isolation protocol involving pH-swing extraction, where the product is selectively partitioned into the organic phase after basification, leaving behind unreacted hydrophilic impurities and enzyme proteins in the aqueous phase, thereby ensuring a high-purity final product suitable for sensitive pharmaceutical applications.
How to Synthesize N-alkyl Cyclohexylamines Efficiently
The synthesis of N-alkyl substituted cyclohexylamines using the RsRedAm biocatalyst involves a streamlined workflow that begins with the preparation of the recombinant expression host. The gene encoding the enzyme is cloned into a standard expression vector such as pET28a and transformed into E. coli BL21(DE3) cells, which are then cultured in rich media to maximize biomass accumulation. Upon reaching the optimal optical density, protein expression is induced using IPTG at low temperatures to ensure proper folding and solubility of the enzyme. The resulting wet cells or crude lysate can be directly employed in the biotransformation reaction containing cyclohexanone, the desired amine donor, glucose, and the GDH cofactor regeneration system. This robust protocol allows for the rapid generation of secondary amines with high conversion rates, and the detailed standardized synthetic steps for implementing this technology in your laboratory are provided in the guide below.
- Clone the target gene (SEQ ID No. 1) from Rhizobium sophorae genomic DNA using specific primers with Nde I and Xho I restriction sites into a pET28a vector.
- Transform the recombinant plasmid into E. coli BL21(DE3) competent cells and culture in TB medium containing kanamycin until OD600 reaches 0.6-0.8.
- Induce expression with IPTG at 16°C for 16-24 hours, harvest cells, and lyse via high-pressure homogenization to obtain the crude enzyme solution.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this enzymatic technology offers compelling strategic advantages that extend beyond mere technical performance. The transition from metal-catalyzed chemical synthesis to biocatalysis fundamentally alters the cost structure and risk profile of the supply chain. By eliminating the reliance on precious metals like palladium or platinum, manufacturers can insulate themselves from the volatile price fluctuations associated with these commodities. Furthermore, the removal of heavy metal catalysts obviates the need for expensive and time-consuming metal scavenging steps during purification, which significantly reduces the consumption of specialized resins and solvents. This simplification of the downstream process not only lowers the direct cost of goods sold but also shortens the overall production cycle time, allowing for faster response to market demands and improved inventory turnover rates for critical amine intermediates.
- Cost Reduction in Manufacturing: The implementation of the RsRedAm enzyme system drives cost efficiency through multiple mechanisms, primarily by operating under ambient conditions that drastically reduce energy consumption compared to high-temperature and high-pressure chemical processes. The use of glucose as a cheap and renewable co-substrate for cofactor regeneration replaces expensive stoichiometric reducing agents, leading to substantial savings in raw material costs. Additionally, the high selectivity of the enzyme minimizes the formation of by-products, which increases the overall yield of the desired secondary amine and reduces the waste disposal costs associated with purifying complex reaction mixtures. These factors combine to create a leaner manufacturing process with a significantly lower environmental footprint and reduced operational expenditure.
- Enhanced Supply Chain Reliability: Biological catalysts offer a level of supply chain security that chemical catalysts cannot match, as they are produced via fermentation using renewable feedstocks rather than being mined or synthesized from finite resources. The recombinant expression system described in the patent utilizes standard E. coli strains and commercially available vectors, meaning that the enzyme production can be easily scaled up or replicated at different geographic locations to mitigate regional supply disruptions. The stability of the freeze-dried enzyme powder allows for long-term storage and transportation without the need for cold chain logistics, ensuring that production facilities can maintain a reliable inventory of the biocatalyst to support continuous manufacturing operations without interruption.
- Scalability and Environmental Compliance: Scaling biocatalytic processes is inherently safer and more compliant with increasingly stringent environmental regulations. The aqueous nature of the reaction medium reduces the volume of volatile organic compounds (VOCs) emitted during synthesis, facilitating easier compliance with air quality standards. The waste streams generated are primarily biological in nature and are more readily biodegradable than those containing heavy metals or cyanide residues from chemical reduction. This environmental compatibility simplifies the permitting process for new manufacturing lines and reduces the liability associated with hazardous waste management, making the technology an attractive option for companies aiming to meet corporate sustainability goals and green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of the RsRedAm reductive amination technology. These insights are derived directly from the experimental data and embodiments disclosed in the patent documentation, providing a clear understanding of the enzyme's capabilities and limitations. Understanding these details is crucial for process engineers and technical buyers who are evaluating the feasibility of integrating this biocatalytic route into their existing production infrastructure. The answers cover aspects ranging from substrate compatibility to reaction optimization, ensuring that stakeholders have the necessary information to make informed decisions about adopting this innovative synthesis method.
Q: What is the substrate scope of the RsRedAm enzyme?
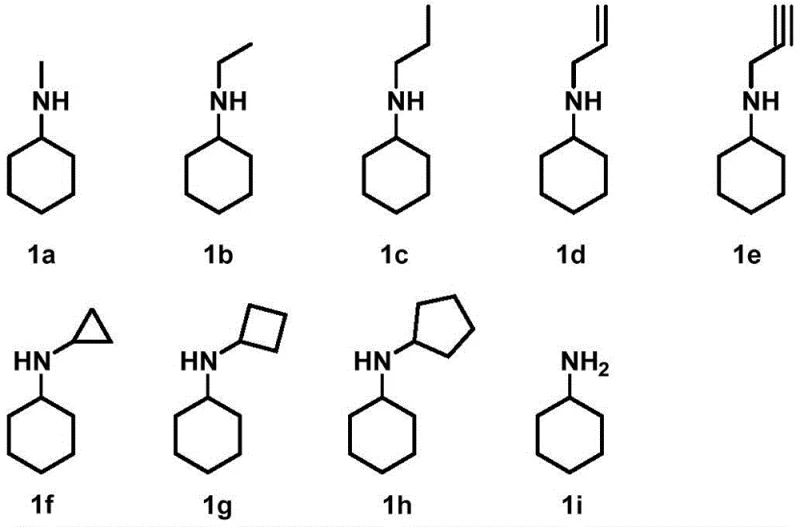
A: The RsRedAm enzyme demonstrates broad substrate tolerance, effectively catalyzing the reductive amination of cyclohexanone with various amine donors including methylamine, ethylamine, propylamine, allylamine, and propargylamine, as well as cyclic amines like cyclopropylamine and cyclobutylamine.
Q: How is the cofactor NADPH regenerated in this process?
A: The process utilizes a coupled enzyme system where glucose dehydrogenase (GDH) oxidizes glucose to regenerate NADPH from NADP+, ensuring a sustainable catalytic cycle without the need for stoichiometric amounts of expensive cofactors.
Q: What are the optimal reaction conditions for this biocatalyst?
A: The enzyme operates optimally at mild temperatures between 20°C and 30°C in a Tris-HCl buffer at pH 8.5, significantly reducing energy consumption compared to traditional thermal chemical processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Secondary Amine Supplier
As the demand for high-purity pharmaceutical intermediates continues to grow, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge biocatalytic technologies such as the RsRedAm enzyme system. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of secondary amine meets the exacting standards required for drug substance synthesis. Our commitment to quality and consistency makes us a trusted partner for global pharmaceutical companies seeking to secure their supply chains with robust and sustainable manufacturing solutions.
We invite you to contact our technical procurement team to discuss how this enzymatic reductive amination technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits of switching to this biocatalytic route. We encourage you to reach out for specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in enzyme engineering and process optimization can accelerate your development timelines and reduce your overall manufacturing costs.
